Synthesis of Poly (Ether Ether) Ketone Sulfonated Membrane with Tin Dioxide Particles ()
Received 22 December 2015; accepted 12 February 2016; published 17 February 2016

1. Introduction
One of the greatest environmental concerns facing modern society is the use of fossil fuels for power generation [1] -[3] . Studies of the synthesis and use of polymeric membranes for fuel cells (FC) in electrolytes have been extensively investigated by several researchers who seek to consolidate the use of polymer electrolyte membrane fuel cell (PEMFC) in environmental with average temperatures until 100˚C [4] -[6] .
Nafion®, which is already established in the membrane market, is one of the products to be improved due to some drawbacks, such as its high cost and reduction in proton conductivity in an environment with low relative humidity [7] -[9] . Aromatic polymers, including poly (ether ether) ketone, arise as alternatives due to the possibility of increasing its conductivity by sulfonation reaction. Depending on the degree of sulfonation of the polymer, it may reduce its own dimensional and thermal stability [10] .
Obtaining of hybrid membrane based on sulphonated poly (ether ether) ketone (SPEEK) has been described in literature by incorporating inorganic particles, such as silica [11] , zirconia [12] , titanium [13] , among others [14] . However, the studies of the polymeric matrix with tin particles were only mentioned in some academic papers where the number of particles introduced in the matrix was around 20% - 50% [15] . The main use of these particles is to maintain, or even improve, its properties, such as the thermal and dimensional stability lost during the sulfonation reaction.
Other studies on organic-inorganic hybrid proton conductive membranes obtained by the sol-gel process showed lower conduction of protons [16] [17] . The tin oxide particles are stable in a wide pH range, allowing modifications with inorganic surfactants and higher working range during the sol gel process. However, it should be considered that the pure particle is not proton conductive, which makes necessary the inclusion of a third component in the system to perform such function.
The phosphotungstic acid (HPW) has a strong interaction with the inorganic phase, which promotes agglomerates inside this phase [16] -[20] . The protons may exist in their isolated acids form or in solvated form creating water molecules protonated (H3O+,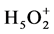 ) [21] -[24] .
) [21] -[24] .
In the present work two membranes groups were synthesized: one of the groups was used as control, containing SPEEK matrix with sulfonation degree (SD) of 55% and tin oxide particles, and the other one with the same composition of the control group but containing HPW particles. In that way, it was possible to evaluate the use of HPW in the membranes doping and its effect on proton conductivity.
2. Experimental
2.1. Instruments and Reagents
Poly (ether ether) ketone (PEEK) from Victrex (molar mass 288 g∙mol−1), sulfuric acid from Sigma Aldrich (ACS 98% of purity), SnCl4∙5H2O from Sigma Aldrich (97.5% of purity, molecular weight 350.58 g∙mol−1), n- methyl-2-pyrrolidone (NMP) from Vetec (99% of purity, molecular weight 99.13 g∙mol−1), and hydrated phosphotungstic acid (H3PW12O40 × H2O) from Fluka (AR). All of them were used as received.
2.2. Synthesis of the Tin Particles
The inorganic tin oxide (SnO2) has been obtained by precipitation of Sn (IV) in the form of oxyhydroxide, with addition of ammonium hydroxide in aqueous solution of SnCl4∙5H2O (0.25 mol∙L−1). The system was then kept in reflux for 2 hours. Further, it was subjected to a static dialysis process for about a week, until the peptization of the dialyzed material was completed, thus obtaining a limpid and transparent suspension. After dialysis the peptized material was dried in an oven at 100˚C, yielding the oxide.
2.3. Preparation of Hybrids Membranes
The polymer was sulfonated as described in the literature, obtaining the DS of 55% measured by titration [25] . First, for the preparation of membranes, the polymer was dissolved in NMP. The mass ratio of polymer/solvent mass was 20%. Subsequently, the oxide was also dissolved in NMP. The polymer solutions and oxide (varying the concentration of 2%, 4% and 6% w/w) already dispersed in the solvent were placed under stirring at 60˚C until complete dissolution. After the HPW was dissolved (40% of the mass of tin oxide) it was also added to the solution. The following mixture was transferred to a preheated glass plate at 70˚C for 24 hours, and then dried in a vacuum oven at 80˚C for 72 hours, finally obtaining the membrane. Two groups of membranes were synthesized: one containing only tin particles and SPEEK matrix, named SS2, SS4 and SS6, according to the amount of oxide in the matrix, and the other that, besides the SPEEK matrix and tin particles contained HPW particles, named SH2, SH4 and SH6.
