Novel Antioxidant Peptides Derived from Enzymatic Hydrolysates of Macadamia Protein ()
Received 19 January 2016; accepted 13 February 2016; published 16 February 2016

1. Introduction
Enzymatic hydrolysis modification of proteins is a particularly attractive technique, as it can avoid severe operational conditions while enabling ease of control over the reaction to reduce by-products, achieve high product yields, and improve the various functional properties of the recovered proteins [1] . Peptides can be generated via enzymatic hydrolysis. Commercial papain, bromelain, actinidin and zingibain have been studied in their abilities of hydrolysis of protein due to their unique functions, wide accessibility, and cost-effectiveness [2] .
Many studies have demonstrated that bioactive peptides, the products of protein hydrolysis, beyond their nutritional value, exhibit biological activities, such as antioxidant activities [3] , opioid [4] , immunological implications [5] , metallocarboxypeptidase inhibitor [6] , antibacterial effect [7] , antihypertensive effect [8] , and antifungal effect [9] . In food industry, incorporation of particular synthetic antioxidants such as butylated hydroxyanisole (BHA), butylated hydroxytoluene (BHT), and tert-butylhydroquinone (TBHQ) into food products has been commonly used for the purpose of improving oxidative stability and shelf-life through inhibition of lipid oxidation [10] . However, there is growing concern about safety of these antioxidant compounds, particularly their potential carcinogenicity and genotoxicity [11] [12] . Thus, the development of specific peptides derived from food with potent antioxidant properties has attracted increasing attention which is believed to heighten safety for naturally derived products [13] .
Researchers have demonstrated that the hydrolysates of a number of food proteins, such as cod skin gelatin [14] , corn [15] , canola [16] , human milk [17] , hemp [18] , and sweet potato [19] , possess significant antioxidant activities. These antioxidant properties are believed to be attributed to their unique sequences, which have been reported to have stronger antioxidant activities than constituent amino acid mixtures [20] .
Macadamia nuts contain around 30% protein [21] and are a very good source of peptides. Here, we study the antioxidant ability of peptides from macadamia protein hydrolysis, aiming to improve our global understanding on macadamia nuts.
2. Materials and Methods
2.1. Materials
2,2-Diphenyl-1-picrylhydrazyl (DPPH radical) and 2,2’-azinobis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) were obtained from Sigma Chemical Co. (St. Louis, Mo., U.S.A.). Total antioxidant capacity research reagent (T-AOC) was purchased from Nanjing Jiancheng Bio-engineering Institute. Papain (enzyme activity > 200 myriad units/g) was a product of Guangxi Nanning Teemreal Biological Products Co., LTD. All other chemicals and solvents used in this study were of analytical grade.
HH-W600 electric heated water bath, 752N spectrophotometer, electronic balance, PHS-25 acidity meter, circulating water vacuum pump and low-speed desktop centrifuge were used in this experiment. HD-9707 computer ultraviolet detector was procured from Shanghai Jingke Industrial Co., LTD.
2.2. Preparation and Purification of Macadamia Protein
Box-Behnken Design (BBD) was used to estimate and optimize the yield of protein. Macadamia nuts were submerged in to PBS buffer pH 9 (9:91) for 2.5 h at 55˚C. The mixtures were centrifuged at 5000 rpm for 10 min. Supernatant was collected, adjusted pH to 4.6 and incubated at 4˚C for 1 h, followed by centrifugation at 5000 rpm for 10 min. The sample of crude macadamia protein was obtained by with vacuum freeze-dry.
DEAE cellulose cation-exchange chromatography and sodium dodecyl sulfatepolya-crylamide gel electrophoresis (SDS-PAGE) were used to purify macadamia protein and determine the distribution of molecular weight (MW). DEAE cellulose cation-exchange chromatography was performed using Chithra and Devaraj method [22] with slight modification. Crude macadamia protein was homogenized with phosphate buffer (0.01 M, pH 7.4) and passed through DEAE-Cellulose (Sigma) column (2.3 × 30 cm). Bound protein was eluted with a linear gradient of NaCl (0 ~ 0.5 M) at a flow rate of 0.8 ml/min and absorbance was monitored at 280 nm via HD-9707 computer ultraviolet detector. MW was determined by using SDS-PAGE according to the method described by Laemmli.
2.3. Enzymatic Hydrolysis of Macadamia Protein
The method of enzymatic hydrolysis of macadamia protein was similar to Hou et al. [23] [24] . Macadamia protein after purification was homogenized with phosphate buffer (0.01 M, pH 7.4) and incubated at 55˚C for 30 min to soften and dissolve the protein. Papain was added into the solution to perform the hydrolysis (20 g/l) at 37˚C for 180 min in a 120 rpm shaking water-bath incubator. The reaction was ended by heating the mixtures at 100˚C for 15 min to inactivate the protease. The suspension was centrifuged at 3000 g for 20 min and the supernatant was collected. Finally, the sample of peptides was obtained with vacuum freeze-dry technique.
