Advances in lignocellulosic biotechnology: A brief review on lignocellulosic biomass and cellulases ()
2. CHARACTERISTICS OF LIGNOCELLULOSIC BIOMASS
Lignocellulosic materials including agricultural wastes, forestry residues, grasses and woody materials have great potential for bio-fuel production. Typically, most of the agricultural lignocellulosic biomass is comprised of about 10% - 25% lignin, 20% - 30% hemicellulose, and 40% - 50% cellulose [3,8]. Cellulose is a major structural component of plant cell walls, which is responsible for mechanical strength and chemical stability to plants. While, hemi-cellulose macromolecules are often repeated polymers of pentoses, and hexoses. Due to the genetic variability among different sources hemicellulose macromolecules are also vary in structural composition [7]. Lignin contains three aromatic alcohols (coniferyl alcohol, sinapyl alcohol and p-coumaryl alcohol) produced through a biosynthetic process and forms a protective seal around the other two components i.e., cellulose and hemicelluloses (Figure 1) [7,9]. In general, the composition of lignocellulose highly depends on its source whether it is derived from the hardwood, softwood, or grasses. Lignocellulosic biomass has a complex internal structure and comprised of a number of major components that have, in turn, also complex structures. Table 1 shows the typical chemical compositions of all these three components in various lignocellulosic materials that vary in composition due to the genetic variability among different sources [7,8,10,11].
3. PROPERTIES OF CELLULOSE
Plant biomass contains 40% to 50% of cellulose molecules which are fibrous in nature, insoluble, crystalline polysaccharide. Being the most abundant and easily available carbohydrate polymer all around the earth which is a major polysaccharide constituent of plant cell wall, composed of repeating (1,4)-D-glucopyranose units, which are attached by β-1,4 linkages with an average molecular weight of around 100,000 [12]. Naturally cellulose molecules are exists as bundles which aggregated together in the form of micro-fibrils order i.e., crystalline and amorphous regions [3,13,14]. The structure of one

Figure 1. Chemical structure of lignocellulosic material; (a) Building blocks/units of Lignin; (b) Xylose unit of hemicellulose; and (c) Cellulose. (Adopted from Iqbal et al. [7]).
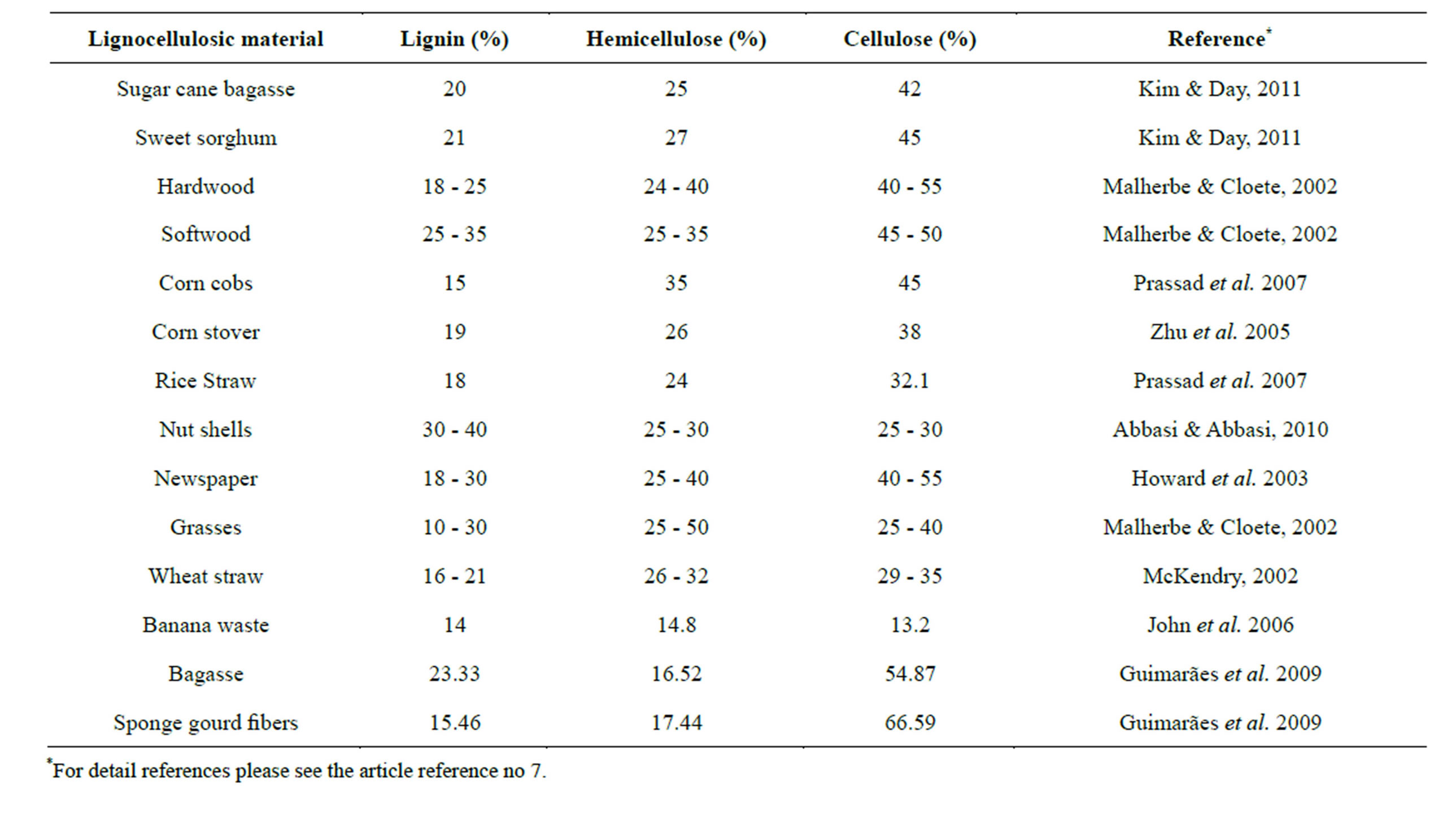
Table 1. Percent composition of lignocellulose components in various lignocellulosic materials (Adopted from Iqbal et al. [7]).
