The ecological context of bilateral symmetry of organ and organisms ()
The phenotypic plasticity of the organisms under stress may have great evolutionary importance, once stress responses can become permanent within the group when fixed by natural selection. However, opposite to the evolutionary importance of the environmental changes, when the level of environmental oscillations starts to be stressed,

Figure 1. Interaction between the processes that generate developmental instability (DI) and fluctuating asymmetry (FA). The phenoltype can be produced by different ways, ranging according to the plasticity of the genotype and the environmental conditions; under non-stress conditions (solid lines) or under stress conditions (dashed lines).
the developmental stability (DS) of the organism may be perturbed. DS is a concept that combines morphogenetic responses to internal and external factors. DS can be denominated by the ability of the organisms to minimize disruption occurred at random during development [2]. DS has been used to monitor the effects of anthropogenic and natural factors of stress on living organisms. The DS of an organism is reflected in its ability to produce an ideal phenotype under a particular set of conditions [3]. Higher DS involves the generation of a phenotype predicted by the genetic information’s associated with the optimal environment, while lower DS is associated with a phenotype interacting with a perturbed ambient (Figure 1). An indicator of DS in plants is the fluctuating asymmetry (FA) of organs or organisms with bilateral structure, which higher DS has been associated with higher symmetry.
Theoretically every organ or organism with bilateral structure has a perfect symmetry, i.e. there is a pattern of symmetry normally distributed in the population [4]. This context can be discussed also at biochemistry level, once there are evidences showing that the symmetry is also important for production and function of proteins [5]. Deviations from the perfect symmetry of organs or organisms with bilateral structure occur due to the inability of the organism to contain disorders from environment or endogenous conditions during its development. It reflects the inability of the organism to maintain precision development during ontogeny [6]. It has been suggested that more symmetrical individuals have greater DS, reproductive success and survival rate [4]. Organs and organisms with bilateral structure may exhibit fluctuations on their symmetry under stress conditions, which is known as FA when it is non-targeted and random [7]. Lower FA is associated with greater DS [2,4]. Individuals unable to exhibit plastic responses to developmental disorders can present slight deviations in the bilateral symmetry (Figure 1). FA, an analysis of the symmetry normally distributed in a population (discussed below), has been often used to measure the effects of environmental perturbations in a wide range of organisms [2,8], since FA can indicate the level of stress which the individual and/or population are subjected to [9,10].
2. THE ECOLOGICAL CONTEXT OF THE SYMMETRY
The symmetry deviations in a bilaterally symmetrical organ can come in the form of directional asymmetry, antisymmetry or fluctuating asymmetry (FA) [11]. Of these three kinds of asymmetry, only FA is useful for assessing the effects of injury during development given that reflect DS of the organism [12]. Studies involving FA in response to stress has been investigated in a wide range of organisms with bilateral structure. The stress has been reported as a disturbing source capable to induce qualitative and quantitative changes in the symmetry of organs or bodies with bilateral structure, such as reported in birds [13], mammals [14,15], fish [16], insects [17,18], human [19-21], and plants [22-27). Thus, FA has been used as a marker of the biologic quality of individuals [19] as well as an ecological indicator to identify environmental stress [12].
2.1. Face Symmetry as a Trait for Sexual Selection in Mammals
In mammals, the face symmetry has been related to attractiveness and is supposed to be an important trait for sexual selection in humans and monkeys [21,28]. Individuals more symmetrical are considered more attractive, at least in occidental human societies. For example, if you reader ask some girl about the attractiveness of Brad Pitt’s face or a man about the attractiveness of Giselle Bündchen’s face, for sure the answer will most likely be a resounding yes. Despite the famous and the financial success of both, there are evidences showing that attractiveness is closed related to the body or face symmetry [29-31] (Figure 2). Higher facial symmetry or lower facial fluctuating asymmetry enhances the perception of facial attractiveness [20]. Moreover, there are evidences showing that the degree of FA is negatively related to woman’s potential fertility [19] and to the health condition in monkeys [28]. More symmetrical women have from 13% to 28% higher average levels of estradiol than

Figure 2. Symmetry-attractiveness relationship. Data from Brown et al. 2008, PNAS, 105:12938-943. ©2008 by National Academy of Sciences—PNAS.
