Liquid-Liquid Extraction of V(IV) from Sulphate Medium by Cyanex 301 Dissolved in Kerosene ()
1. Introduction
Vanadium is widely used to prepare ferro-vanadium and the oxidative catalyst, V2O5. Almost 90% vanadium is used to prepare ferro-vanadium (0.1% - 0.3% V) for automobile parts and high speed tools. Besides its use as oxidative catalyst, vanadium compounds are used in ink, dye, paint and varnish, insecticide, photographic chemicals, medicine and glass industries [1]. From carnotite (K2U2V2O11∙3H2O), it is extracted with uranium by the DAPEX process. It is also extracted from vanadinite (3Pb3(VO4)2∙PbCl2). But in the present world, these ores together with patronite (V2S3) are rare on the earth’s crust. So to meet up the demand, the processing of low grade ores and waste materials (tar sand, waste desulphurization catalyst, waste SO2-oxidation catalyst of contact process, slag, ash, rock etc.) are desirable. Prior roasting and leaching [2,3] followed by solvent extraction is convenient for such purpose. It can build up concentration by using low organic to aqueous phase ratio (O/A) in extraction and high O/A in stripping.
Works published before 1976 on solvent extraction of V(IV) by various extractants have been summarized by Sekine and Hasegawa [4]. D2EHPA is a promising extractant for V(IV) and V(V) [5-11]. Saji and Reddy [12] have reported the extractions of V(IV) and V(V) by EHEHPA. In the field of solvent extraction, a delayed development is the use of organo-phosphinic compounds (Cyanex reagents) introduced by American Cyanamid and Cytec Canada Inc. Cyanex 302 and Cyanex 301 are mono-and di-sulphide analogues of Cyanex 272 (bis- 2,4,4-trimethylpentylphosphinic acid). The sulphur substitution decreases the pKa values (6.4, 5.6 and 2.6 for Cyanex 272 [13], Cyanex 302 [14] and Cyanex 301 [15], respectively) permitting to work at lower pH [16]. Cyanex reagents differ from other commercial organophosphorous reagents (e.g. D2EHPA, DDPA, TBP, EHEHPA etc.) in that the former reagents contain P-C bonding, whereas the latter reagents contain P-O-C bonding. The presence of P-C bonding in Cyanex reagents renders them to be less susceptible to hydrolysis and less soluble in water [17].
In recent past, the extraction behavior of V(IV) from 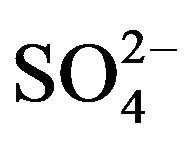 medium by Cyanex 272 [18,19] and by Cyanex 302 [20] and of V(IV) and V(V) in presence of Mo(VI), W(VI), U(VI), Ti(IV), Al(III), Cr(III), Fe(III) from Cl- medium by Cyanex 272 and HA [17] had been reported. There is no report on the equilibrium of the extraction of V(IV) from
medium by Cyanex 272 [18,19] and by Cyanex 302 [20] and of V(IV) and V(V) in presence of Mo(VI), W(VI), U(VI), Ti(IV), Al(III), Cr(III), Fe(III) from Cl- medium by Cyanex 272 and HA [17] had been reported. There is no report on the equilibrium of the extraction of V(IV) from 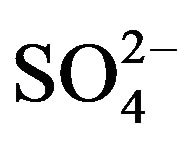 medium by HA. The present paper reports the extraction characteristics of V(IV) from
medium by HA. The present paper reports the extraction characteristics of V(IV) from 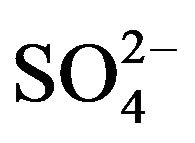 medium by HA dissolved in kerosene in order to determine the effects of various parameters on extraction ratio and the value of Kex; and also to propose mechanism. The loading of HA by V(IV) and the stripping ability of various mineral acids are also investigated. Finally, the possibilities of separation of V(IV) from some 3d-block cations in binary mixtures have been examined.
medium by HA dissolved in kerosene in order to determine the effects of various parameters on extraction ratio and the value of Kex; and also to propose mechanism. The loading of HA by V(IV) and the stripping ability of various mineral acids are also investigated. Finally, the possibilities of separation of V(IV) from some 3d-block cations in binary mixtures have been examined.
2. Materials and Methods
2.1. Materials
Cyanex 301 is a product of Cytec Canada Inc. and received as a gift. It contains 75% - 83% R2PS2H, 5% - 8% R3PS, 3% - 6% R2PSOH and ~2% unknown compound [21] and used without further purification since R3PS and R2PSOH have the extracting powers too. Kerosene purchased from the local market is distilled (200˚C - 260˚C) to collect the colorless aliphatic fraction. As-received NH4VO3 (99%, Riedel-deHaen) and VOSO4∙xH2O (99.9%, Alfa Aesar-Johnson-Mathey), H2O2 (30% (w/v), MerckGermany) are used. Analytically pure diluents (besides kerosene) are the products of Riedel-deHaen and E. Merck-India.
2.2. Analytical
The aqueous [V(IV)] has been measured by the HNO3 oxidative-H2O2 method [22] at 450 nm using a UV-visible Spectrophotometer (UV-1650 PC, Shimadzu, Japan). For standard and test solution preparations, NH4VO3 and VOSO4∙xH2O, respectively, are used. A Mettler Toledo pH meter (model MP 220) is used for pH measurement.
2.3. Procedure for Extraction
The procedure for extraction is given elsewhere [20,23]. Two phases are agitated at O/A = 1 (O = 20 mL) and 303 K (otherwise stated) for a predetermined time (15 min). The phase separation is quick; and the aqueous phase after disengagement is analyzed for its pH(eq) and V(IV)- content. Then “D” is calculated as usual [20,23].