chain of the cellulose polymer is presented in Figure 1 [7]. Cellulose has attracted worldwide attention as a renewable resource that can be converted into bio-based products of commercial interests. Therefore, cellulose has been used as a potential energy source for a wide variety of organisms including fungi and bacteria to extract many useful products e.g., enzymes.
4. PROPERTIES OF HEMICELLULOSE
The second most abundant polymer after cellulose is hemicellulose which is heterogeneously branched in nature. The backbone of the hemicellulose polymer is built up by sugar monomers like xylans, mannans and glucans, with xylans and mannans being the most common [15], in this case xylanases are the enzymes involved in its degradation. Similar to cellulases the xylanases can act synergistically to achieve hydrolysis, predominant enzymes within this systemare endo 1, 4 b-xylanases which attack the polysaccharide backbone, and b-xylosidases. Hemicellulosic biomass contains 25% to 35% of hemicellulose, with an average molecular weight of <30,000. Cellulose and hemicellulose binds tightly with non-covalent attractions to the surface of each cellulose microfibril. Hemicellulose, degrade quickly due to its amorphous nature [16]. Among other important aspects of the structure and composition of hemicellulose are the lack of crystalline structure, mainly due to the highly branched structure, and the presence of acetyl groups connected to the polymer chain.
5. PROPERTIES OF LIGNIN
Lignin is generally the most complex and smallest fraction, representing about 10% to 25% of the biomass. It has a long-chain, aromatic polymer composed largely of phenyl propane units. Lignin acts like a glue by filling the gap between and around the cellulose and hemicellulose complexion with the polymers. It is present in almost all kind of cellulosic plant biomass and acts as a protective sheet against cellulosic and hemicellulosic components of the biomass materials. Lignin is consists of multifarious and large polymer of phenyl-propane, methoxy groups and non-carbohydrate poly phenolic substance, which bind cell walls constituent together [16]. Among them phenyl-propanes are the main blocks of the lignin share in biomass residues. These phenyl-propanes denoted as 0, I, II methoxyl groups attached to rings give special structure I, II and III. These groups depend on the plant source which they are obtained. Structure I exist in plants (grasses) and structure II found in the wood (conifers) while structure III present in deciduous wood.