2.4. Energy-Dispersive Analysis of X-Ray Emission (EDX Analysis)
As electrons of a given atom have different energies it is possible that the point of incidence of the beam determine the chemical elements in the sample, and thus identify which mineral is observed. Samples of the synthesized tin oxide were analyzed in the form of powder, free of water, and the analysis was performed under vacuum for 320 s. A Shimadzu EDX-720 analyzer was used.
2.5. Dynamic Light Scattering
The average diameter of the particles of tin oxide was determined by light scattering using the Malvern Zetasizer ZS equipment. The method used by the equipment is PCS (photon correlation spectrocopy) or dynamic light scattering, this method correlates the Brownian motion of the particles with its average diameter. The rating distribution of average particle diameter was made by dissolving 20 mg of particles in 2 mL of 0.5 M ammonium hydroxide. A 1 M HCl solution was used to perform measurements in neutral pH.
2.6. X-Ray Diffraction Patterns (XRD Analysis)
The diffraction spectra were obtained by a Rigaku Miniflex diffractometer using CuK source, with a wavelength of 1.5418 Å. Operating conditions were 30 kV voltage, 15 mA current, and diffraction angle scan range between 2 - 80, with a speed of 0.05˚/min.
2.7. Thermal Gravimetric (TGA) and Derivative Thermal Gravimetric (DTG) Analysis
The thermal stability of the membranes was investigated using thermogravimetric analysis (TGA). The analysis was performed on a TA Instruments Q-50 V20 10 Build device 36. The variation of weight loss versus temperature of the material was measured under continuous flow of N2, using a temperature range from 30˚C to 700˚C and speed heating of 10˚C/min.
2.8. Differential Scanning Calorimetry (DSC) Analysis
The glass transition temperature (Tg) of the membranes was determined by differential scanning calorimetry (DSC). The first run was used to eliminate the thermal history of the polymer and initially the samples were heated from 30˚C to 200˚C, at a rate of 10˚C/min. under N2 atmosphere. The samples were then cooled to 30˚C and a second heating was performed at a rate of 10˚C/min. The Tg was determined from the curves and data obtained in the second heating scan. It was used a DSC 204 F1 Phoenix Netzsch equipment.
2.9. Water and Ethanol Solution Uptake Test
Adsorption tests in water and in 20% ethanol solution (v/v) were performed at different temperatures (30˚C, 60˚C and 80˚C) and 24 hours interval. The samples were cut into circles and immersed in the solution. The weight taken at the end of the time range was taken as wet weight (WW). After the test the samples were dried in a vacuum oven. This weight was taken as dry weight (DW). The total of water content or adsorbed ethanol (TS) was calculated using the Equation (1).
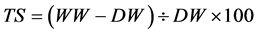 (1)
(1)
2.10. Pervaporation Test
The pervaporation test consists in forcing a fluid to permeate through a membrane due to the application of vacuum in one of its sides, while the other is in contact with the solution to be permeated. For this test it was used a device assembled in the laboratory. The membrane was cut into a disk-shape and then placed in the cell, where the upper side is continuously fed with an aqueous solution of ethanol or deionized water, and the lower face was under constant vacuum permeation. The vapor which permeated the membrane was condensed and collected in traps immersed in liquid nitrogen. The test was performed with the flow of water and with an aqueous solution of 20% ethanol by mass in the temperatures of 30˚C and 50˚C. The pervaporated materials were collected three times, on intervals of 1 hour between them. The amount of pervaporated material was calculated by the difference in mass of the dry trap, before running the test, and the trap with pervaporated material, calculating the average of the three measures.
2.11. Proton Conductivity
Impedance spectroscopy was used in this work. Membranes were preconditioned in deionized water at 60˚C for 1 hour and then placed between two graphite electrodes. The whole membrane/electrode set was placed in a stainless steel cell. The cell was then placed in a greenhouse and analyses were performed at the temperatures of 30˚C, 60˚C and 80˚C using an Autolab PGSTAT-30 equipment. The proton conductivity was calculated using the Equation (2).
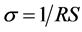 (2)
(2)
where σ = conductivity, R = resistivity and S = membrane area.
3. Results and Discussion
3.1. Energy-Dispersive Analysis of X-Ray Emission (EDX Analysis)
From the EDX analysis results it is possible to verify the synthesized components present in tin oxide and identify their compositions. The results (Table 1) showed that the synthesized oxide has 96.84% of tin oxide, a value considered suitable for applications as a load on the membranes.