2.4. Determination of Antioxidant Activities
2.4.1. Scavenging Activity of DPPH Radical
The scavenging activity of DPPH radical was measured according to the method of Rao [25] with a slight modification. An aliquot of 4 ml of sample solution was supplied with 4 ml of 0.1 mM DPPH in 95% ethanol. The mixture was incubated in the darkness for 20 min at room temperature. Subsequently, the absorbance of the resulting solution was measured at 517 nm with the spectrophotometer. The sample solutions were homogenized with 0.01 M PBS buffer pH 6.75 at a series of concentrations (0.5, 1, 1.5, 2, 2.5, 3, 3.5, 4, 4.5 and 5 mg/ml). Ethanol was used as a control. The absorbance of DPPH radical was recorded and radical scavenging capacity of the samples was calculated by using the following Equation (1):
DPPH scavenging activity (%) = 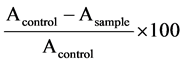 (1)
(1)
2.4.2. Scavenging of ABTS+ Radical
The ABTS radical cation (ABTS+) discoloration assay was performed according to Janet et al. [26] with modifications. ABTS+ was prepared by reaction of ABTS (7 mM in water) with potassium persulfate (2.45 mM final concentration) in the dark at room temperature for more than 12 h. Subsequently, the solution was diluted in ethanol and equilibrated in the dark at room temperature to give an absorbance of 0.50 ± 0.02 at 734 nm. Samples of 4 ml were homogenized in 0.01 M PBS (pH 6.75) at a series of concentrations (5, 10, 15, 20, 25 and 500 μg/ml), and then incubated for 20 min with 4 ml ABTS+ radical cation solution before measuring absorbance at 734 nm. Percentage inhibition of the ABTS+ radical was calculated using the Equation (2):
% Inhibition =  (2)
(2)
2.4.3. Evaluation of the Total Antioxidant Capacity
The total antioxidant capacity of peptides derived from enzymatic hydrolysates of macadamia protein was evaluated by the method of Hossain and Rahman [27] , with some modifications. An aliquot of 4 ml of sample solutions (0.5, 1, 1.5, 2, 2.5, 3, 3.5, 4, 4.5 and 5 mg/ml) were combined with reagent solutions in T-AOC as the procedures in Table 1. Subsequently, the solution was mixed well and incubated at room temperature for 10 minutes before the absorbance was measured at 520 nm against blank. One unit per mg peptides of total antioxidant capacity was determined by the absorbance, and was defined as the absorbance necessary to increase 0.01 per minute under assay conditions. The total antioxidant capacity was quantified using the following formulas (3) at our laboratory:
The total antioxidant capacity (unit/mg) = 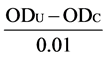 ÷ 30 × N ÷ Csample (3)
÷ 30 × N ÷ Csample (3)
where ODU and ODC were the absorbance values of samples and controls respectively, respectively. N and Csample were dilution multiple of reaction system and the concentration of the samples.
![]()
Table 1. The standard added samples in determination of antioxidant.
2.5. Statistical Analysis
All measurements were performed in triplicate and all results are presented as mean ± standard deviation of triplicate assays.
3. Results and Discussion
3.1. Characterization of Macadamia Protein by Chromatographic Analysis
DEAE cellulose cation-exchange chromatographic separation of macadamia protein samples was performed by the computer ultraviolet detector technique. Compared with membrane purification technology described by Selmane, Christophe and Gholamreza [28] , this technique allowed very fast and convenient separation of the proteins as the automated computer test. The traditional methods of processing for DEAE cellulose cation- exchange chromatographic separation used to measure absorbance and estimated the order of the peak in the main proteins with all kinds of spectrophotometers. These methods obviously affect the retention of bioactives and medicinal natural components that aid in the treatment of diseases. Besides, they are cost-effectiveness. As shown in Figure 1, the resolution achieved by this method enabled the complete separation and purification of the five major macadamia proteins. The preliminary results show that macadamia protein mixtures can be separated into five groups (peak 1, 2, 3, 4 and 5) after being eluted with a linear gradient of NaCl.
3.2. Characterization of Macadamia Protein by SDS-PAGE
SDS-PAGE was used to characterize the purified protein from DEAE cellulose cation-exchange chromatography. The distribution of molecular weight (MW) was also determined and the profile was shown in Figure 2. The SDS-PAGE separation of macadamia protein exhibited different protein bands of different sizes, and the apparent MW of each protein was estimated by gel-pro analyzer software to be 13.988, 18.40, 39.122 and 45.741 kDa for peak 3. Protein bands from the sample of crude macadamia protein were referred to as peak 1.