2.4. Procedure for Loading
The loading procedure is given elsewhere [24]. An aliquot of 100 mL 0.20 mol/L HA-kerosene solution has been used for V(IV)-loading from an aqueous solution containing 1.0 g/L V(IV) and 0.10 mol/L 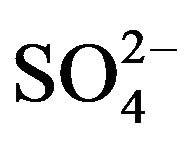 at pH(ini) = 2.60. After each stage of contact, cumulative [V(IV)](o,eq) is calculated to monitor the progress of stripping.
at pH(ini) = 2.60. After each stage of contact, cumulative [V(IV)](o,eq) is calculated to monitor the progress of stripping.
2.5. Procedure for Stripping
The stripping procedure is similar to the extraction procedure. The fully loaded organic solution obtained in the loading study is diluted to contain 200 mg/L V(IV) and 0.10 mol/L HA in kerosene. Vanadium(IV) in this solution has been stripped by (0.1, 0.3 or 1.0) mol/L (H2SO4, HCl or HNO3) solution. A shaking time of 1 h is allowed arbitrarily. After equilibration and phase separation, the aqueous phase is analyzed for its [V(IV)] in order to calculate % V(IV) stripped by:
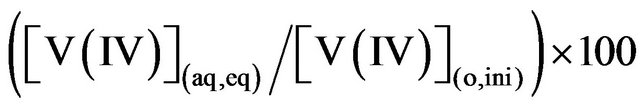
2.6. Treatment of Extraction Equilibrium Data
R2PS2H is the principal constituent of Cyanex 301. This species is monomeric in non-polar diluents (owing to low electronegativity of sulphur) [21,25]. Consequently, Cyanex 301 has been abbreviated as HA. In aqueous solution, VO2+ can form complexes with co-existing OH- and  or
or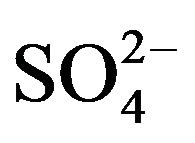 . On considering the existence of [VO(OH)jLk] (L is
. On considering the existence of [VO(OH)jLk] (L is  or
or 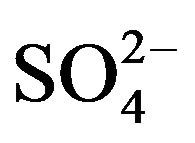 and the charge of the complex is neglected for simplicity) in the aqueous phase and the monomeric charge-less extracted complex does not contain any OH- and
and the charge of the complex is neglected for simplicity) in the aqueous phase and the monomeric charge-less extracted complex does not contain any OH- and  or
or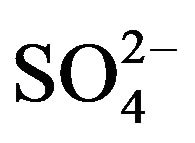 , the equilibrium for its extraction by HA can be represented as (“x”, “2 − j” and “k” are experimental extractant, pH and co-existing ligand dependences, respectively):
, the equilibrium for its extraction by HA can be represented as (“x”, “2 − j” and “k” are experimental extractant, pH and co-existing ligand dependences, respectively):
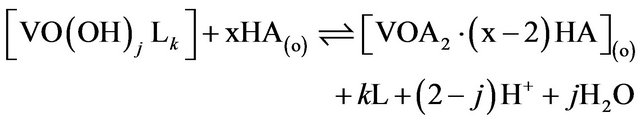 (1)
(1)
On defining “D” as
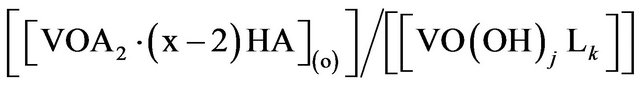
Kex of Equation (1) can be expressed as:
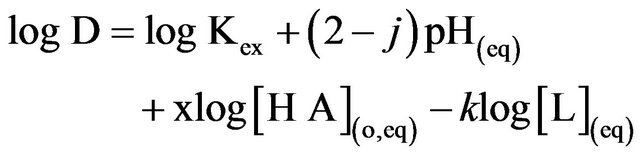 (2)
(2)
The Equation (2) represents the basic equation for solvated chelate formation by reaction of a metal ion with an acidic extractant. All concentration terms and pH in Equation (2) refer to the equilibrium values. Although it is difficult to collect D-values experimentally at a constant set of pH(eq), [HA](o,eq) and [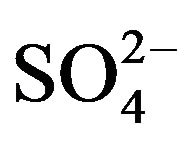 ](eq), it is possible to modify the experimental D-values to CD values at a chosen set of constant pH(eq), [HA](o,eq) and [
](eq), it is possible to modify the experimental D-values to CD values at a chosen set of constant pH(eq), [HA](o,eq) and [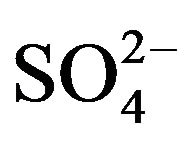 ](eq). Since in almost all experiments, 0.10 mol/L
](eq). Since in almost all experiments, 0.10 mol/L 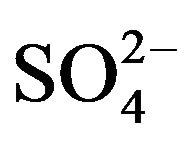 (~25 times greater than [V(IV)]) has been used; it can be assumed that [
(~25 times greater than [V(IV)]) has been used; it can be assumed that [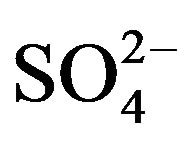 ](eq) will not be significantly changed from [
](eq) will not be significantly changed from [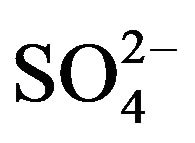 ](ini). Consequently, after determining the approximate pH and extractant dependences and rounding up these values, log CD can be calculated by:
](ini). Consequently, after determining the approximate pH and extractant dependences and rounding up these values, log CD can be calculated by:
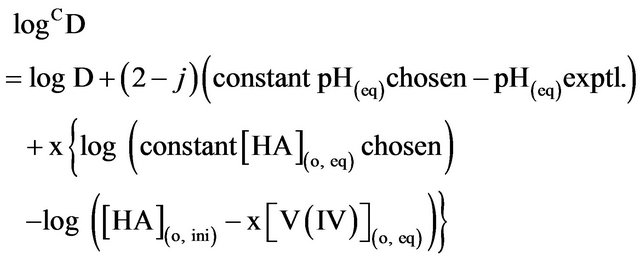 (3)
(3)
Moreover, [H2A2](o,eq) is equal to
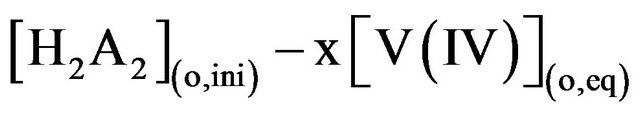
Consequently, according to Equation (2), log CD should be independent of [V(IV)] if the solutions behave ideally; while it should depend on pH(eq), [HA](eq) or [L] at a constant set of other parameters. Moreover, as Kex is related to temperature by Van’t Hoff equation, log CD will also depend on temperature.
