Short term effects of fire intensity and fire regime on vegetation dynamic in a tropical humid savanna (Lamto, central Côte d’Ivoire) ()
1. INTRODUCTION
Fire is a natural process, integrated into the function of many ecosystems, particularly savannas. The study of fire ecology focuses on understanding the processes that link fire behavior and its ecological effects. This includes the relationship between fire, living organisms, and the physical environment [1]. Fires strongly influence ecosystem dynamics and are subject to the effects of climate variability on two time scales [2]: 1) on the long-term (1 year): primary production of grasses constitutes the bulk of the fuel; 2) on the short-term (~10 days): local weather conditions, a short period before fires have a considerable influence on their dynamics. We therefore expect a possible impact of climate change on the dynamic of fires, while their effects on ecosystems are not yet fully characterized.
In the Guinea savanna region of West Africa, the phenomenon of savanna invasion by trees despite the annual occurrence of fire is a major concern, as in other American [3] Australian [4] and African [5] savannas. Indeed, the reasons for such changes in vegetation are not well known. The effect of climate change and changes in fire intensity (severity) are suggested [3,6].
The lack of information on fire intensity makes it even more difficult to understand its impact on vegetation, specially in a climate change context. Fire intensity or the rate of energy release, is an important descriptor used in studies of fire behavior and the ecological impacts of fires [7]. Fires are considered more intense in forests than in savannas [8-11], but there is no estimation available to make the difference between fire intensity in dry vs. wet savanna. In humid tropical savanna, the high rainfall ensures the development of a biomass that allows relatively violent fires [12]. Woody cover is controlled by fire in those savannas [13-16], but changes induced by fire in the vegetation structure is not sufficiently studied.
In Africa, every year, on average, respectively 10%, 38% and 70% of the Sahel, Sudanese and humid Guinea zone, of savanna burn [17]. In West African savannas, fire is well integrated with human habits such as hunting, traffic and livestock [8]. Large savanna areas are burnt each year, but very few studies are devoted to fire ecology unlike in Australia where several long-term experiences were established to measure and understand the impact of fire on living organisms [18-21].
The ability of plants to persist after fire by sprouting is known to be decisive for tree demography [22]. However, fire can affect adult trees [23-25]. N’Dri et al. [26] showed that mortality of tree stems increased with fire intensity. The same authors showed that fire interacts with termites to cause external openings on adult tree trunks. This study suggested that fire could damage trees, which would favor their attack by termites to cause internal cavities. Fire could cause thereafter the opening of internal cavity and the breakage of these trees. Our aim here is to test the hypothesis that fire plays an important role in causing possibly life-threatening damage on tree trunks. Despite the importance of fire in determining the occurrence of savannas, it remains difficult to understand how its seasonal nature and intensity interact to influence the vegetation structure, while the severity of a fire depends on its intensity and on the season in which it occurs [1]. Fire behavior is influenced by many factors [27], among which one of the most important is fuel quantity and quality [28,29]. In this study carried out in the Lamto savanna, fire intensity was determined by reference to the quantity of fuel and the season. The impact of fire intensity and its regime on adult trees and on the speed of regrowth of the grass layer were evaluated.
2. MATERIAL AND METHODS
2.1. Study Area
This study was conducted in the Lamto reserve, located in the center of Côte d’Ivoire (6˚13'N, 5˚02'W), in the transition zone between semi deciduous forest and the Guinea savanna [30]. The average yearly rainfall reaches ~1200 mm in the region and is used to classify the Lamto climate in the rainy climates regime [31]. The climate is of a sub-equatorial type with four seasons: a long rainy season from March to July, a short dry season in August, a short rainy season from September to November, and a long dry season from December to February. Each year, the fire (mid-season fire) is set by scientists in the Lamto reserve during the long dry season (in January). In this reserve, the fire has a stabilizing function and explains the persistence of the savanna in an area where the climate favors the existence of forests [8, 30]. This mid-season fire is the usual fire management regime as in most countries [32-36]. The reasons given are that these fire reduce the quantity of biomass and therefore the risk of intense fire and create habitat heterogeneity. The late fire regime is generally not recommended because it has a negative impact on trees [25].