6. BIOTECHNOLOGICAL IMPORTANCE OF LIGNOCELLULOSIC BIOMASS
A large magnitude of lignocellulosic biomass resources are available as potential candidate that are convert able into high value bio-products like bio-ethanol/bio-fuels [2]. The detailed step by step information on the conversion of lignocellulosic biomass into fuel ethanol is illustrated in Figure 2 [7]. From the last few decades there has been an increasing research and developmental interests in the value of lignocellulosic biomass. In this regard a considerable improvement from the green biotechnology related to lignocellulose biomass has appeared. The ever increasing costs of fossil fuels and their greenhouse effects are a major concern about global warming. Therefore, all these issues are creating a core demand to explore alternative cheaper and eco-friendly energy resources [16-18].
7. FROM CELLULOSE TO CELLULASES
Cellulose is a fibrous, insoluble, crystalline polysaccharide. It is a major polysaccharide constituent of plant cell walls, composed of repeating D-glucose units linked by β-1,4-glucosidic bonds [19] and being the most abundant carbohydrate polymer on earth. Cellulose has attracted worldwide attention as a renewable resource that can be converted into bio-based products and bio-energy. But nowadays, enormous amounts of agricultural, industrial and municipal cellulose wastes have been accumulating or used in efficiently due to the high cost of their utilization processes [20]. Therefore, it has become of considerable economic interest to develop processes for the effective treatment and utilization of cellulosic wastes as cheap carbon sources. Cellulose is used as a food source by a wide variety of organisms including fungi, bacteria, plants and protists, as well as a wide range of invertebrate animals, such as insects, crustaceans, annelids, mollusks and nematodes. These organisms possess cellulases and the complete enzymatic system of them include three different types, that is, exo-β-1, 4-glucanases (EC 3.2.1.91), endo-β-1,4-glucanases (EC 3.2.1.4), and β-1,4-glucosidase (EC 3.2.1.21) [3,5]. These enzymatic components act sequentially in a synergistic system to facilitate the breakdown of cellulose and the subsequent biological conversion to an utilizable energy source, glucose. The endo-β-1,4-glucanases randomly hydrolyzes the β-1,4 bonds in the cellulose molecule, and the exo- β-1,4-glucanases in most cases release a cellobiose unit showing a recurrent reaction from chain extremity. Lastly, the cellobiose is converted to glucose by β-1,4-glucosidase.
8. STATUS & PROSPECTS OF CELLULASES
Cellulase is an important and essential kind of enzyme for carrying out the depolymerization of cellulose into fermentable sugars. As a major resource for renewable
