Occurrence of fluoride in the drinking waters of Langtang area, north central Nigeria ()
1. INTRODUCTION
Fluorine often occurs in combine form in minerals as fluoride. It is highly reactive and represent about 0.06% to 0.09% of the earth crust [WHO, 1994]. Fluorine presence in groundwater is mainly a natural phenomenon, influenced mainly by local and regional geology. The main source of fluorine in groundwater is basically from mafic minerals which are concentrated in rocks. Robinson and Edington in 1964 reported the main sources of fluoride in ordinary soil consist of clay minerals. Fluoride rich minerals which are present in rocks and soils, when in contact with water of high alkalinity are released into groundwater by hydrolysis.
The effect of fluoride could either be beneficial or harmful. Hussain et al. in 2004 summarise both the beneficial and harmful effects of fluoride to be: prevention of dental caries and anti-carcinogenic agent; harmful effects to be dental fluorosis, cardiovascular disorder, gastro-intestinal disorder, endocrine effects, neurological, reproductive effects, developmental inhibition, genetic damges and effects on the pineal gland. The most common symptoms of fluoride effects is dental fluorosis, a condition involving interaction of fluoride with tooth enamel, which involves staining or blackening, weakening and possible eventual loss of teeth. With high exposure to fluoride, skeletal fluorosis can result. This manifest in the earliest stages of osteoclerosis which involves hardening and calcifying of bones and causing pain, stiffness and irregular bone growth. At its worst, the condition can result in severe bone deformation.
Dental flourosis is a common manifestation in the inhabitants of Langtang area among adults, youth and children. This has inspired the authors to determine the occurrence of fluoride in the drinikng water sources of Langtang area.
2. THE STUDY AREA
The study area is defined by Latitudes 9˚00'' and 9˚11''N and Longitudes 9˚45'' and 9˚54''E. It is part of the Crystalline Hydrogeological Province of Nigeria (Figure 1). Over 60% of the area is entirely underlain by coarse
to porphyritic granite intruded by syenites on the western part, fine grained biotite granites and small scale intrusions of aplitic-, pegmatiticand microgranites with xenoliths of the coarse to porphyritic granite a common sight (Figure 2). Small volcanic rocks are also common. The southern portion of the map is underlain by migmatites with intrusions of non orogenic granite, which probably belong to the Younger Granite series of Nigeria. Pegmatitic dykes have also intruded the migmatites. A large scale fault was formed as a result of the syenite intrusion trending in an almost N-S direction. The study area is drained by the River Bapkwai which takes its course from the hills on the western side and flow towards the south east direction. Several other small streams drain the area which flow in the same direction.
Hydrogeologically, groundwater is found in three different aquifers; recently deposited alluvium found along river and stream channels, Weathered Overburden (Soft Overburden Aquifer) and Fractured Crystalline Aquifer. Groundwater is encountered in the alluvium at depths of between 4 - 5 meters, 10 - 15 meters in the Weathered Overburden Aquifer and between 30 - 35 meters in the Fractured Crystalline Aquifer. In the dry season, most of the hand dug wells dry up and water is obtained from scooped water holes in streams and river sands.
3. METHODOLOGY
3.1. Field Sampling
Hand dug well samples were collected with a clean plastic bucket as recommended by Davies, 1994 [1]. Samples from boreholes were pumped for several minutes with the view that stagnating water within pipe is pumped out in order to sample those coming from the formation. Spring samples were collected close to emission points to minimize contact with the atmosphere. Field parameters (temperature, pH) were measured right in the field with an Oakton pH/MV meter. Water samples were filtered with UNICEF standard filter to free them from suspended particles. Two samples were collected at each point. One was acidified with two drops of concentrated nitric acid to a pH of 2.0. Samples were kept in a field cooler throughout the period of the sampling exercise.
