1. Introduction
To keep good condition sanitarily, various method and compound have been proposed. Inorganic materials containing silver have been attractive because of the effectiveness and the broad antibacterial spectrum and have been used at various scenes such as water sanitization [1, 2]. Various reports have been published about the mode of action of inorganic materials containing silver [3-5]. Among them, to investigate mode of action of the antibacterial activity of reactive oxygen species (ROS), single species of ROS was generated and affected to bacterial cells, then changes occurred inside cell was observed. We reported that ROS was detected in the suspension of silver loaded zeolite (Ag-Z) [6]. To the best of our knowledge, no report is found about the effect of reactive oxygen species on the chemotherapy. If the application of Ag-Z increases the susceptibility of pathogen to antibiotics, the appearance of the microorganism with the resistant to antibiotics might decrease. It will help to raise the level of the healthcare environment.
In this study, we investigated the effect of Ag-Z on the susceptibility of Escherichia coli to the antibiotics. The long time attack of hydroxyl radicals, highly reactive species, to the bacterial cell leads to decrease the viable cell count markedly. This phenomenon was the outcome after the various reactions proceeded. We studied to bring up the function of Ag-Z in detail. Therefore, we focused the early stage of the bactericidal processes. To observe the early stage of the antibacterial effect of Ag-Z, we treated the bacterial suspension of E. coli by Ag-Z to be the level which the decrease of viable cell count was not observed. It is also investigated the case of treatment of silver ions. The results might show the meaningful information to use silver loaded zeolite effectively and safely.
2. Materials and Methods
Nalidixic acid, rifampicin, benzyl penicillin and chloramphenicol were purchased from Sigma (MO. USA). Ag-Z was prepared by the ion-exchange method, as described previously [6]. Sodium-type zeolite (Na-Z, faujasite structure, Mizusawa Chemical Co. Ltd.) was used as the host compound. For antibacterial assay, 0.1 g of Ag-Z was fixed with polyvinylidene fluoride.
Escherichia coli NIHJ JC2 was used as a test microorganism. Bactericidal activity was estimated by counting viable cell after each treatment.
Ag-Z was suspended in the bacterial suspension and the suspension was stirred for 3 min. at 37˚C and aerated condition. The initial viable cell count was c.a. 107 CFU·ml–1. Ag-Z was removed from the bacterial suspension and the bacterial suspension was diluted by ten fold serial dilution with sterilized saline.
One hundred micro litter of each dilution was spread on Mueller Hinton (MH; Difco, MI, USA) agar plate (f: 90 mm). The MH agar contained antibiotics (nalidixic acid, rifampicin, benzyl penicillin and chloramphenicol) at various concentrations. After overnight incubation, colony found on the agar plate was counted and the viable cell count was calculated.
The effect of silver ion was also investigated. Solution of silver nitrate was added into bacterial suspension to be 10–5 mol·l–1 at the final concentration and the suspension was stirred at room temperature and aerated condition for 3 min. After that, the suspension was diluted immediately by ten fold serial dilution with sterilized saline. Following procedures were same as the case of Ag-Z.
3. Results and Discussion
Figure 1 showed the change in the viable count of E. coli on MH agar plate containing various concentration of rifampicin. The viability of E. coli decreased at higher than 2.5 μg·ml–1 of rifampicin without Ag-Z treatment. By the treatment of Ag-Z, no viable cell was observed higher than 2.0 μg·ml–1 of rifampicin. The treatment of silver ion decreased the viability at 2.2 μg·ml–1 of rifampicin and no viable cell was found higher than 2.5 μg·ml–1. The treatment of Ag-Z increased the sensitivity to rifampicin apparently. Figure 2 showed that the change of sensitivity to nalidixic acid of E. coli with treatment of Ag-Z and silver ion. By treatment of silver ion, no viable cell was found at higher than 1.2 μg·ml–1 of nalidixic acid. In the case of Ag-Z treatment and control condition, the concentration of nalidixic acid at which no viable cell was observed was higher than 1.5 μg·ml–1. The increase of sensitivity to nalidixic acid was observed in the case of silver ion treatment. The viable cell count with no treatment decreased at higher concentration of 14.5 μg·ml–1 of benzylpenicillin and no viable cell was found higher than 15.5 μg·ml–1 (Figure 3). Same change in the viable cell count was observed for the case of Ag-Z and silver ion treatment. Higher concentration of 2 μg·ml–1 of chloramphenicol decreased the viable cell count and no viable cell was found at higher than 2.5 μg·ml–1 for the control condition (Figure 4). No difference of sensitivity to chloramphenicol was found after the treatment of Ag-Z and silver ion.
