Influence of Traditional Inoculum and Fermentation Time on the Organoleptic Quality of "Attiéké” ()
1. Introduction
Cassava is an important staple food in many tropical countries [1] with an annual World production estimated to 200 million tons in 2002 [2,3]. The majority of production is located in Africa where 99.1 million tons were grown; 51.5 million tones were grown in Asia and 33.2 million tones in Latin America and the Caribbean. Over 90% of the cassava in Africa as some fermented products for human consumption [4]. In the humid and sub-humid areas of tropical Africa, cassava is either a primary staple food or a secondary co-staple. Cooked in various ways, cassava is used in a great variety of dishes. The root has a delicate and distinctive flavour once boiled and can replace boiled potatoes in many uses as an accompaniment for meat or fish dishes. Many diversity of food products are resulting from the starchy cassava root flour which include “attoukpou”, “gari” and “attiéké” [5].
“Attiéké” is an essentially flavour starchy ingredient, produced from fermented cassava root. The product is widely consumed in Côte-d’Ivoire and in neighbouring countries [6]. To produce that staple food, cassava roots are peeled, cut into pieces, washed and milled. During grating, the cassava paste is mixed with a traditional prepared inoculum or starter culture. The inoculum is prepared by storing boiled cassava roots for three days in an unwashed jute bag previously used for inoculum preparation. According to [7], the use of an inoculum results in a decrease in fermentation time of “attiéké” compared with other fermented cassava products, such as “gari” or “chickwangue”, prepared without inocula [8,9]. The inoculated paste is fermented overnight in covered bins. After fermentation, the pulp obtained is filled into bags and pressed for several hours to remove water. The pressed cake is taken from the bags and squeezed through a sieve to obtain granules or semolina that are partially sun-dried and then, cleaned to remove fibers and waste. The dried granules are steamed to produce “Attiéké” or semolina, which is sold in small plastic bags as a readyto-eat food [10].
Fermentation applied to cassava processing is very important because it helps to eliminate cyanogenic compounds, to preserve, to soften and to produce important organic acids for the organoleptic properties of attiéké [11]. Several researchers have studied the “Attiéké” microflora [12,13] but the variability of inoculum amount and fermentation time still remains unknown. Therefore, the aim of this study is to determine parameters affecting the microflora and sensory qualities of “Attiéké”.
2. Materials and Methods
2.1. Materials
Local cassava variety IAC (Improved Africa Cassava) was used for this study. Mature plants (aged between 12 and 18 months) were harvested from the experimental station of University of Abobo-Adjame (Abidjan) in Cote d’Ivoire.
2.2. Preparation of “Attiéké”
The processes described by Aboua [12] were followed with some modifications. The cassava roots were peeled, cut into small pieces and washed with cold water. The pieces were inoculated by 6%, 8%, 10% and 12% of traditional inoculums, respectively, palm oil (4 ml) and grated to a pulp. The pulp was fermented in bowls for 6, 12 and 18 hours. 2 Kg of paste was then filled into jute sacks and pressed until attiéké production. Some samples (10 g) were taken each step for microbial analysis and also “Attiéké” step. After that, the sensory evaluation was lead to compare the different types of “Attiéké”.
2.3. Determination of pH of Microbial Suspension
The pH of microbial suspension was determined with a pH Meter (pH-meter P 107, CONSORT, Bioblock, France). It was determined from the suspension obtained from 10 g of starter in 90 ml of distilled water.
2.4. Enumeration of Groups of Micro-Organisms
Twenty one (21) samples of the fermented cassava were aseptically used and processed as follows: 10 g of sample were diluted with 90 ml of peptone water (0.1% casein peptone, 0.85% NaCl) before homogenization in a stomacher (Colworth 400, Müller + Krempel AG, London) for 3 min.
