Xenotransplantation of embryonic pig pancreas for treatment of diabetes mellitus in non-human primates ()
1. INTRODUCTION
There are approximately 1000 pancreas and islet transplants performed per year in the USA, the diabetic population in which exceeds 20 million individuals [1]. Transplantation is rarely used for type 2 diabetes which is almost 20 times more common than type 1 disease. When performed, whole pancreas or islet transplantation requires use of potent immunosuppressive medications that have significant complications [2]. Pig insulin works well in humans. For that reason, the pig could be a physiologically suitable beta cell donor [2]. The severity of humoral rejection effectively precludes the use of pigs, as whole pancreas donors. However, because they are vascularized by the host post-transplantation, islets are not subject to humoral rejection [3]. Unfortunately, porcine islets are rejected by the cellular immune system within days of transplantation in non-immune suppressed non-human primates. Experience with non-transgenic or transgenic pig to primate islet or neonatal islet transplantation in immune suppressed non-human primates shows that sustained insulin independence can be achieved, but only through the use of agents that are not approved for human use or that result in a high level of morbidity and mortality [3,4]. Thus, the need for host immune suppression is a barrier for pig-to-human islet xenotransplantation.
The use of embryonic tissue for transplantation offers theoretical advantages relative to transplantation of either pluripotent embryonic stem (ES) cells, or of fully differentiated (adult) organs [reviewed in 4]: 1) Unlike ES cells, organ primordia differentiate along defined organ-committed lines. There is no requirement to steer differentiation and no risk of teratoma formation; 2) The growth potential of cells within embryonic organs is enhanced relative to those in terminally-differentiated organs; 3) The cellular immune response to transplanted primordia obtained early during embryogenesis is attenuated relative to that directed against adult organs; 4) Early organ primordia are avascular. The ability of cellular primordia to attract a host vasculature renders them less susceptible to humoral rejection than are adult organs with donor blood vessels transplanted across a discordant xenogeneic barrier; and 5) Organ primordia differentiate selectively. In the case of embryonic pancreas, exocrine pancreatic tissue does not differentiate following transplantation, obviating complications that can result from exocrine components such as the enzymatic autodigestion of host tissues.
Approximately a decade ago, we observed that embryonic pig kidney, transplanted across a wide xenogeneic barrier into immune-suppressed rodent (inbred mouse or rat) mesentery undergoes differentiation into an anatomically precise renal organ at the transplant site [5,6]. When we attempted the same approach using embryonic pig pancreas rather than kidney, we discovered using two inbred rat strains as hosts [Lewis rats rendered diabetic using streptozotocin (STZ) [6-9], a model for type 1 diabetes in humans or Zucker Diabetic Fatty (ZDF) rats [10], a model for type 2 diabetes] that unlike kidney, embryonic pancreas does not differentiate in situ into an anatomically precise pancreas. Rather: 1) pancreas differentiates selectively in that only endocrine cells survive; 2) endocrine cells differentiate and migrate into host mesenteric lymph nodes from where they release insulin in a glucoseregulated manner, the host serving as its own in vivo bioreactor; and 3) host immune suppression, required for engraftment of transplanted embryonic pig kidney into mice or rats is not required for engraftment of early-stage embryonic pancreas.
Strategies for xenotransplantation in humans are modeled immunologically using old world monkeys or baboons as hosts. This is because the non-human primates share uniquely with humans a complex and redundant immune system that includes the capacity for humoral rejection of vascularized implants originating from pigs, a frequently used donor-species, outbred diversity and MHC expression patterns known to present barriers to long-term graft survival [3]. Employing protocols developed for embryonic-pig to STZ-diabetic Lewis rat embryonic pancreas transplantation and recapitulated in ZDF rat hosts, we showed that embryonic pig pancreas engrafts, and improves glucose tolerance in STZ-diabetic rhesus macaques without the need for immune suppression [11,12]. Here we review the adaptation of pig-into-diabetic rat transplantation technology to a pig-into-diabetic non-human primate model.