3.2. Dynamic Light Scattering
From the synthesis performed polydisperse particles were obtained. Figure 1 shows the size distribution of the particles of the synthesized tin oxide. The values were in the micrometer range varying from 0.19 - 0.64 μm.
3.3. X-Ray Diffraction Patterns
The tin oxide has amorphous characteristics due to the amount of water absorbed, and depending on the heat
![]()
Table 1. Percentages of elements present in SnO2.
treatment performed, some peaks can be narrowed forming nanocrystals [15] [26] . Comparing the spectra obtained for pure tin oxide with other studies, the formed oxide corresponds to form crystallographic rutile. By the analysis of Figure 2(a) shows that the introduction of tin oxide increased the crystallinity of the membranes, verified by the narrowing of the peak in the region of 2θ = 20 as the amount of the oxide introduced into the membrane increased. Such behavior can be associated with the good dispersion of the oxide particles in the polymer matrix, generating points for nucleation.
Membranes that possess HPW, in addition to the tin oxide particles, exhibit behavior similar to that presented hybrid membranes with pure oxide particle. From Figure 2(b), it was verified that the introduction of HPW somehow interacted with the polymer, since the displacement of the amorphous halo with center 2θ = 20 at larger angles. This behavior can be explained, for the insertion of HPW may have hindered the packing of polymer chains and nucleation of smaller particles of tin oxide.
3.4. Thermal Gravimetric (TGA) and Derivative Thermal Gravimetric (DTG) Analysis
The thermal degradation of SPEEK occurs after three major events. The first phase occurs at approximately 100˚C and is related to the loss of free water. In other words, water which is not bound in the polymer structure. In the second step, around 200˚C, there is a loss of sulfonic acid groups, which do not always occur at the same temperature due to uneven distribution of it in polymer chain, the loss of residual solvent (in some cases, after the drying process, it can remain approximately 5% of the solvent), and also the loss of water attached in the polymer chains. These water molecules can be attached through either chemical bonds or interactions. The third stage, which takes place around 450˚C, is related to the degradation of the polymer chain [27] .
The values of mass loss and maximum degradation temperature displayed for each phase were taken from the TGA and DTG analysis presented in Figure 3(a) and Figure 3(b). In general, by adding tin oxide in the membranes, they showed a reduction in mass loss, even compared to the pure polymer.
In the values shown in Table 2 it can be seen that, regardless of the studied group of membranes, all had smaller amount of mass lost in the first stage, showing that the hybrid membranes have better conditions to maintain the polymer hydrates domains. However, the amount of particles on the membrane had little influence on the variation of weight loss in this region.
In the first event of the second region, the greater weight loss compared to pure polymer may be a consequence of the increased amount of solvent, which has been used to solubilize the polymer and it was also used to disperse the tin oxide and HPW particles. Regarding the maximum temperature of degradation, by increasing the amount of tin oxide it was observed a slight decrease of the maximum degradation temperature. Meanwhile, in the membranes of the SH group, there is an increase in the maximum degradation temperature with the increase in the percentage of HPW in SPEEK matrix. That is, an improvement in thermal stability, which means that the
![]() (a) (b)
(a) (b)
Figure 2. XRD patterns of the membranes containing SnO2 particles (a) and SnO2 and HPW particles (b).
![]()
Figure 3. TGA analyses of the membranes containing SnO2 particles (a) and SnO2 and HPW particles (b).
![]()
Table 2. Values taken from TGA and DTG analysis of membranes.
conductor groups remain on the membrane for a longer time when it is exposed to a higher temperature.
In the third phase, it was observed a mass loss reduction in both groups of hybrid membranes due to the increase of inorganic portion at the polymer matrix. There was an improvement in the thermal stability of the synthesized membranes since in the SS group to increase the oxide amount in up to 6% (SS6) showed a slight increase in the maximum degradation temperature relative to SPEEK membrane. Already in SH group the maximum temperature degradation was achieved with the SH4 membrane.
3.5. Differential Scanning Calorimetry (DSC) Analysis
The DSC analysis results, shown in Figure 4, investigated the Tg of the membranes. From Table 3 it was found that the incorporation of both particles, HPW and SnO2, decreased the Tg when compared to the pure polymer, thus providing greater mobility of the amorphous part of the polymer chain. The lower Tg of SH membranes group compared to SS group is related to the state of hydration of the membranes because, besides the change in
![]()
Figure 4. Influence of the particles in the membranes Tg measured by DSC.