The samples were plated in duplicate with the convenable dilution of the microbial suspension. Total counts of different groups of organisms were made for each of the 21 samples. Plate Count Agar (PCA, NF T90-401, NF T90-402 and ISO 4833), set at 30˚C for 24 - 72 h, was used to enumerate the mesophilic aerobic microorganisms. Colonies were counted in all Petri dishes which contained 30 - 300 colonies. Enterococci were counted on Bile Esculin Azide agar (BEA, ISO 7899/1) at 37˚C for 24 - 48 h. Enumeration of Lactobacillus were done on Man Rogosa Sharpe agar (MRS, NF V 08-051) at 30˚C over 24 - 72 h. Yeasts and moulds were counted on SABOURAUD + Chloramphénicol agar as used according to standard XP V 08-059 at 25˚C for 3 - 5 days.
2.5. Sensory Analysis [14]
A panel of 15 assessors were used to taste the products (Attiéké) with the descriptive test. The responses made by them were recorded using the procedure outlined by Touraille (1990).
2.6. Statistical Analysis
Software Stat Soft with Duncan test (Statistica 99ème edition, France) was used for the statistic analysis of the sensory data. Significance was at p < 0.05. The threshold of significance was used in order to test whether there were significance differences between “Attiéké” data of different time of fermentation quantity of inoculums.
3. Results
3.1. Evolution of pH of Microbial Suspension
The starter rate and the pH were inversely proportional (Figure 1(a)). The time of fermentation and the pH were also inversely proportional. The pH remained slightly stable between 6 and 12 h then dropped considerably to 18 h to reach 3.79 (Figure 1(b)).
3.2. Isolation and Enumeration of Micro-Organisms
The highest number of Mesophilic aerobic micro-organisms was observed with a quantity of 6% of the traditional starter. But generally, Lactobacillus constitutes the most important microflora followed by enterococci, yeasts and moulds. The peak of Lactobacillus (9.14 Log cfu/g), enterococci (7.64 Log cfu/g), yeasts and moulds (7.30 Log cfu/g) was observed with 10% of the traditional starter (Figure 2). The number of Lactobacillus grown gradually between 6 and 12 h then got down at 18 h respectively 7.74, 9.14 and 8.30 Log cfu/g. Then followed enterococcus sp. with 6.17 cfu/g proportions at 6 h but reached its maximum (8.66 Log cfu/g) at 18 h. Finally, yeasts and moulds remained minority at 6 h and decreased the following times (12 to 18 h) between 7.30 and 7.04 Log cfu/g (Figure 3).
3.3. Sensory Analysis
Generally, fermentation time affected the organoleptic quality of “Attiéké”. Statistical analysis on the data showed significant differences (p < 0.05) on the color, texture, aroma and granulometry. It was also noted that “Attiéké” produced after 12 hours of fermentation had a good general acceptability score (Figure 4). The inoculums quality caused differences on all the attributes and the most perceived were at the level of the taste, texture, the aroma and granulometry. However it was noted that
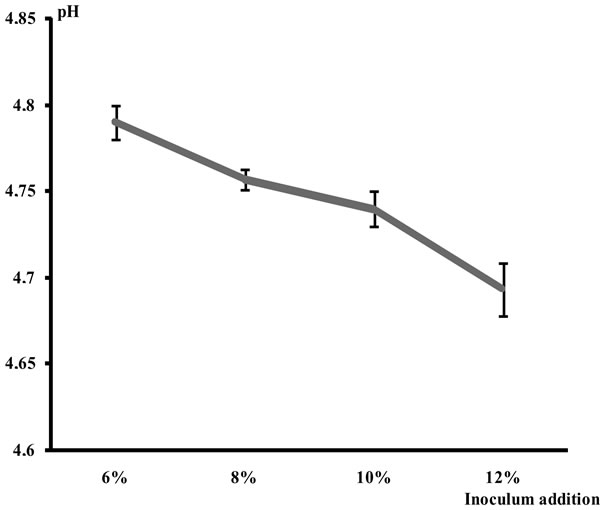 (a)
(a) (b)
(b)
Figure 1. (a) Evolution of the pH according to the inoculum addiction; (b) Evolution of the pH according to the fermentation time.
“Attiéké” produced with starter of 10% had a better general acceptability. Statistical analysis showed that there were significant differences on the taste, texture and granulometry according to the starter rate used (Figure 5).