The canopy stratum at Lamto is composed for over 90% adult trees belonging to four species [37]: Bridelia ferruginea Benth (Phyllantaceae) Crossopteryx febrifuga (Afzel. ex G. Don) Benth. (Rubiaceae), Cussonia arborea Hochst. ex A. Rich. (Araliaceae) and Piliostigma thonningii (Schum.) Milne-Redhead (Caesalpiniaceae). Some species such as Annona senegalensis Pers. (Annonacea) are sometimes present as resprout or small shrubs. Others, such as Terminalia schimperiana Hochst. (Combretaceae), are mainly in woody savannas.
2.2. Study Plots
This study was conducted in 2009 and 2010 on two plots of shrubby savanna, of 3.72 ha each (310 m × 120 m). Each plot was divided into seven subplots (80 m × 30 m) separated by 10 m-wide fire-breaks (Figure 1). Three subplots were subjected to normal fire (that occurs in January) and three others to a late fire (in April). For each group of subplots, they were three levels of grass biomass: C0, C1 and C2 (Figure 2). On C0, grasses were removed and distributed on C2 (where grass biomass was then considered doubled). On C1, grass biomass is as the surround savanna. Only C1 and C2 were burnt. The seventh subplot represented the control, where there was no slashing and no burning (UBS, Unburned Site).
2.3. Fire Intensity Measurements
According to [38], fire intensity is a measure of the energy released along the fire front, and is defined as the product of the heat yield of the fuel (H), the weight of standing fuel consumed in the flaming zone (W), and the rate of forward spread of the fire line or perimeter (R). This fire intensity was determined on subplots C1 and C2 that have burned, and during the two different fire regimes.

H: is a constant measured in laboratory. Its recommended value is 18,700 KJ·kg–1 [39]; W (kg·m–2) is the quantity of fuel. It was determined by weighting grass samples (fresh grasses are cut in 1 m2 quadrat at four points distributed over each subplot C1 and C2) after oven drying at 60˚C for 72 hours; for R (m·s–1) measurement, stakes were set at 10 m intervals on the different subplots. Fire spread was measured thereby for each of the eight intervals obtained as the time needed for the fire front to cover the distance between two successive stakes. This time determined using a stopwatch and the
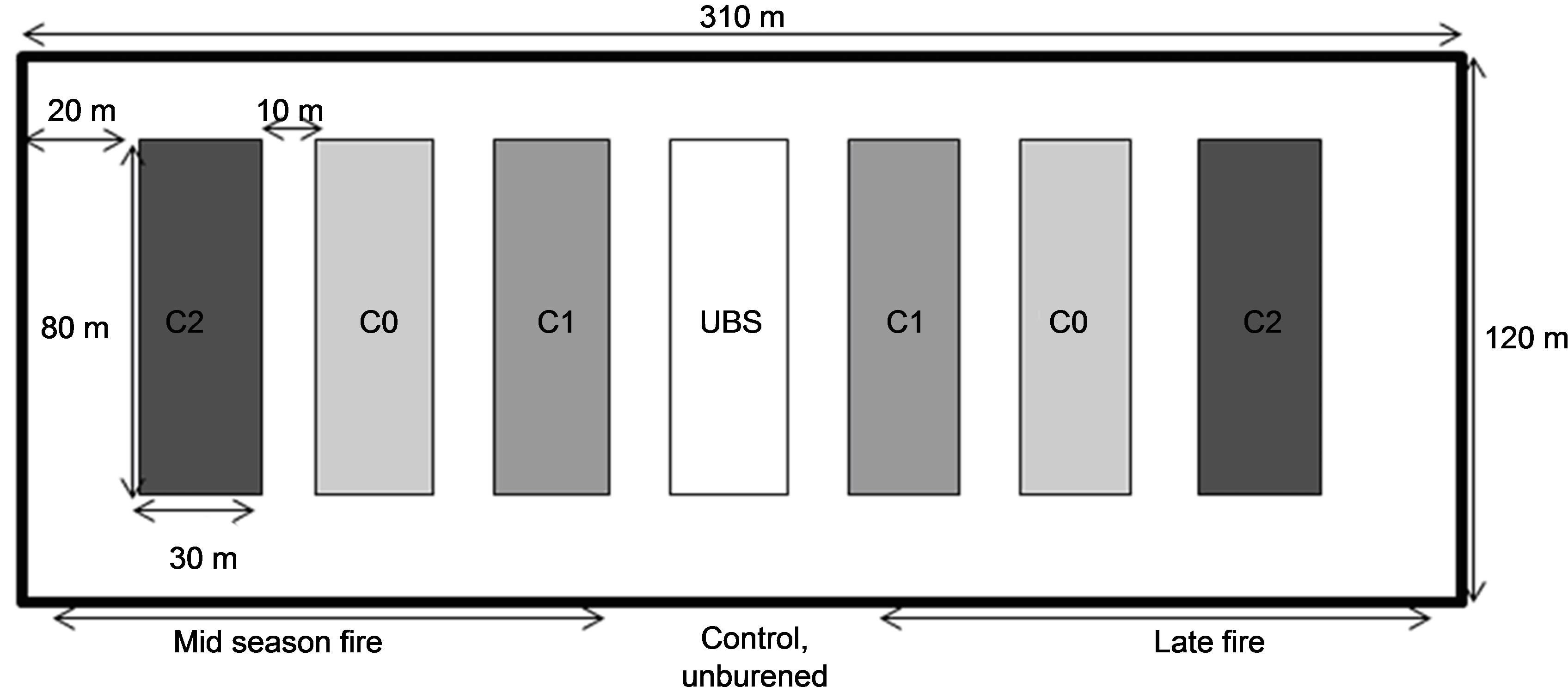
Figure 1. Experimental design of fire experiment. On C0, grasses were removed and distributed on C2 (where grass biomass was considered doubled). Only C1 (normal biomass) and C2 were burnt. The seventh subplot represented the control, where there were no slashing and no burning (UBS).

Figure 2. Subplots with different grass biomass level.
average time was calculated for each subplot during the two different fire regimes experiments.
2.4. Impact of Fire on Grasses Regrowth