the less symmetrical one along the menstrual cycle [19]. Furthermore, the women perception to detect male facial symmetry also ranges along the menstrual cycle. It occurs according to the progesterone levels, whereas women are best at detecting male facial symmetry during the menses phase of their cycle (the phase with lowest progesterone levels) [20]. In summary, in animals, especially in mammals, the symmetry of traits that are considered attractive can be used as reliable markers of the biologic condition and reproductive potential of individuals, which may be sufficient to create a selection pressure to choose symmetrical mates [28].
2.2. Symmetry and Good Runners: Why You Cannot Run Like Usain Bolt?
Beyond of the face symmetry, other parts of the human body has been studied. The symmetry of human legs traits have been recently used to explain the success of Jamaican athletes in sprint running [32]. The transition from humans ancestors (more suited to living in trees) to modern humans (with a skeleton that could support walking and running) was pressured by climate change in Africa, whereas change from a forested ecosystem to savannah. Briefly, humans evolved to run [33]. However, remains unclear why some genotypes can run faster than others, including friends of training which have near phylogenetic origin and the same conditions for training. What’s the advantage of good runners? Should be better morphologically adapted; genetically privileged; or both? Trivers and collaborators developed a nice long-term study in Jamaican measuring the symmetry of legs of Jamaican children’s associating this symmetry with the running performance of the adults 14 years latter [32,34]. Interesting, lower FA measured in 1996 was positively related to sprinting performance in 2010 [34]. It means that children’s more symmetrical have more capability to develop a good adult runner. Thus, FA of legs can be used as a marker for selection of runner’s while children, which can anticipate the training of genotypes which have higher potential to develop an Olympian athletes.
Trivers and collaborators suggest that high symmetry in the legs—especially knees—may underlie at least some of the advantage that Jamaican sprinters enjoy over their competitors, which can have a genetic origin related to heterozygosity [32]. It’s important to highlight that FA have a genetic origin [4,8]. In this context, Enriquez and Gullans [35] discuss that the ability of the Olympian athletes to run faster than the “normal people” comes from a minimum set of particular performance-enhancing genes. It means that the capability to run is due the genetic information of the individual, separating gifted from the ungifted people [35]. Altogether, is notable that best runners may have both advantages; i.e. is better morphologically adapted to run (have lower FA and greater DS) as well as is genetically privileged (have a set of particular performance-enhancing genes).
2.3. Flower Symmetry Determine the Success of Pollination
Similar to the studies about leaves, flowers symmetry has been extensively studied also. Such as leaves, flowers are a very sensitive organ to environmental changes. Their morphology is highly influenced by environmental stress. The deviations on flowers symmetry affect not only their morphology but also the success of the pollination process. It was showed that insects prefer symmetrical flower over asymmetrical ones independent of pollinator rewards, floral color, floral odor and flower size [22]. The hypothesis that higher symmetry in an individual or organ can be a marker of the phenotype quality (already discussed above for humans [19]) is also valid for insect-flower interaction. Symmetrical flowers produce more nectar and enriched in quality (sugars) compared to asymmetrical flowers [31,36]. It suggests that the preferential visits of insects to symmetrical flowers comprise one selection pressure for symmetry [22], whereas these preferences may have an innate basis [6].