![]()
Table 3. Tg values taken from the middle of the inflection point obtained from the second run in the DSC analyses.
the sulfonic group of the polymer, HPW particles provide greater hydrophilic character and increase the free volume present in these membranes [28] .
3.6. Water and Ethanol Solution Uptake Test
Figure 5 shows the result of swelling in water. It can be seen that all hybrid membranes maintains better dimensional stability at temperatures up to 80˚C, compared to pure polymer since a lower percentage of swelling of these membranes. It was also noted increased swelling at 80˚C for all membranes synthesized, this is due to the intense movement of the chains caused by temperature. With respect to SS membranes group to introduce up to 4% of SnO2 particles approximately 5% of reduced swelling compared to pure polymer when measured at 30˚C, and reduced the swelling by over 30% when measured at 80˚C. The HPW is a hygroscopic material [28] -[30] , and therefore the greatest degree of swelling of this membrane group than in the SS membranes group was expected. However, even when introducing the larger amount of HPW tested in this work the SH membranes showed less swelling than pure SPEEK membrane, especially when tested at 80˚C. This result shows that the particles, despite the increased mobility of the chains, promoted a degree of crosslinking to prevent excessive swelling.
Figure 6 shows the swelling results in ethanol solution. This test followed the same trajectory in the swelling test in water, but since the beginning of the test, at 30˚C, the hybrid membranes have been less swelling compared to pure polymer.
3.7. Selective Permeability of Membranes
Among the search by different sources hydrogen, alcohols such as methanol and ethanol have been widely considered for use in fuel cells. The Nafion presents problems known as methanol crossover, reducing the performance
![]()
Figure 5. Results of the water uptake tests.
![]()
Figure 6. Results of ethanol solution uptake tests.
of the cell [29] [31] . Therefore, to place the membranes in the current scenario, in order to be able to use alcohol as fuel, the developed membranes were also tested for permeability alcohol and the obtained values were shown in Table 4.
In Figure 7(a) and Figure 7(b) it can be seen that both groups of hybrid membranes have a better ethanol barrier property than pure SPEEK membrane. According to Xiang et al. (2011) this behavior can be explained by the amount of sulfonic acid groups in the polymer chains, since the passage of ethanol through the membrane preferentially occurs by hydrophilic domains. The ability of the hybrid membranes to be less permeable to ethanol solution is associated with the change in the structure of the membranes, since the insertion of the oxide and acid particles reduces the available cross channels that the alcohol molecules pass. That is, they form a physical barrier which prevents the passage path of alcohol molecules [22] .
![]()
Table 4. Values of the permeation test.
![]()
Figure 7. Permeability of the membranes in water and in ethanol solution at 30˚C (a) and 50˚C (b).
![]()
Table 5. Proton conductivity results of synthetized membranes.
3.8. Proton Conductivity
Table 5 shows the proton conductivity values obtained for the two groups of membranes. It is possible to verify a different behavior for each group. In the group composed only by tin oxide particles (membranes SS2, SS4 and SS6), there was an increase in the proton conductivity of the membranes. But when it was used the proportion greater than 4% of SnO2 proton conductivity values dropped again. In this group the maximum conductivity reached was 67.8 mS/cm at 80˚C. In the second group, membranes with tin oxide and HPW, it was found that the proton conductivity increased due to the addition of HPW. However, there were no major changes in the proton conductivity values comparing composite membranes with the SPEEK/SnO2 ones. So, larger amounts of HPW are needed to achieve higher conductivity values seen, showing that when HPW was introduced it preferably interacted with the polymer chains, giving them a more amorphous character.
4. Conclusion
The developed membranes series showed good capacity of absorption of water in temperatures up to 80˚C when compared to SPEEK. The introduction of tin oxide and HPW increased the mobility of the polymer chains. However, they caused no major changes in the thermal stability, with lower mass loss values than SPEEK ones, as verified at the TGA analyses. The highest value of conductivity was 67.80 mS/cm for the SS4 membrane. An increase was observed in proton conductivity in the SH group membranes due to the addition of HPW. This suggests that the concentration of HPW particles used in this work was not the best to achieve better proton conductivity results in the SH group membranes and the amount of HPW should be increased.
Acknowledgements
The authors thank CNPq for the financial support for this research.
NOTES
![]()
*Corresponding author.