3.3. Antioxidant Activity of Enzymatic Hydrolysates
The hydrolysates of macadamia protein were freeze-dried and assayed for antioxidative activity using various
![]()
Figure 1. DEAE cellulose cation-exchange chromatography.
![]()
Figure 2. SDS-PAGE for macadamia protein, the numbers of 1, 2, 3, 4 and 5 were corresponding to the peaks in chromatography. 0 represented protein ladder.
antioxidant assays, including scavenging activity of DPPH radical, scavenging of ABTS+ radical and evaluation of the total antioxidant capacity assay.
3.3.1. DPPH Free Radical-Scavenging Activity
Figure 3 showed the DPPH radical-scavenging activity of the hydrolysates of macadamia protein at a series of concentrations (0.5, 1, 1.5, 2, 2.5, 3, 3.5, 4, 4.5 and 5 mg/ml). After the addition of different peptide fractions, increase in DPPH scavenging activity was observed. The results clearly indicated that the hydrolysates for 5 mg/ml exhibited the highest radical-scavenging activity on DPPH radical (56.43% ± 4.03%), while the lowest DPPH radical-scavenging was obtained from 0.5 mg/ml (10.31% ± 2.99%). This was in accordance with the results of Bougatef et al. [3] who found that the BHA (2 mM) and the hydrolysates from sardine viscera reached a maximum of 92.5% ± 1.7% and 53.76% ± 1.2% at only 500 μl, respectively, all hydrolysates at the series of concentrations showed lower radical-scavenging activity. However, they were obviously higher than SCPH-II (11.1% at 2 mg/ml) what has been reported by Zhong [15] . Moreover, the price graph for Figure 3 made an interesting point that we might get better at judging the difference and connation between the actual and ideal value of samples, and took action to reduce this difference.
3.3.2. Scavenging of ABTS+ Radical
The discoloration assay of ABTS+ radical can be used to determine antioxidant activity of both lipophilic and hydrophilic molecules, and is based on the reaction with hydrogen donating antioxidants. The higher activity was found in fractions from the hydrolysates of macadamia protein as shown in Figure 4 than the results observed in Figure 3. Moreover, the dynamic increase trended steeper than the former. When the concentration of hydrolysates was 50 ug/ml, the scavenging of ABTS+ radical was up to the standard of 96.30%. Compared with a parallel experiment described by Janet et al. [26] , there were similarities and differences between the process of conducting such a test and results. The similarities lied in higher scavenging activity and the differences between the two were that the latter dealt with molecular weight of peptides.
3.3.3. Evaluation of the Total Antioxidant Capacity
The total antioxidant capacity can be used to evaluate the potential antioxidant activity of the hydrolysates of macadamia protein based on its ability to reduce ferric (Fe3+) to ferrous (Fe2+) ion through the donation of an electron. All hydrolysates at various concentrations (0.5 - 5 mg/ml) showed some degree of electron donation capacity were shown in Figure 5. Moreover, the hydrolysates at higher concentrations were found to be better potent on total antioxidant capacity. However, this dynamic increase trend was not a linear development. The results clearly indicated that hydrolysate for 5 mg/ml exhibited the highest total antioxidant capacity (12.95 unites), while the lowest total antioxidant capacity was obtained from 0.5 mg/ml (3.24 unites).
![]()
![]()
Figure 3. DPPH scavenging activity of the hydrolysates of macadamia protein. Maximum value, median, minimum value were lines of large, medium and small values in triplicate measurements, respectively; average value represented average value in triplicate measurements. M: mg/ml.
![]()
![]()
Figure 4. Scavenging of ABTS+ radical of the hydrolysates of macadamia protein. Maximum value, median, minimum value were lines of large, medium and small values in triplicate measurements, respectively; average value represented average value in triplicate measurements.
![]()
Figure 5. Evaluation of the total antioxidant capacity of the hydrolysates of macadamia protein.
4. Conclusions
In the present study, DEAE cellulose cation-exchange chromatographic separation and SDS-PAGE of were performed to purify and characterize macadamia protein. The relative antioxidant ability of the peptides derived from enzymatic hydrolysates of macadamia protein was investigated through some in vitro models such as scavenging activity of DPPH radical, scavenging of ABTS+ radical and evaluation of the total antioxidant capacity assay. The antioxidant activity of peptides derived from macadamia protein on DPPH radical was lower than on ABTS radical. This might because ABTS radical was easier to scavenge than DPPH radical as indicated by previous studies [29] .
The enzymatic hydrolysates of macadamia protein could be a good source of antioxidant peptides. The difference and connation between the actual and ideal antioxidant values of the hydrolysates were well studied. Further studies were warranted for the isolation and identification of individual peptide and also in vivo studies were needed for a better understanding of their mechanism of action as antioxidant.
Acknowledgements
We gratefully acknowledged Key Laboratory of Tropical Crop Products Processing Ministry of Agricuture in China for providing some support and facility for this work.