3. Results and Discussion
3.1. Extraction Equilibrium
Some preliminary experiments indicate that V(IV) is extractable by HA at pH ~ 0.50. When 0.20 g/L V(IV) containing 0.10 mol/L 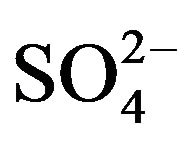 at pH(ini) of 2.30 is extracted by 0.10 mol/L HA in kerosene at 303 K and O/A = 1, then it is found that [V(IV)](o) is increased up to phase contact of 10 min. It is therefore concluded that the equilibration time is 10 min (15 min is allowed in subsequent experiments).
at pH(ini) of 2.30 is extracted by 0.10 mol/L HA in kerosene at 303 K and O/A = 1, then it is found that [V(IV)](o) is increased up to phase contact of 10 min. It is therefore concluded that the equilibration time is 10 min (15 min is allowed in subsequent experiments).
Variation of “D” with [V(IV)](ini) is found out at four different set of experimental parameters. It is found in all cases that [V(IV)](o) is increased, but ‘D’ is decreased continuously with increasing [V(IV)](ini) in the aqueous phase. This is contrary to Equation (2) which is valid at constant [HA](o,eq) and pH(eq). The observed decreasing behavior might be due to the non-constancy of [HA](o,eq) and pH(eq) for various extents of V(IV) extraction. On calculating log CD (by Equation (3) on considering “2 − j” = 1.50 at constant pH(eq) of 1.40 or 1.67 at constant pH(eq) of 1.80 and x = 2.00 for all systems), log CD vs. log ([V(IV)](ini), mol/L) plots are drawn in Figure 1. Plots indicate the independency of CD on [V(IV)] at least up to 300 mg/L. Systems with higher [V(IV] behave nonideally.
At a constant [HA](o, eq), the plot of log D vs pH(eq) should be a straight line with slope equaling to “2 − j” (cf. Equation (2)). For low [ ] of 0.10 mol/L, Figure 2 represents log CD vs pH(eq) plots at constant [HA](o,eq) of 0.20 and 0.30 mol/L. In both cases, straight lines are not obtained. Three or four points at lpHr produce straight lines of slope 2. At hpHr, the slope is decreased gradually and at pH 2.15, it becomes 1.6. It is concluded that single type of extractable species is formed from two different types of aqueous V(IV) species, viz. VO2+ and VO(OH)+. In the first case, two H+ are liberated; whereas, in the second case, one H+ is liberated, per V(IV) being extracted into the organic phase. As an evidence to this statement, the formation of same extractable species from VO2+, VO(OH)+ and VO(OH)2 by D2EHPA may be cited [5]. The effect of pH on extraction at hcr of [
] of 0.10 mol/L, Figure 2 represents log CD vs pH(eq) plots at constant [HA](o,eq) of 0.20 and 0.30 mol/L. In both cases, straight lines are not obtained. Three or four points at lpHr produce straight lines of slope 2. At hpHr, the slope is decreased gradually and at pH 2.15, it becomes 1.6. It is concluded that single type of extractable species is formed from two different types of aqueous V(IV) species, viz. VO2+ and VO(OH)+. In the first case, two H+ are liberated; whereas, in the second case, one H+ is liberated, per V(IV) being extracted into the organic phase. As an evidence to this statement, the formation of same extractable species from VO2+, VO(OH)+ and VO(OH)2 by D2EHPA may be cited [5]. The effect of pH on extraction at hcr of [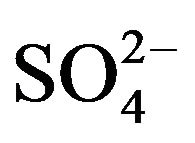 ] (1.50 mol/L) has also been investigated (cf. Figure 2). A
] (1.50 mol/L) has also been investigated (cf. Figure 2). A
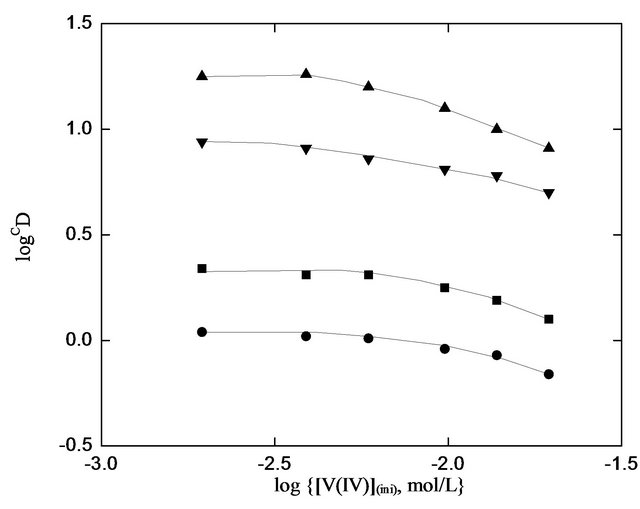
Figure 1. Effect of [V(IV)] on its extraction. Temp. = 303 K, [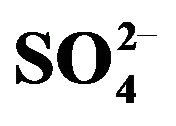 ] = 0.10 mol/L, Equilibration time = 15 min, O/A = 1 (O = 20 mL). (¢), pH(ini) = 1.52, constant pH(eq) chosen = 1.4, [HA](o,ini) = 0.30 mol/L = [HA](o,eq); (), pH(ini) = 1.52, , constant pH(eq) chosen = 1.42, [HA](o,ini) = 0.20 mol/L = [HA](o,eq); (p), pH(ini) = 1.90, constant pH(eq) chosen = 1.78, [HA](o,ini) = 0.30 mol/L = [HA](o,eq); and (q), pH(ini) = 1.90, constant pH(eq) chosen = 1.78 [HA](o,ini) = 0.20 mol/L = [HA](o,eq).