When the viable cell count decreased or the ability to growth was lost, it is considered various interaction including reactions between bacterial cell and reactive species and reactions followed had been preceeded. Our objective in this study is to investigate the early stage of antibacterial process. Therefore, as the condition of antibacterial assay, the contact time of Ag-Z and the concentration of silver ion were determined to be 3 min and 10–6 mol·l–1, respectively. At the experimental condition, the treatment of Ag-Z and silver ion did not decrease the viable cell count of E. coli (data not shown).
The target process of antibiotics used in this study, rifampicin, nalidixic acid, benzylpenicillin and chloramphenicol are ribonucleic acid (RNA) synthesis, deoxyribonucleic acid (DNA) synthesis, cell wall synthesis and
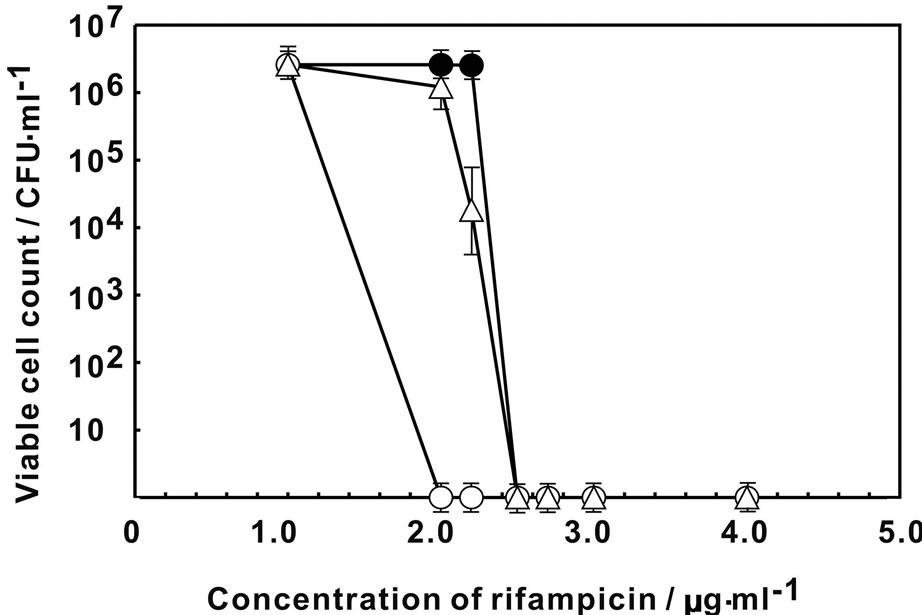
Figure 1. Change in the viable cell count of E. coli on MH agar plate containing various concentration of rifampicin. Open circles: E. coli treated with Ag-Z; Open triangles: E. coli treated with silver ion; Closed circles; Control condition.