This experiment aims at determining the speed of grasses regrowth according to the fire intensity and its regime. The height of the grass layer was measured using a herbometer (Figure 3), this 3, 6 and 9 months after the different fire regime. This regrowth was measured at four points of each subplot C1, C2 and C0 (as control).
2.5. Damage Caused by Fire on Trees
The purpose of this study is to evaluate debarking caused by fire in order to correlate it to the fire intensity and fire regime. After the fire, all the damages on trees were recorded, whether related to an immediate fire (Figure 4(a)) or previous ones (Figure 4(b)). The main savanna species were considered: B. ferruginea, C. febrifuga, C. barteri and P. thonningii. The impact of fire on young and adult trees was evaluated by detecting areas debarked by fire on the main branches. Fire can cause damage in all directions for straight trees [40] or to the leeward side of the branch or trunk [26,41]. When the trunk or branch was debarked or presented an external opening, we recorded whether trees were leaning and whether debarked parts or external openings were facing downwards or upwards. Trees were considered leaning if more than 30˚ from the vertical. According to [41-44], these debarked parts could be entry points for termites into trees and could be easily burnt at the next fire because not protected by the bark. According to the scenario proposed by [26], the external openings observed on trees would start by debarking which would allow termites to enter into tree trunks. Once termites have

Figure 3. Herbometer used to measure the height of grass stratum.
entered the trunk and started piping, they restrict their action to heartwood. Once the tree is hollowed by termites, unless the internal cavity is very large, a single fire could cause an external opening (Figure 4(c)). These external openings are responsible for an increase in tree mortality through mechanical trunk breakage [45, N’Dri “unpublished data”).
2.6. Statistical Analyses
Statistical analyzes were processed using the R software (http://www.r-project.org/). The linear model (lm) was used to analyse 1) the change in the fuel quantity according to the treatment (C1 and C2) and the fire regime (mid-season and late fire); 2) the intensity of fire according to its regime; 3) the height of grass regrowth according to the treatment (C0, C1 and C2), the fire regime and the measurement period (3, 6 and 9 months after the fire). The test ranks sign of Wilcoxon was used to analyse the intensity variation as a function of treatment (C1 and C2). The generalized linear model (glm) was used to analyse the frequency of debarked trees according to the different species and the development stages. Chi-square tests were used 1) to test the distribution of debarking parts and opening directions; and 2) to test its correlation with the leaning direction.