] = 0.10 mol/L, Equilibration time = 15 min, O/A = 1 (O = 20 mL). (¢), pH(ini) = 1.52, constant pH(eq) chosen = 1.4, [HA](o,ini) = 0.30 mol/L = [HA](o,eq); (), pH(ini) = 1.52, , constant pH(eq) chosen = 1.42, [HA](o,ini) = 0.20 mol/L = [HA](o,eq); (p), pH(ini) = 1.90, constant pH(eq) chosen = 1.78, [HA](o,ini) = 0.30 mol/L = [HA](o,eq); and (q), pH(ini) = 1.90, constant pH(eq) chosen = 1.78 [HA](o,ini) = 0.20 mol/L = [HA](o,eq).
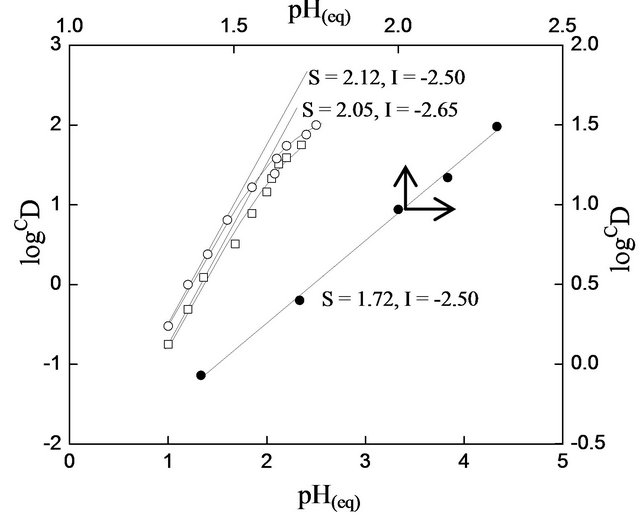
Figure 2. Effect of pH(eq) on extraction. [V(IV)](ini) = 200 mg/L. (), [HA](o,ini) = 0.30 mol/L = [HA](o,eq); (£), [HA](o,ini) = 0.20 mol/L = [HA](o,eq); (), [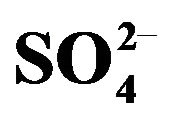 ] = 1.50 mol/L, [HA](o,ini) = 0.15 mol/L= [HA](o,eq). Other parameters are as in Figure 1.
] = 1.50 mol/L, [HA](o,ini) = 0.15 mol/L= [HA](o,eq). Other parameters are as in Figure 1.
straight line having slope of 1.72 is obtained. It is therefore concluded that the pH dependency in the present system depends on [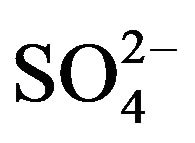 ]. As [
]. As [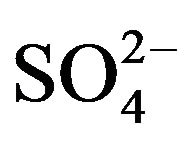 ] is increased, pH dependency is decreased.
] is increased, pH dependency is decreased.
According to Equation (2), the plot of log CD vs. log [HA](o, eq) at a constant pH(eq) should be a straight line with slope giving moles of HA(x) associated with 1 mol of V(IV) in extracted species. The log D vs log [HA](o, ini) plots (as there will be a very little variation between pH(ini) and pH(eq) and between [HA](o, ini) and [HA](o, eq)) are shown in Figure 3. Straight lines having slope of 2 are indeed obtained at both lcr and hcr of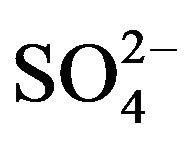 . It is
. It is
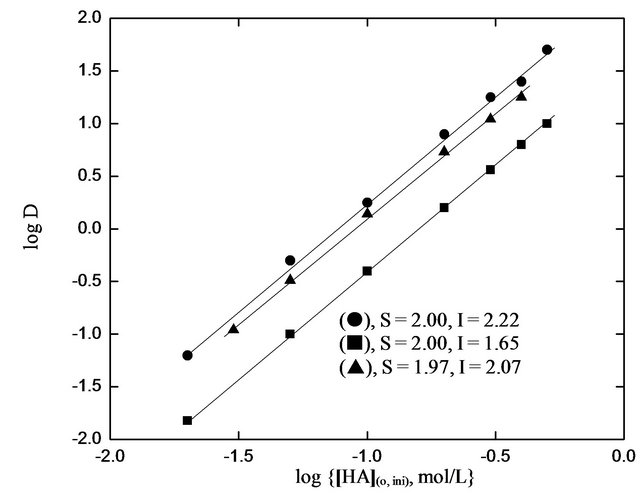
Figure 3. Effect of [HA](o,eq) on extraction. (), pH(ini) = 2.00; (¢), pH(ini) = 1.50; (p), pH(ini) = 1.40, [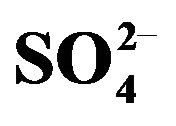 ] = 1.50 mol/L. Other parameters are as in Figure 1.
] = 1.50 mol/L. Other parameters are as in Figure 1.
therefore concluded that the HA-dependency is always 2 irrespective of [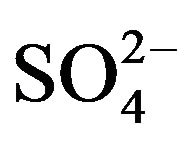 ], though the pH-dependency is dependent of [
], though the pH-dependency is dependent of [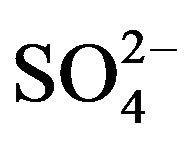 ].
].