4. Discussion
When ready for use, the fermented cassava paste has a high level of micro-organism, indicating a microbial deterioration strongly related to earlier report by Assanvo [13]. That was justified because the fermented paste contained many micro-organisms (Lactobacillus, enterococci, yeasts and moulds) brought during the traditional starter incorporation, and which constitutes the
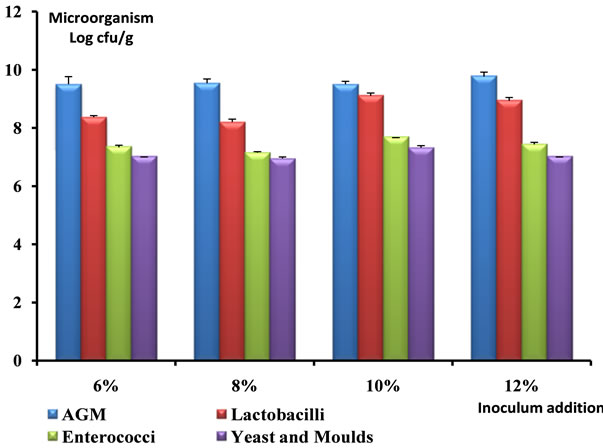
Figure 2. Evolution of the microbial load according to the inoculums addiction after 12 hours of fermentation.

Figure 3. Evolution of the microbial load according to the fermentation time with 10% of inoculums.
primary source of micro-organisms according to several authors [15-17]. The major micro-organisms present, were Lactobacillus sp. followed by enterococci, yeasts and moulds depending of the variability of inoculums amount and fermentation time used. This result confirms those of [13] which found the same order during their work on “Attiéké”. That could be explained by the fact that the lactic bacteria resist better to acid pH. Indeed, it is to be noted a fast acidification of pulp in less than 12 hours. This acidification, according to [18] would be associated with a high concentration of lactic acid (produced by lactic microflora) which prevents a degradation of the food product thereafter. It also should be noted that lactic bacteria, owing to the fact that they do not have system of breathing, are not very sensitive to the toxicity of free cyanides [19,20]. Moreover the hydrolysis of the starch places at the disposal of Lactobacillus an important source of dextrose which is the basic substance of their nutrition and their growth. In general, there were significant differences on the organoleptic attributes of

Figure 4. Evolution of the organoleptic attributes according to the fermentation time with 10% of inoculums.

Figure 5. Evolution of the organoleptic attributes according to the quantity of inoculums at 12 h of fermentation.
“Attiéké” produces according to the inoculums level and the duration of fermentation. Fermentation is related to the micro-organisms activity, their proportions (weak or strong) in a product caused necessarily a variation of the organoleptic quality. The factor combination “starter rate —time of fermentation” that was more appreciated by the panelists was 10%; 12 h, and thus having the most score for general acceptability. That could be due to the fact that Lactobacillus represent, in all the cases, the dominant microflora and that their metabolic properties are proven. According to [21], the lactic bacteria hydrolyzed the released starch of the cassava pulp leading to the production of sugars, lactic acids and acetic acids. According to [18], the lactic bacteria lead to develop the organoleptic qualities of the food products. That is possible thanks to the production of lactic acid, acetates, acetaldehyde, diacetyl, peptides and amino-acids which are precursors of flavour. The fermentation process was therefore important for the improvement of flavour of the product as well as for food safety by the reduction of cyanogenetic glucosides [3].
In brief, this study shows that “Attiéké” semolina (i.e. fermented cassava mash) texture is influenced by the inoculums level and the duration of fermentation, influencing texture and sensory qualities.
5. Conclusion
Sensory analysis revealed significant effects (p < 0.05), of inoculums quantity and fermentation time on the “Attiéké” quality. “Attiéké” obtained with 10% inoculums and after 12 hours of fermentation was identified as the optical inoculums amount and time for “Attiéké” production.
6. Acknowledgements
We thank Dr. Emmanuel Afoakwa for valuable comments, advice. This study was carried out with the technical financial support of Food Technology Laboratory (University of Abobo-Adjamé) and National Public Health Laboratory (LNSP) of Abidjan.