3. CONNECTING STRESS TO PLANT ASYMMETRY
According to the classical definitions the stress can be considered as environmental pressures on organisms [37] or responses of organisms to environmental disturbances [38]. Pahlich [39] argues that stress can be described as the state in which the increase in demand for the plant can take it to an initial destabilization of functions, followed by a standardization and development of resistance. However, if the tolerance limits are exceeded and adaptive capacity is overcome, the result is often permanent damage or death. When the tolerance limits are exceeded, the stress tends to increase the phenotypic variation of the organism [40], reduce the homeostasis of a given biological system [41], generate symmetry deviations in organs with bilateral symmetry as well as to decrease the DS (2,4). Further, as plants are the basis of the trophic chain, perturbation on plant fitness tends to perturb the microclimate and the fauna around [42].
Although the stress concept has been used mainly as a dangerous factor for plants, it’s important to highlight that stress period are important to promote biological diversity through phenotypic plasticity. The adaptation, in a biological context, occurs along the evolutionary time, under pressure of natural selection, which involves the selection of complex of genes co-adapted [4]. Thus, adaptation can be processed not only at individual scale and cannot be confounded only with morphological changes in response to stress, since a character may have or not an adaptive role [43]. The genetic information of the individual can determine tolerance or susceptibility to the stress. Tolerance may be associated with complex of genes co-adapted to the stress condition [4] as well as to an internal memory of the genotype [44,45]. The ability of a plant genotype to acclimate to the stress period depends on a complex network of interactions that generate responses at different levels and organs of the plant in order to keep the growth and the development under these conditions. Usually the plasticity of the higher levels of the hierarchy of biological systems (e.g. anatomical and morphological) are more stable than low levels (e.g. molecular, biochemical and physiological). Probably the higher stability of the highest levels occurs to prevent disturbances at low levels, contributing to greater stability of the whole plant [41,46]. In this context, when the stress is identified at morphological level generally means that the plasticity of the physiological and molecular networks were unable to avoid the effect of the stress. It highlights the importance of tools to detect the fitness of the plant at morphological level, such as FA analysis.
Plants under Stress Tends to Produce Asymmetrical Leaves
Land plants exhibit FA in response to anthropogenic and natural factors of stress. FA were reported in leaves of a wide range of species under different levels of pollution [23], water deficit [26], ultraviolet radiation (UV) [27,46], soil disturbance [25], shade conditions [47], and post-fire [42]. Souza et al. [26] reported that the increase in FA in leaves, performed through the determination of symmetry deviation (sd) (see below), was due the variations in net photosynthesis, which induce the individual to a non-uniform growth of the leaves. Daloso et al. [46] showed that high energy level of UV may promote disturbances in the process of differentiation of tissues of young leaves, resulting in asymmetrical leaves. In another study under UV stress, FA also showed to have a direct relationship with UV stress. Further, there is a linear correlation between FA and potential quantum yield of photosystem II ( ) (Figure 3), a sensitive and usefulness physiological parameter to detect plants under stress [48]. It demonstrates the sensitivity of FA to analyse the fitness of the plant under disturbances occurred during its development. Altogether, these studies showed that FA in leaves can be a promise strategy to determine the level of stress which the individual is subjected.
) (Figure 3), a sensitive and usefulness physiological parameter to detect plants under stress [48]. It demonstrates the sensitivity of FA to analyse the fitness of the plant under disturbances occurred during its development. Altogether, these studies showed that FA in leaves can be a promise strategy to determine the level of stress which the individual is subjected.