The log CD vs log ([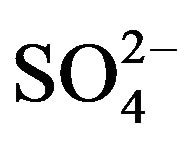 ], mol/L) plot is displayed in Figure 4" target="_self"> Figure 4. Experimental points fall on a curve. In lcr of
], mol/L) plot is displayed in Figure 4" target="_self"> Figure 4. Experimental points fall on a curve. In lcr of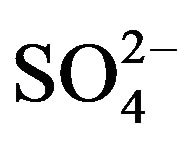 , CD is seldom changed; whilst in the hcr, it is considerably increased with increasing [
, CD is seldom changed; whilst in the hcr, it is considerably increased with increasing [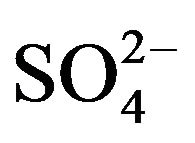 ]. The curve in the figure is theoretical and represented by:
]. The curve in the figure is theoretical and represented by:
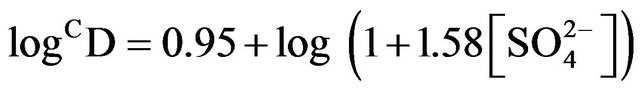 (4)
(4)
which is obtained to fit experimental points as described in the caption of Figure 4. It is seen that [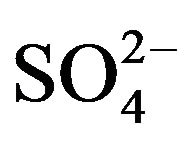 ] has little effect on extraction when its concentration is kept ~0.10 mol/L; but at hcr of [
] has little effect on extraction when its concentration is kept ~0.10 mol/L; but at hcr of [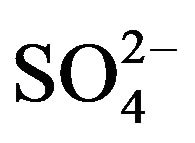 ], log CD is almost directly proportional to log [
], log CD is almost directly proportional to log [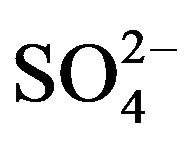 ].
].
The Van’t Hoff plots are shown in Figure 5. It is found that CD is increased with increasing temperature and the straight line relationship holds. Slopes of the lines are −870 (DH = 16.70 kJ/mol) and −830 (DH = 15.95 kJ/mol) for pH = 1.35 and 1.25 systems, respecttively. Therefore, the process is endothermic with low DH value of ~16 kJ/mol.
It is evident from these studies that the value of “x” is 2 irrespective of the experimental parameter but the value of “k” is 0 at low [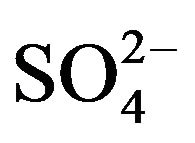 ] and −1 at high [
] and −1 at high [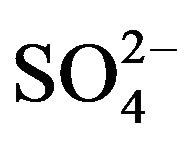 ]. The value of “2 − j” is 2 in low pH(eq) and <2 in high pH(eq). At lcr of
]. The value of “2 − j” is 2 in low pH(eq) and <2 in high pH(eq). At lcr of 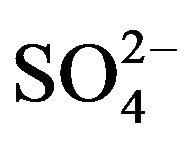 and at lpH(eq)r, “2 − j” = 2 implies that “j” = 0; but at hpH(eq)r, “2 − j” < 2 (but >1) implies that 1 < j < 2. On the other hand, at hcr of SO42- and at both lpH(eq)r and hpH(eq)r, “2 − j” < 2 implies j > 0.
and at lpH(eq)r, “2 − j” = 2 implies that “j” = 0; but at hpH(eq)r, “2 − j” < 2 (but >1) implies that 1 < j < 2. On the other hand, at hcr of SO42- and at both lpH(eq)r and hpH(eq)r, “2 − j” < 2 implies j > 0.
3.2. Evaluation of Extraction Equilibrium Constant
The foregoing results give the equation for logCD at 303
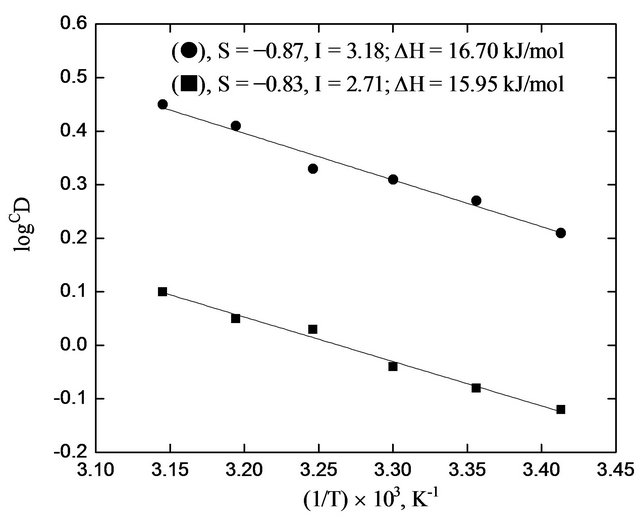
Figure 5. Effect of temperature on extraction. (), pH(ini) = 1.35, [HA](o,ini) = 0.30 mol/L = [HA](o,ini); (¢), pH(ini) = 1.25, [HA](ini) = 0.20 mol/L = [HA](o,ini). Other parameters are as in Figure 1.
K in lcr of sulphate as:
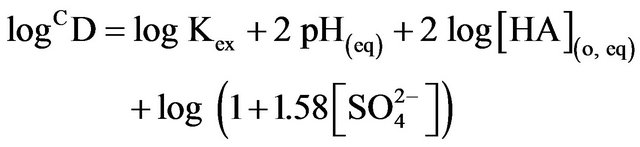 (5)
(5)
Based on Equation (5), value of log Kex has been evaluated from intercepts of the straight lines or asymptotic lines in Figures (2)-(4) and tabulated (Table 1). The average log Kex is −1.419 at lcr of 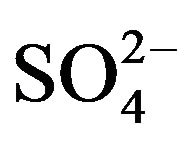 with stand. dev. of 0.105. Besides, log CD at 303 K in hcr of
with stand. dev. of 0.105. Besides, log CD at 303 K in hcr of 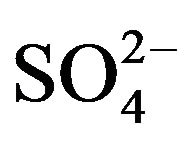 can be put as:
can be put as:
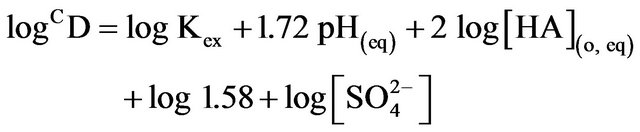 (6)
(6)
On using Equation (6), log Kex is evaluated as −0.94 with stand. dev. of 0.026.