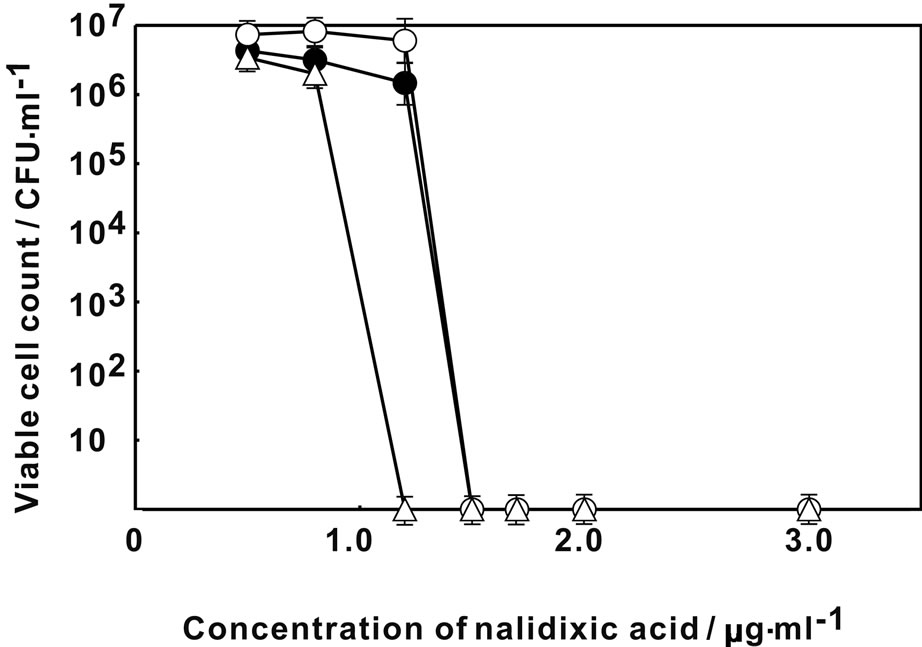
Figure 2. Change in the viable cell count of E. coli on MH agar plate containing various concentration of nalidixic acid. Open circles: E. coli treated with Ag-Z; Open triangles: E. coli treated with silver ion; Closed circles; Control condition.
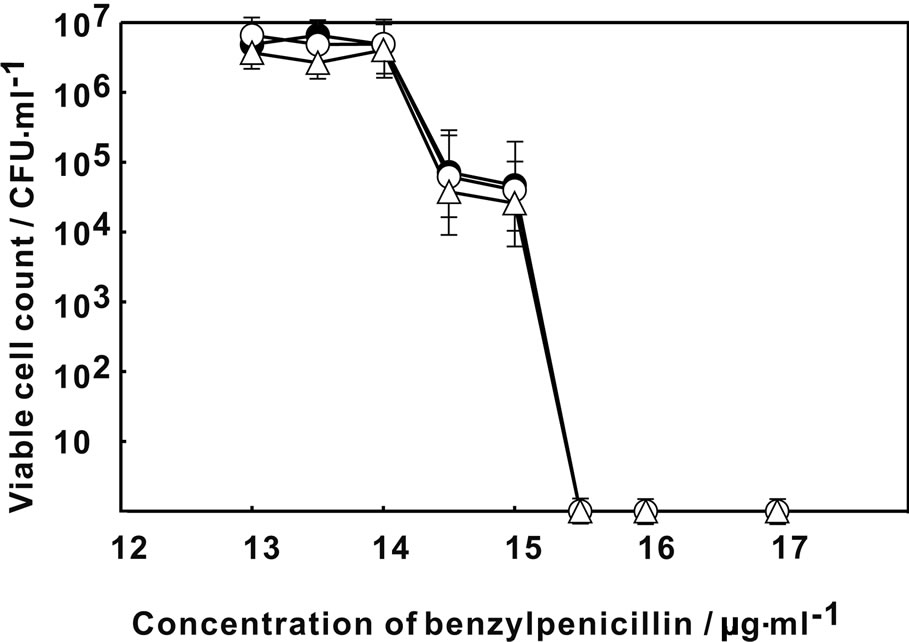
Figure 3. Change in the viable cell count of E. coli on MH agar plate containing various concentration of benzylpenicillin. Open circles: E. coli treated with Ag-Z; Open triangles: E. coli treated with silver ion; Closed circles; Control condition.