2. TRANSPLANTATION OF EMBRYONIC PANCREAS
2.1. Transplantation of Embryonic Pig Pancreas in Diabetic Rhesus Macaques Is Followed by Engraftment of Pig Endocrine Cells in Mesenteric Lymph Nodes
The developmental stage of donor pig embryos from which primordia are obtained impacts on the host immune response. We showed E35 pig pancreatic primordia are rejected in Lewis rats following transplantation employing conditions under which E28 pig pancreatic primordia engraft [6]. While we have not determined whether E30-E34 pig pancreas has a fate similar to organs obtained on E35, we always use E28 pig pancreas as a source for transplantation because it engrafts predictably. It follows that the importance of obtaining accurately and precisely-timed pregnant Yorkshire sows from which to obtain E28 embryos is critical to the success of experiments. An E28 pig pancreatic primordiumwith separate dorsal pancreas (dp) and ventral pancreas (vp) components is shown as an inset (Figure 1(A)) in Figure 1. It is approximately 1 mm in diameter (much smaller than primordia obtained from older embryos). and its dissection from pig embryos requires a good deal of skill. Our ability to employ E28 embryonic pig pancreas for transplantation is dependent on obtaining pregnant sows that are exactly 28 days pregnant from which to obtain embryos. There is no pancreas in embryos obtained from sows that are 26 days pregnant. Thus there is little or no margin for error.
Following incubation for 45 minutes in vitro of dissected E28 pig pancreatic primordia with selected growth factors and cytokines, we implant individual dorsal and ventral components, within a tunnel dissected between tissue planes of mesentery. Figure 1(B) shows the mesentery of a STZ-diabetic rhesus macaque at the time of transplantation. A primordium between sheets of mesentery is delineated (arrowhead). Accumulation of fat and proliferation of lymphatic vessels are observed in mesentery within 6 weeks post-transplantation in non-human primates [11,12]. Figure 2 shows photomicrographs originating from a mesenteric lymph node of a rhesus macaque transplanted previously with E28 pig pancreatic primodia in mesentery. Sections in Figures 2(A) and (C) are stained with an anti-insulin antibody. Sections in Figures 2(B) and (D) are incubated with control serum. Individual cells that stain positive (red) are present in medullary sinus (arrow A). The cells are polygonal, consistent with a beta cell identity (Figure 2(C) arrow). No positive-staining cells are found in sections incubated with control serum (Figures 2(B) and (D)). No insulinpositive cells are present in mesenteric lymph nodes of
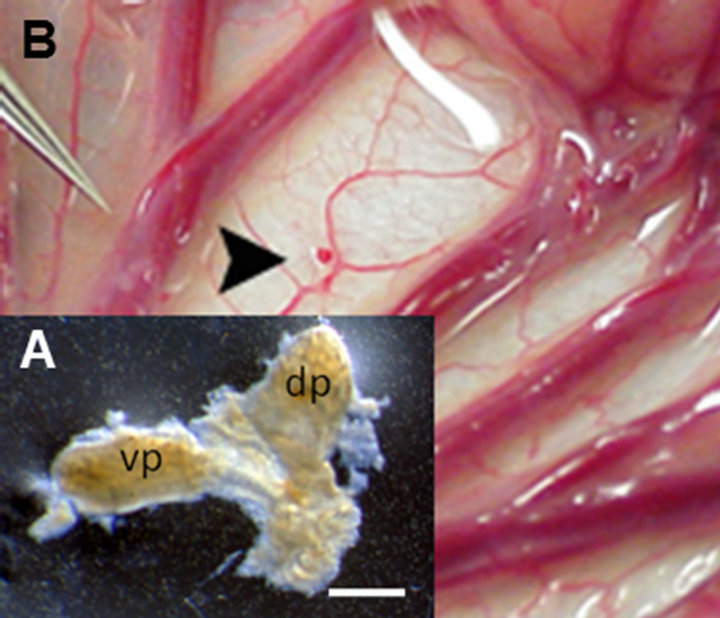
Figure 1. (A) Photograph of a pancreatic primordium freshly dissected from an E28 pig embryo; (B) A pancreatic primordium implanted between sheets of mesentery in a rhesus macaque. dp, dorsal pancreas; vp, ventral pancreas. Scale bar 10 um (A). Reproduced with permission [11].
non-transplanted rhesus macaques [11]. Engraftment of pig tissue in the mesenteric lymph nodes is documented using in-situ hybridization for porcine proinsulin mRNA. Cells expressing porcine mRNA stain with use of an antisense probe to porcine proinsulin mRNA (Figure 2(E)), but not a sense probe (Figure 2(F)). The presence of porcine tissue engrafted in lymph nodes of primates is confirmed by detecting porcine proinsulin mRNA using reverse-transcriptase polymerase chain reaction (RTRCR), porcine X chromosomes using fluorescence insitu hybridization, and cells with insulin granules in lymph node tissue using electron microscopy [11,12].
2.2. Improvement of Glucose Intolerance Following Transplantation of E28 Pig Pancreatic Primordia and Demonstration of Porcine Insulin in Circulation
Figure 3 shows the results of intravenous glucose tolerance testing in rhesus macaques prior to administration of STZ (Pre-STZ) and in the same animals following induction of STZ-diabetes (Post-STZ) and after transplantation of E28 pig pancreatic primordia (Post TX).