3.3. Extraction Mechanism
The empirical equation for Kex at 303 K is:
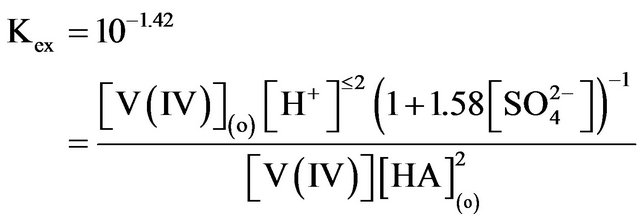 (7)
(7)
The 1st and 2nd ionization constants of H2SO4 are 103 [26] and 10−2 [27], respectively. These values suggest that 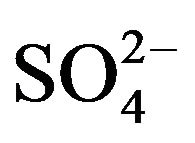 will be more available than
will be more available than  in the working pH region. So, L in Equation (1) represents
in the working pH region. So, L in Equation (1) represents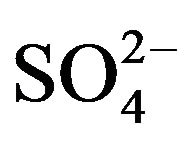 . As the values of “x”, “k”, “l” and “(2 − j)” are known at different experimental conditions, Equation (1) will provide extraction mechanisms. Although in Equation (1), “L” is presented as a product (liberated during complex formation); experimental results indicate that it is associated with V(IV) during complex formation. As “x” is always 2, non-solvated chelate (VOA2) is formed at lcr of
. As the values of “x”, “k”, “l” and “(2 − j)” are known at different experimental conditions, Equation (1) will provide extraction mechanisms. Although in Equation (1), “L” is presented as a product (liberated during complex formation); experimental results indicate that it is associated with V(IV) during complex formation. As “x” is always 2, non-solvated chelate (VOA2) is formed at lcr of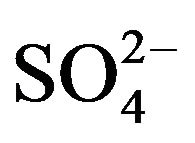 , whereas, solvated complex (VOSO4·2HA) is formed at hcr of
, whereas, solvated complex (VOSO4·2HA) is formed at hcr of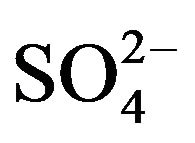 . Typical equilibria are suggested as:
. Typical equilibria are suggested as:
1) in lcr of 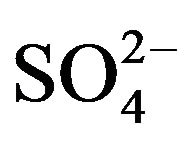 and lpHr:
and lpHr:
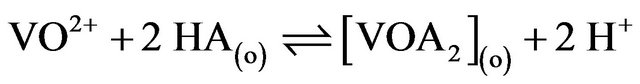 (8)
(8)
2) in lcr of 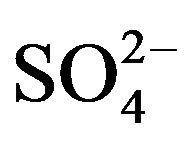 and hpHr (limiting):
and hpHr (limiting):
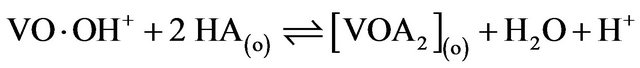 (9)
(9)
3) in hcr of 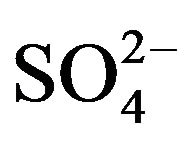 (limiting):
(limiting):
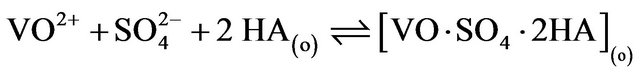 (10)
(10)
An alternative option of the formation of [VO∙SO4∙2HA](o) may be the formation of [VO(HSO4)(A)∙HA](o) with the simultaneous liberation of a proton. These are only presumptions from the experimental results and not proven by other means.
3.4. Effect of Diluent
In order to determine the effect of diluent on V(IV)-distribution, D-values have been measured when the same aqueous phase has been extracted separately by 0.10 mol/L HA in different diluents keeping all other parametric conditions ([V(IV)] = 200 mg/L, pH(ini) = 2.00 and [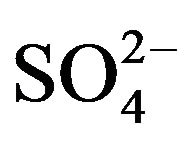 ] = 0.01 mol/L) identical. It is observed that the extraction ratio increases in the following order with the variation of diluent: CHCl3 (e = 4.807; D = 0.42) < 1,2-C2H4Cl2 (e = 10.42; D = 0.54) < C6H4-(CH3)2 (e = 2.26; D = 0.68) < cyclo-C6H12 (e = 2.02; D = 1.31) = C6H5Cl (e = 5.69; D = 1.31) < C6H5-CH3 (e = 2.385; D = 1.64) < n-C7H16 (e = 1.921; D = 2.08) = C6H6 (e = 2.274; D = 2.08) < CCl4 (e = 2.228; D = 2.34) < petroleum benzin (D = 3.62) < kerosene (e = 2.00; D = 3.93). The study helps draw the conclusion that kerosene is a very good diluent followed by petroleum benzin and CCl4 for the extraction of V(IV) by Cyanex 301. CHCl3, 1,2-C2H4Cl2 and C6H4 (CH3)2 are not recommended. 79.72% V(IV) extraction in kerosene phase is decreased to only 29.70% V(IV) extraction in CHCl3 phase.
] = 0.01 mol/L) identical. It is observed that the extraction ratio increases in the following order with the variation of diluent: CHCl3 (e = 4.807; D = 0.42) < 1,2-C2H4Cl2 (e = 10.42; D = 0.54) < C6H4-(CH3)2 (e = 2.26; D = 0.68) < cyclo-C6H12 (e = 2.02; D = 1.31) = C6H5Cl (e = 5.69; D = 1.31) < C6H5-CH3 (e = 2.385; D = 1.64) < n-C7H16 (e = 1.921; D = 2.08) = C6H6 (e = 2.274; D = 2.08) < CCl4 (e = 2.228; D = 2.34) < petroleum benzin (D = 3.62) < kerosene (e = 2.00; D = 3.93). The study helps draw the conclusion that kerosene is a very good diluent followed by petroleum benzin and CCl4 for the extraction of V(IV) by Cyanex 301. CHCl3, 1,2-C2H4Cl2 and C6H4 (CH3)2 are not recommended. 79.72% V(IV) extraction in kerosene phase is decreased to only 29.70% V(IV) extraction in CHCl3 phase.