4. HOW TO MEASURE SYMMETRY DEVIATIONS?
The symmetry deviations have been performed basically measuring the width of the left side (L) and right side (R) of bilateral structures, such as leaves (Figure 4). Firstly, symmetry deviations (sd) are established comparing L and R sides through Student t-test (P < 0.05) in order to check if there is occurrence of an directional asymmetry [11]. Further, in case that the asymmetry is not directional (P > 0.05), i.e. is fluctuating asymmetry, there are different methodologies which can be used to calculate the sd: 1)—The sd can be determined using the methodology proposed by [49], in which the data are log transformed and normalized using Box-Cox transforma-
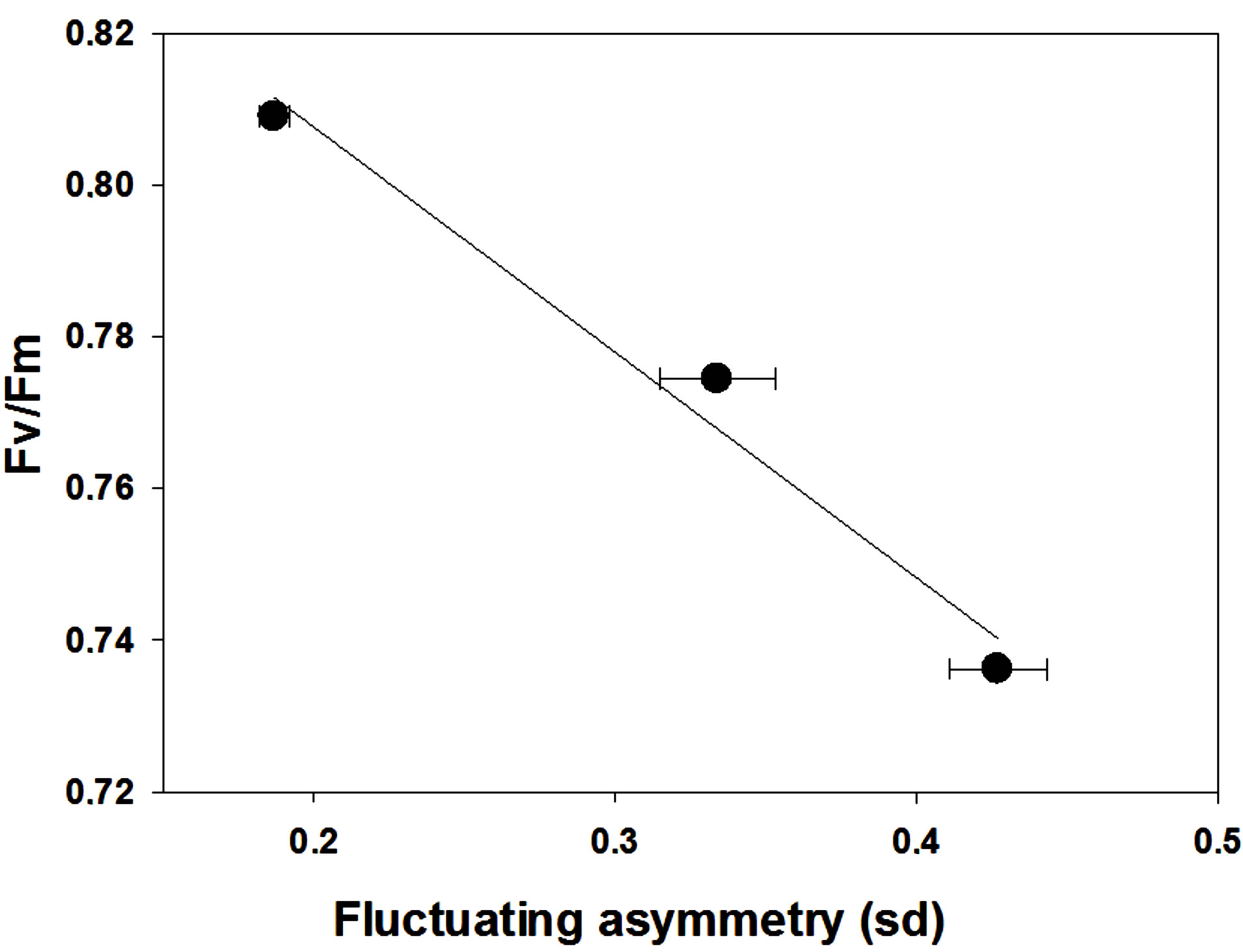
Figure 3. Effect of ultraviolet radiation on potential quantum yield of photosystem II ( ) (leaft) and fluctuating asymmetry relationship in Glycine max leaves. The leaves were submitted to 0’, 30’ or 60’ minutes per day of ultraviolet radiation (UV) during four weeks. Fluctuating asymmetry was measured through symmetry deviations (sd) of leaves following the methodology proposed by Cowart and Graham (1993). The exposition to UV results in decrease of both sd and Fv/Fm. A linear regression was performed following the equation f = y0 + a*x (R2 = 0.95). Data from Silva et al. 2011. Idesia, 29(3): 45-52.