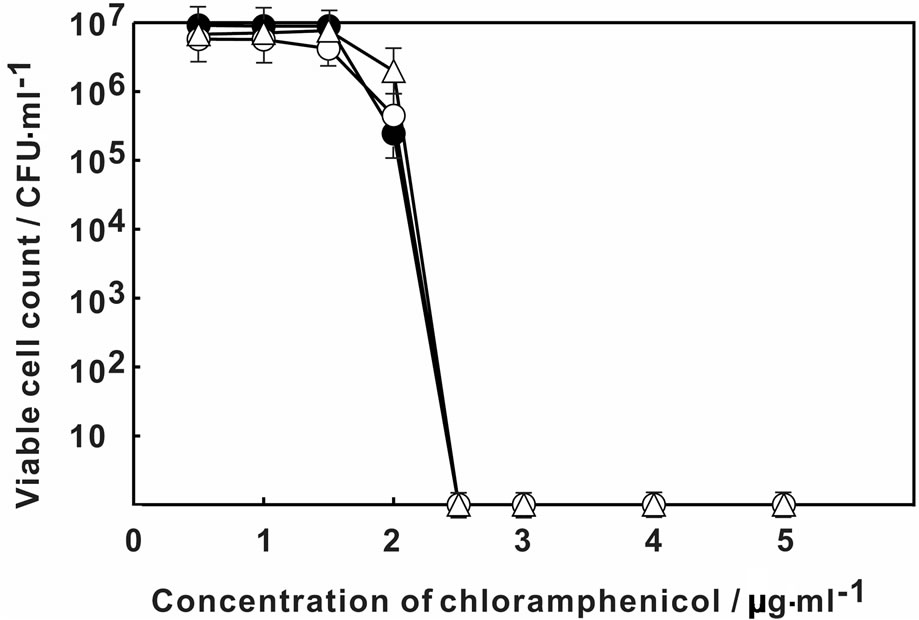
Figure 4. Change in the viable cell count of E. coli on MH agar plate containing various concentration of chloramphenicol. Open circles: E. coli treated with Ag-Z; Open traingles: E. coli treated with silver ion; Closed circles; Control condition.
protein synthesis, respectively. Neither treatment of Ag-Z nor silver ion change the sensitivity to benzylpenicillin and chloramphenicol at the comparison with control condition. Ag-Z treatment increased the sensitivity to rifampicin. Silver ion treatment increased the sensitivity to nalidixic acid and also increased to rifampicin for small amount. These results mean that Ag-Z enhances the effect of rifampicin whose target is synthesis of RNA. The effect is different from the one of silver ions.
During Ag-Z suspended in bacterial cell, ROS was generated [6]. Among them, hydroxyl radical was the final reduced species and showed the high reactivity. Hydroxyl radical damaged cell effectively by direct attack. Some authors insisted that hydroxyl radical plays important roll to the bactericidal activity of silver contained material [7-9]. Our results suggested that the Ag-Z treatment enhanced the inhibition of RNA synthesis process which proceeded inside the cell. Amount of hydroxyl radical might not be so much since the contact time was determined to be 3 min in this study. It is difficult for hydroxyl radical to penetrate cell membrane since at the contact it may be disrupted. Therefore, the species which can penetrate cell membrane and enhance the inhibition of RNA synthesis process might be super oxide anion.
We reported the formation of super oxide anion in the system studied previously [6], Katori et al. also pointed out the possibility for super oxide anion plays the important role to damage RNA synthesis system [10]. It is considered that at the early stage, super oxide anion is generated by Ag-Z and enhance the inhibition of RNA synthesis.
As the effect of silver ions to bacteria, some authors reported the interaction with ribosome to inhibit ATP production [11], lipid peroxidation [12], reactive oxygen species formation [13], and so on. In their experimental condition, the concentration of silver ion reached the level which induced the growth inhibition. At the condition, detectable phenomenon might include secondary and followed reactions. Our experimental conditions were shorter contact time and lower concentration of silver ion than theirs’. Results obtained suggested that silver ions could suppress DNA production process at mild condition as the growth behavior was similar to the control condition. Same result was reported by author which the experimental condition was determined similar level to ours [14].
Ag-Z can generate ROS at the environment where we live, not only in water. Therefore, ROS can interact with microbe suspended in air. Pathogen spread through the air induce serious situation and patient should be isolated. When Ag-Z is contained in the wall of hospital room, the pathogen suspending in air might interact with ROS generated by Ag-Z to increase the susceptibility to rifampicin. It is helpful to quell the nosocomial infection, such as tuberculosis.
Ag-Z could produce various kinds of reactive species. Bacterial cell was attacked by various patterns to lose the colony forming ability. Since the bactericidal activity of Ag-Z was supported with some mode of action, the pathogen with resistance to silver-loaded zeolite might be hard to generate.