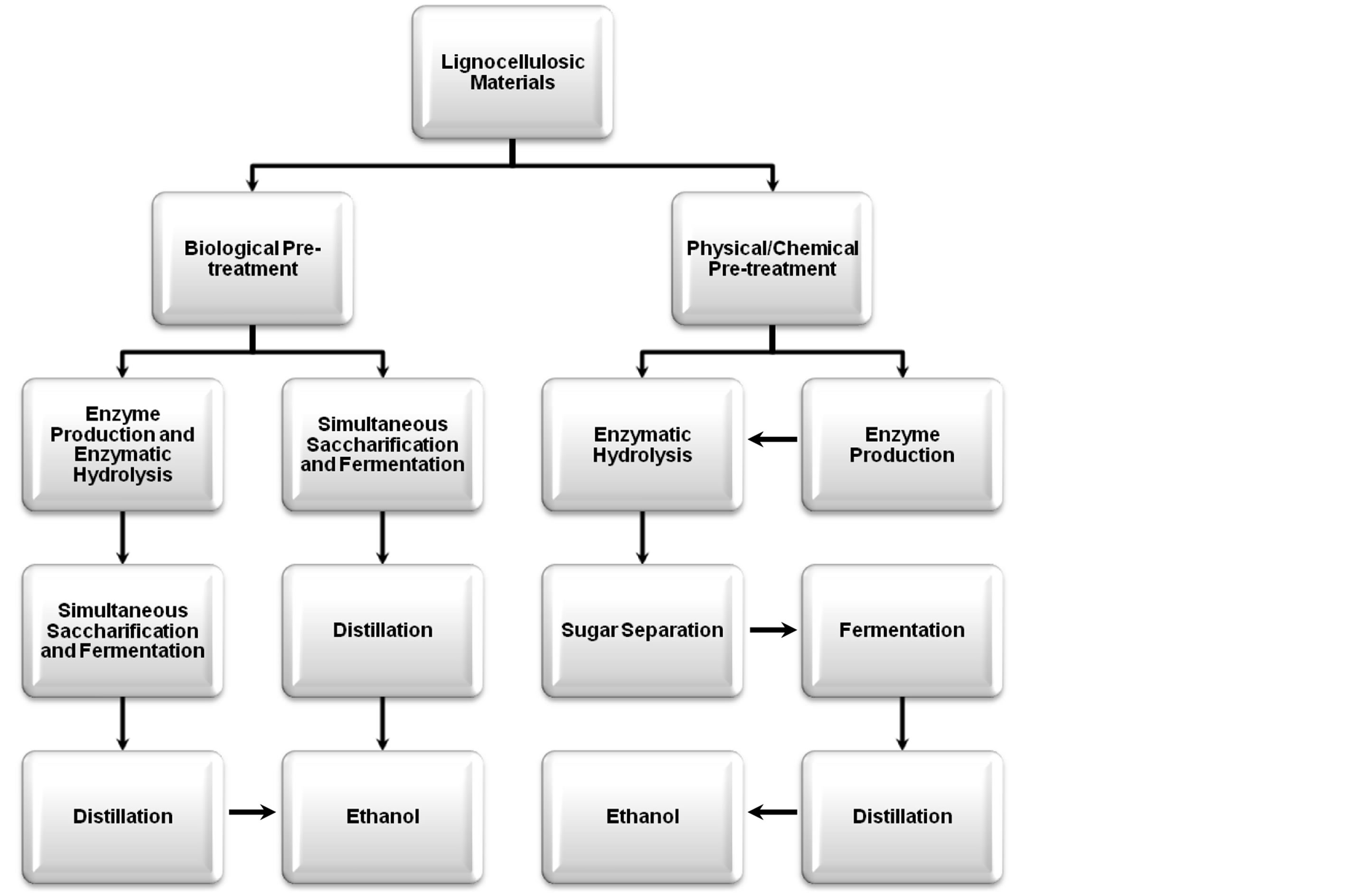
Figure 2. Generalized schematic representation of lignocellulosic materials bio-conversion into ethanol. (Adopted from Iqbal et al. [7]).
energy and raw materials, it is widely used in the bioconversion of renewable lignocellulosic biomass. Glucose, from appropriate hydrolysis of this lignocellulosic biomass under the treatment of advanced biotechnology can be used in different applications such as production of fuel ethanol, single cell protein, feed stock, industrially important chemicals and so on [21-23]. A number of fungi and bacteria capable of utilizing cellulose as a carbon source have been identified. Among the cellulolytic fungi, Trichoderma reesei has the strongest cellulose-degrading activity, and its cellulase has been widely investigated. Many other industrially important enzymes produced by other fungi such as Trametes versicolor, Trichoderma, Aspergillus and Rhizopus species have also been extensively studied by several researchers [24-29].
9. CONCLUDED REMARKS
The whole enzymatic process to hydrolyze lignocellulosic materials could be accomplished through a complex synergistically reaction of these various enzymatic components in an optimum proportion. Cellulases provide a key opportunity for achieving tremendous benefits of biomass utilization. Currently, two significant points of these enzyme-based bioconversion technologies are reaction conditions and the production cost of the related enzyme system. Therefore, there has been much research aimed at obtaining new microorganisms producing cellullase enzymes with higher specific activities and greater efficiency. In addition, using lignocellulosic materials, such as agricultural residues, grasses, forestry wastes, and other low-cost biomass can significantly reduce the cost of raw materials for ethanol production compared to corn. It is also predicted that the use of genetically engineered raw materials with higher carbohydrate content combined with the improvement of conversion technology could reduce the cost of ethanol a lot. All those will give a great help for solving the problems of energy and food in the world. In a word, the cellulase enzymes will be commonly used in many industrial applications, and the demand for more stable, highly active and specific enzymes is also growing rapidly.
ACKNOWLEDGEMENTS
This review is a part of work conducted by Tanzila Shahzadi (MPhil Biochemistry student) under the supervision of Dr M Irshad. Being course coordinator, on providing technical expertise and data review skills Dr Sajid Mahmood is thankfully acknowledged. Authors are also great full to the Department of Biochemistry, University of Gujrat, Pakistan on providing the literature facilities.
NOTES