) (leaft) and fluctuating asymmetry relationship in Glycine max leaves. The leaves were submitted to 0’, 30’ or 60’ minutes per day of ultraviolet radiation (UV) during four weeks. Fluctuating asymmetry was measured through symmetry deviations (sd) of leaves following the methodology proposed by Cowart and Graham (1993). The exposition to UV results in decrease of both sd and Fv/Fm. A linear regression was performed following the equation f = y0 + a*x (R2 = 0.95). Data from Silva et al. 2011. Idesia, 29(3): 45-52.
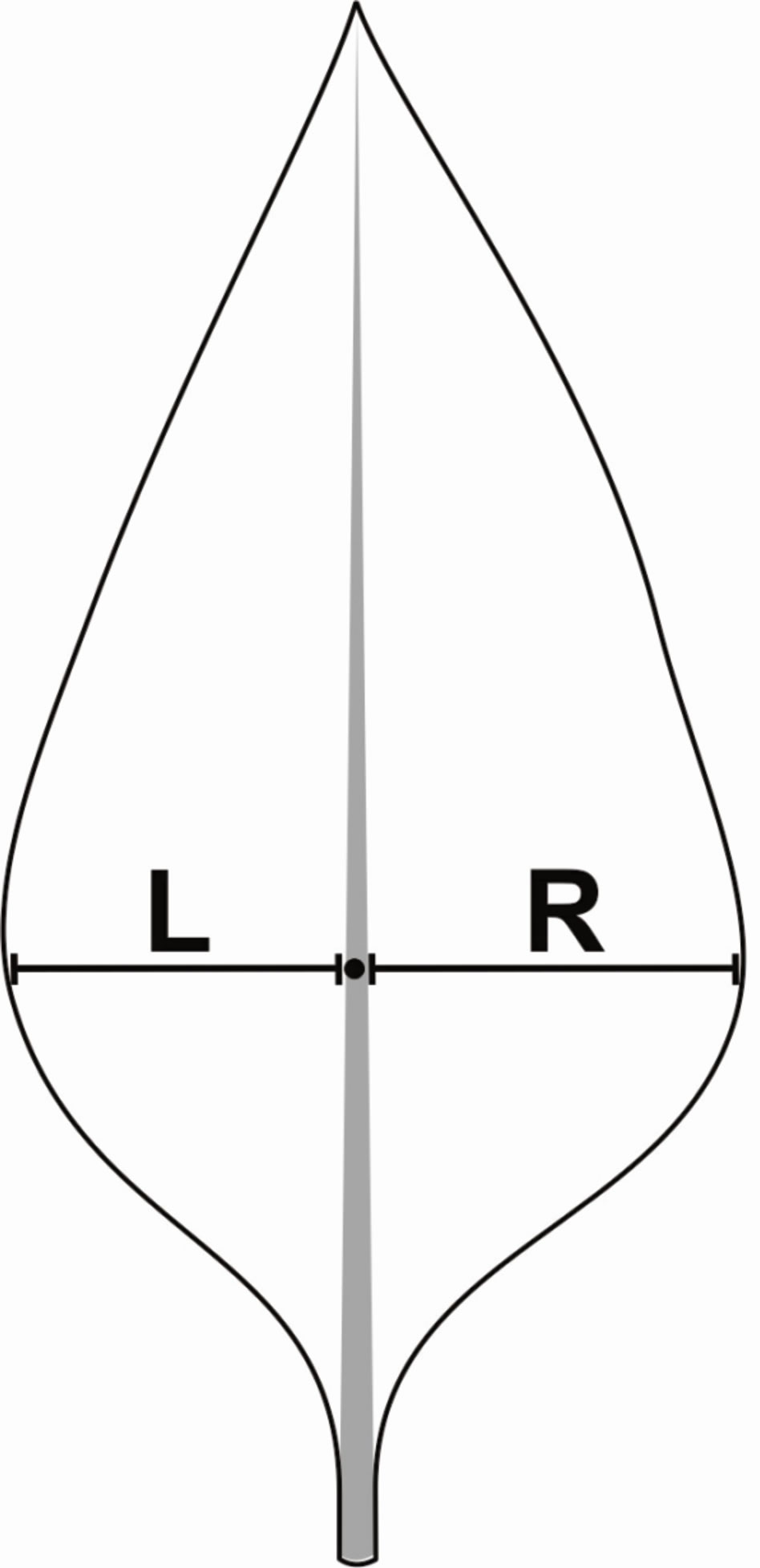
Figure 4. Hypothetical leaves of Glycine max showing the sample units of symmetric analysis. L – Leaf left width, R – Leaf right width. The L and R values are used for determinate maximum width of leaf and for calculation of symmetry deviations (sd).
tion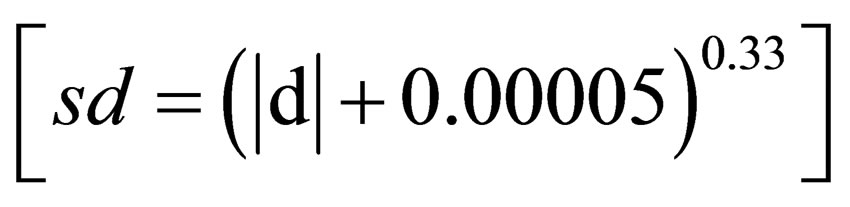 , where
, where
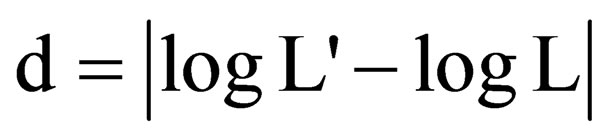 ; or 2) using the methodology proposed by Nagamitsu et al. [22], whereas the sd is calculate following the equation
; or 2) using the methodology proposed by Nagamitsu et al. [22], whereas the sd is calculate following the equation . Both methodologies can be used to determine sd, however the methodology proposed by Cowart and Graham [49] can be higher sensitive to detect slights deviations on the symmetry. Moreover, the Box-Cox transformation used in this methodology can remove some differences between the area of the leaves, given a greater homogeneity for the data [26,46].
. Both methodologies can be used to determine sd, however the methodology proposed by Cowart and Graham [49] can be higher sensitive to detect slights deviations on the symmetry. Moreover, the Box-Cox transformation used in this methodology can remove some differences between the area of the leaves, given a greater homogeneity for the data [26,46].
5. CONCLUDING REMARKS
The nature is constantly changing over the time. The changes in the environment conditions are essential to keep the functionality of the ecosystem. The level of oscillations ranges according to the time and space and can be stressed for the organisms when the limits of tolerance of a given genotype are exceeded. Under stressful conditions, the genotypes may show some deviations in their perfect bilateral symmetry, which can be identified through the analysis of fluctuating asymmetry (FA). Higher symmetry or lower fluctuating asymmetry has been associated with higher genotype quality. Further, symmetry of bilateral organs, such as flowers, is important for the maintenance of the rates of pollination, showing that symmetry in the nature is not a particular trait of the individual, but it is a trait that contributes for the functioning of the whole ecosystem. Although this review showed examples of FA in a small number of organisms, it’s important to highlight that this kind of analysis can be carried out in every organ or organism with bilateral structure.