3.5. Loading of Cyanex 301 with V(IV)
The cumulative [V(IV)](o) (g/L) has been plotted against the number of phase contact in Figure 6. It is observed that the loading of V(IV) in the organic phase is ended up at the 13th contact. An aliquot of 1 L 0.20 mol/L HA is saturated with 5.07 g V(IV) and so the loading capacity is calculated as 7.87 g V(IV) per 100 g HA. The loading capacity is considerably high, and so it can be recommended for a large scale separation of V(IV) from an aqueous solution. The extraction of 5.07 g V(IV)/L by 1 L 0.20 molar HA at saturated loading implies the HA/V(IV) mole ratio of 2.01 which is identical to that obtained from the extractant dependence study. The loading results indicate that the mechanism of extraction at high loading is not changed from that suggested at low loading.
3.6. Stripping of V(IV)-Loaded Organic Phase by Mineral Acids
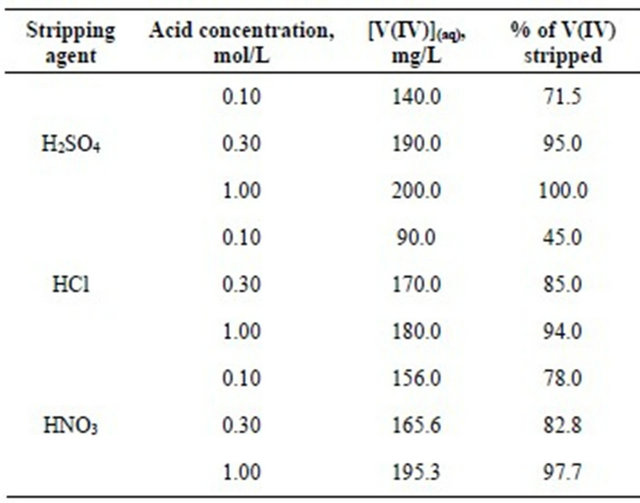
The maximum V(IV) loaded organic phase containing 5.07 g/L V(IV) with theoretically no free extractant, after proper dilution and adjustment of free [HA], has been subjected for stripping by 0.1, 0.3 and 1.0 mol/L H2SO4, HNO3 and HCl solutions 303 K and O/A = 1. The stripping results are given in Table 2. It is found that stripping percentage is more or less acceptable in all three acids used alone. In all cases, stripping percentage is increased with increasing concentration of acid. It is seen that 71.50% stripping by 0.10 mol/L H2SO4 is increased to 100% stripping by 1 mol/L H2SO4. Similarly, 45% stripping by 0.10 mol/L HCl is increased to 94% stripping with 1 mol/L HCl; whereas, 78% stripping by 0.10 mol/L HNO3 is increased to ~98% stripping by 1 mol/L HNO3. Sulphuric acid (1 mol/L) is sufficient to strip off V(IV) quantitatively. Nitric acid and hydrochloric acid
Table 1. Evaluation of the values of Kex at 303 K.
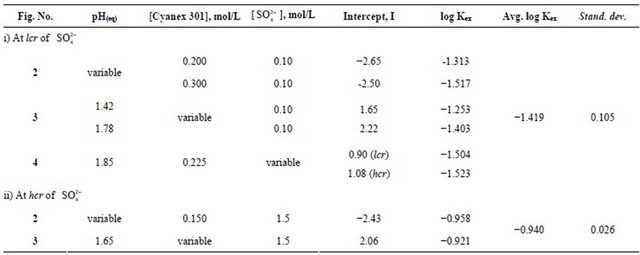
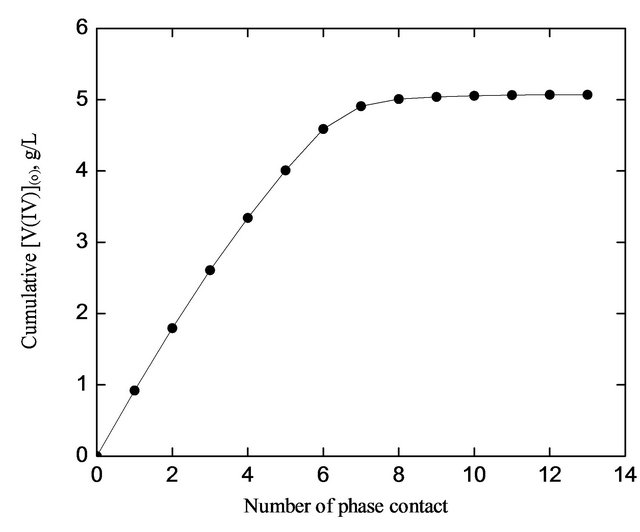
Figure 6. Loading of HA with V(IV). [V(IV)](ini) = 1000 mg/L, [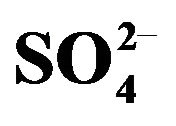 ] = 0.10 mol/L, pH(ini) = 2.60, [HA] = 0.20 mol/L, Stage equilibration time = 15 min, Temp. = 303 K, O/A = 1 (O = 100 mL).
] = 0.10 mol/L, pH(ini) = 2.60, [HA] = 0.20 mol/L, Stage equilibration time = 15 min, Temp. = 303 K, O/A = 1 (O = 100 mL).
can also be used in stripping if two stage stripping is practiced.