3. RESULTS
3.1. Fire Intensity
The C2 treatment led to a much higher fuel for this treatment (ANOVA, F1,6 = 14.99 and p = 0.001, Table 1). These effects in turn affected fire intensity, which increased with the fuel quantity (Test rank sign of Wilcoxon, p < 0.05, α = 5%) and is independent on the fire regime (ANOVA, F1,4 = 1.770, p = 0.254, Table 2). There is no fuel variation between the burnt plots at different fire date (ANOVA, F1,6 = 0.015 and p = 0.906).

Figure 4. Different stages of damages caused by fire. (a) Scorching of the year; (b) scorching following many fires facing downward at the leaning side; (c) Exposition of internal cavities at the downward face of branch leaning side.
3.2. Impact of the Fire on the Regrowth of Grasses Layer
The regrowth speed of grasses varied with the level of fuel used (treatment): C0, C1 and C2 (ANOVA, F2,126 = 8.04 and p = 0.0005), and the measurement period: 3, 6 and 9 months after each fire (ANOVA, F2,126 = 411.59 and p < 0.001). Regrowth varied according to the inter-

Table 1. Grass biomass (fuel) quantity according to fire regime treatement.

Table 2. Mean intensities of fire according to fire regime and fuel quantity.
action between fuel level and the measurement period (ANOVA, F4,126 = 2.78 and p = 0.03). It was only after 9 months that the difference in regrowth was significant among treatments (ANOVA, F2,42 = 7.49 and p = 0.0016); on average of 70.25 cm height, 67.31 cm and 63.56 cm respectively for treatments C2, C1 and C0. The heights of grasses 3 months after the fire in mid-season are higher than those observed for the same duration after the late fire (ANOVA, F1,42 = 9.36 and p = 0.004). This same tendency is observed 9 months after each type of fire (ANOVA, F1,42 = 29.39 and p < 0.001).
3.3. Damages Caused by Fire on the Woody Stratum
The recent debarking observed following the mid-season fire and caused by it showed that a single fire rarely caused debarking on trees: at most, in double fuel load (C2), 8% of trees were debarked while the fire under normal fuel (C1) did not cause any debarking.
Older debarking, resulting from several previous fires affected 48% of the sampled trees. The proportion of these trees varied with species, C. febrifuga (82%) was significantly more often debarked (deviance = 63.846, df = 3, χ2 = 100.657, p = 0.001) than P. thoningii, C. barteri and B. ferruginea, respectively, 45%, 35% and 32% (Figure 5). Moreover, adult trees were more often significantly debarked (65%) than young trees (35%) (deviance = 31.448, df = 1, χ2 = 101.015, p = 0.004).
After observation of 580 main branches, 57% were debarked. The directions in which these branches were debarked were not randomly oriented. Most of them

Figure 5. Proportion of scorched trees according to the different species. Test post-hoc of TukeyHSD. BRI: Bridelia ferruginea; CRO: Crossopteryx febrifuga; CUS: Cussonia arborea; PIL: Piliostigma thonningii.
(89%) had their debarking facing downward at the leaning side (χ2 = 190.56, df = 1, p < 0.001). In addition, 66% of the leaned branches had external hollows. The majority of these branches (79%) had their hollow facing downward at the leaning side of trees, this observation is statistically significant (χ2 = 92.60, df = 1, p < 0.001).
4. DISCUSSION
4.1. Relation between Fire Intensity and Severity
The average fire intensity did not vary according to the fire regime (mid-season and late fires). Indeed, [46-48] as well as studies conducted in Kakadu National Park [35], showed that timing is likely to be an important factor determining the ecological effects of fire, regardless of intensity. The impact of fire on tree survival is more related to physiological stage of the tree at the fire moment than the intensity of this fire. For example, it may be that savanna fires have the greatest impact on plant during periods of active growth (late fire) because carbohydrate and nutrient reserve have been depleted [7, 46,49,50]. Whereas the period of mid-season fires (midJanuary) coincides with the senescence phase of the epigeous parts of plants and the vegetative activity is lower.
As expected, the fire intensitiy values are higher for double fuel load than simple fuel load. Indeed, abundant biomass on the sites that remain unburned for years increases the risk of fire of very high intensities [51,52].
4.2. Impact of the Intensity and Regime of Fire on Vegetation
According to [2], fire intensity can cause tree death or damage to them through the debarking of trunks or the burning of leaves or buds. Here we show that in the long term, fire causes the debarking of 48% of trees. These debarked areas would favor termites entry into trees [26, 41-44] to cause internal hollow, exposed later by fire action [26]. Once exposed, tree trunk mechanical resistance is affected [45].
The tree species C. febrifuga was most subjected to hollowing: 84% of adult trees were hollowed [26]. It is also the one with the most debarked individuals: 82%. As the debarked branch parts, the external cavity facing downward at the leaning side of the trees. This shows the link between debarking and hollowing.
The fire intensity affects the rate of regrowth of grasses. Nine months after the mid-season and late fires, grass heights on compartments C2 were higher than that on C1, which in turn was higher than that on C0. This suggests a stimulation of growth where the fuel was doubled, that through the ash resulting from burning grasses.
The late fire decreased the regrowth rate of grasses compared to mid-season fire in spite of a supposedly better water supply due to the onset of the rainy season. This trend is contrary to that observed by [8], which showed that grasses regrowth was higher after the late fire than the mid-season one (1 - 250 days following the fire). He explained this higher regrowth following the late fire in April, by the fact that the rainy season which extends from March to July, promotes growth of grasses after this fire regime, while the mid-season fire (January) took place during the long dry season which runs from December to February. The contrary tendency observed in this study, could be explained by the fact that the rainy season tends to shift more and more due to climate change. Indeed, rainfall is very variable from one year of study to another. In 1963, when Monnier [8] measured the speed of grass regrowth, rainfall after mid-season fire was lower than after the late one. About 40, 150 and 100 mm of rain respectively in February, March and April and approximately 400 and 300 respectively in June and July following the late fire. The highest rainfall following the late fire has indeed favored greater regrowth of grasses. However, during the year 2009, rainfall following the mid-season fire was higher than after the late fire. Rainfalls were 177, 158 and 128 mm in February, March and April, respectively following the mid-season fire and 122 and 169 in June and July respectively, following late fire. The opposite trend about rainfall in this study compared to that of Monnier [8] may explain the faster grass regrowth following the mid-season. These results suggest that abundant rainfall in a year, would favor higher fire intensity the following year. Now, if the climate is not conducive to good combustion of vegetation for a year, it would through the accumulated biomass, lead to a high intensity of fire the following year. This high fire intensity will in turn promote regrowth of more biomass the following year. The climate of the year and meteorological conditions before the fire control in fact, the occurrence and speed of fire spread in almost all ecosystems [2]. The climatic conditions of the year, in fact determine the amount of fuel that burns.
5. CONCLUSION
This study reveals that fire intensity does not vary significantly from one regime to another (mid-season and late fires) but depends on the level of fuel. The rate of regrowth of grasses following fire also varies depending on the level of fuel: grasses on sites with double fuel load grow faster than those on the sites with simple fuel load. The mid-season fire regrowth ensures greater biomass than the late fire. The impact of fire on mature trees is also linked to its intensity; they are more commonly damaged in double-fuelled sites compared to simple fuelled-sites. There is a positive feedback between fuel load and grass production: grass regrowth tend to be faster following more intense fires; if this yields a higher grass biomass at the end of the growing season, then locally the fire will be more intense. If a low intensity fire breaks this feedback loop, dead grass biomass will accumulate and yield a more intense fire the following year. We can thus conclude that the grasses of Guinea savannas maintain a resilient high-intensity fire regime. The fire remains the recommended mid-season fire regime, because supports a significant regrowth of herbaceous biomass fires that promotes more and more important, ensuring the maintenance of balance between grasses and trees.
6. ACKNOWLEDGEMENTS
This study was conducted as part of the RIPIECSA-project in Côte d’Ivoire. Thanks to all the Lamto reserve students who helped us during handling for determining the intensity of the fire. We are grateful to Kanvaly Dosso, N'Golo Kone, Kone Mouamadhou, Tamia Kouakou, Evelyne Toure, Josiane Ahoussi, Tra-Bi. Thank you to all the reserve technicians who have also helped us during this work. Among them, we especially thank Francois N’Guessan and Kounan Honoré who accompanied us during all fieldwork. We are grateful to Karen Tinland for her contribution in editing the text.