It is reported that Cyanex 302 and Cyanex 301 undergo oxidation in oxidizing environment [28-31] (oxidation products being R2(P=S)-S-S-(S=P)R2, R2(P=S)OH and R2(P=O)OH). It can be demonstrated, however, that when V(IV) is extracted repeatedly from fresh aqueous solutions ([V(IV)] = 0.20 g/L, [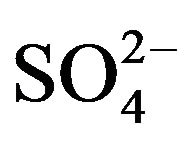 ] = 0.10 mol/L, pH(ini) = 1.50) by 0.30 mol/L HA in kerosene (fresh in the first step and regenerated afterwards) and stripped subsequently with 1 mol/L H2SO4, then (79 ± 2)% extraction and 100% stripping are observed from the 1st - 25th extraction-stripping steps. It is therefore concluded that HA does not undergo any sort of oxidation as also reported by Sole et al. [32].
] = 0.10 mol/L, pH(ini) = 1.50) by 0.30 mol/L HA in kerosene (fresh in the first step and regenerated afterwards) and stripped subsequently with 1 mol/L H2SO4, then (79 ± 2)% extraction and 100% stripping are observed from the 1st - 25th extraction-stripping steps. It is therefore concluded that HA does not undergo any sort of oxidation as also reported by Sole et al. [32].
Table 2. Stripping of V(IV) loaded organic phase using different acid solutions. [V(IV)](o) = 200 mg/L, [Cyanex 301] = 0.10 mol/L, Equilibration time = 1 h, Temp. = (303 ± 0.5) K, O/A = 1 (O = 20 mL).
3.7. Separation Ability of V(IV) from Some Other Metal Ions
In order to examine the effectiveness of HA towards the mutual separations of some 3d-block metal ions viz. Ti(IV), V(IV), Fe(III), Co(II), Ni(II), Cu(II) and Zn(II), the extraction percentages of these metal ions have been estimated. For this purpose, 0.20 g/L metal ion is extracted from 0.10 mol/L 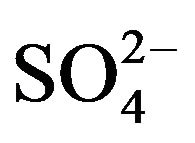 (or, [
(or, [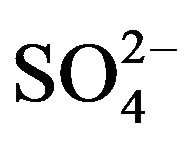 ] = H2SO4 when [H2SO4] > 0.10 mol/L) medium at different pH(eq) values by 0.10 mol/L HA in kerosene at 303 K and O/A = 1 (O = 20 mL) on equilibration for 1 h. The extraction results given in Table 3 predict the following:
] = H2SO4 when [H2SO4] > 0.10 mol/L) medium at different pH(eq) values by 0.10 mol/L HA in kerosene at 303 K and O/A = 1 (O = 20 mL) on equilibration for 1 h. The extraction results given in Table 3 predict the following:
1) V(IV) can be separated from Cu(II) at pH 0 in single step (0% V(IV) extraction and 100% Cu(II) extraction).
2) On using counter-current extraction stages, V(IV)
Table 3. Solvent extraction data of some 3d-block elements by Cyanex 301 dissolved in kerosene. [Cyanex 301] = 0.10 mol/L (in kerosene); [Metal ion] = 0.20 g/L; [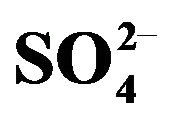 ] = [H2SO4] or 0.10 mol/L, Temp = 303 K, O/A = 1 (O = 20 mL), Equilibration time = 1 h.
] = [H2SO4] or 0.10 mol/L, Temp = 303 K, O/A = 1 (O = 20 mL), Equilibration time = 1 h.
NE: non-extractable, CE: complete extraction; *Aqueous solution becomes cloudy before extraction but becomes clear after extraction.
can be separated from:
• Zn(II) at pH ~ 0.5 (1% V(IV) extraction and 98% Zn(II) extraction)• Fe(III) at pH 1.0 (4.3% V(IV)-extraction and 99% Fe(III)-extraction)• Co(II) at pH 1.5 (38.7% V(IV)-extraction and 91% Co(II) extraction), and
• Ni(II) at pH 1.5 (38.7% V(IV)-extraction and 95% Ni(II) extraction).
3) Separation of V(IV) from Ti(IV) is difficult but not impossible. Separation can be achieved at pH 2.0 on using counter-current multistage extraction.
4. Conclusions
The following conclusions are drawn:
1) Vanadium(IV) can be extracted by HA at pH above 1. The equilibration time 10 min. Up to at least 0.30 g/L V(IV), the extraction ratio (D) is independent of V(IV) concentration in the aqueous phase.
2) The corrected extraction ratio (CD) is proportional to [H+]−2 at its lcr, [HA]2 and the factor (1 + 1.58 [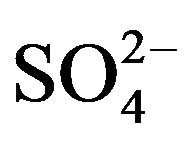 ]). The Kex values at 303 K are 10-1.419 and 10−0.94 in 0.10 and 1.50 mol/L
]). The Kex values at 303 K are 10-1.419 and 10−0.94 in 0.10 and 1.50 mol/L 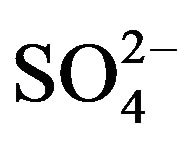 medium, respectively.
medium, respectively.
3) The extraction process is endothermic with a DH value of 16 kJ/mol.
4) The extracted species are VOA2 at lcr and VO·HSO4·A·HA or VOSO4 at hcr of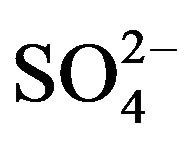 .
.
5) 100 g HA can extract as much as 7.87 g V(IV). This gives HA/V(IV) mole ratio of 2.01 indicating that the mechanism of extraction is not changed with loading.
6) Among the diluents used, kerosene is the best. The least effective diluent is CHCl3 followed by 1,2-C2H4Cl2 and xylene.
7) 1 mol/L H2SO4, HCl and HNO3 can strip off 100%, 94% and 97.7% V(IV), respectively, in single step.
8) Using HA as extractant, V(IV) can be separated from Cu(II). It can be separated from Zn(II) at pH 0.5 but for clear-cut separation counter-current 2-3 stage extractions may be required. In a similar way, V(IV) can be separated from Fe(III) at pH 1.
Separations V(IV) from Ti(IV), Co(II) and Ni(II) by HA appear to be difficult.
Notations and Abbreviations

NOTES