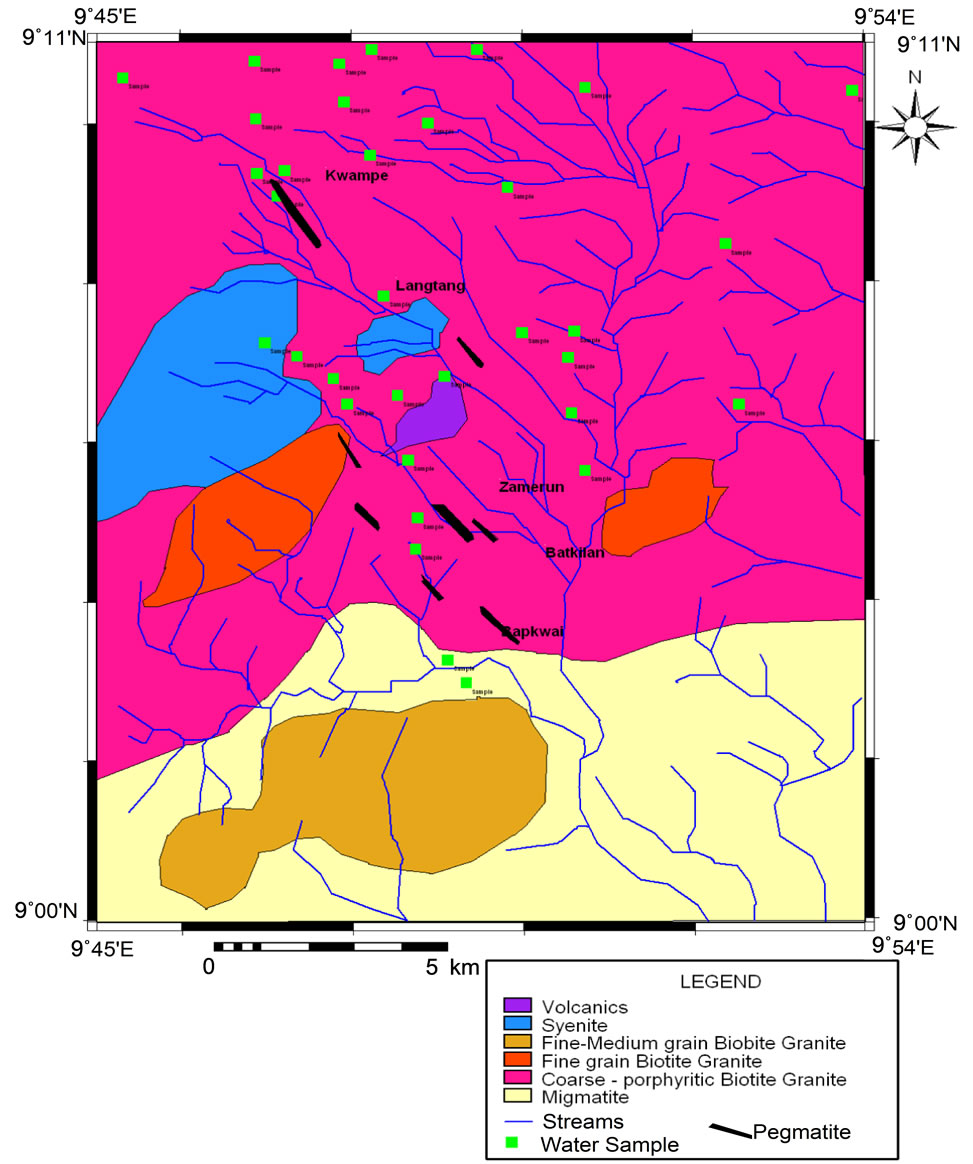
Figure 2. Geological map of Langtang area showing water sampled point [2].
3.2. Laboratory Analysis
Major cations and some trace elements were analysed using the Inductively Coupled Plasma Optimal Emission Spectrometry (ICPOES model 3000 ICP) at the Department of Geology and Mining, University of Jos. The composition of the anions (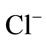 ,
,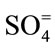 ,
, ,
,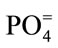 ) were determined using the Ion Chromatography and Fluoride by Ion Selective Electrode (ISE) at the ACME Laboratory Vancouver, Canada. Bicarbonate alkalinity was determined by titration. Total Dissolved Solids and Total Hardness were modeled with the AqQA software.
) were determined using the Ion Chromatography and Fluoride by Ion Selective Electrode (ISE) at the ACME Laboratory Vancouver, Canada. Bicarbonate alkalinity was determined by titration. Total Dissolved Solids and Total Hardness were modeled with the AqQA software.
3.3. Statistical Analysis
The data were statistically treated with the SPSS version 10 for windows to evaluate relationship among variables and behaviours of the elements in the hydrogeological environment. The descriptive statistical method was employed to calculate mean, minimum and maximum values of the data, while bivariate pearson moment correlation was employed to look at the relationship among the parameters.
4. RESULTS AND DISCUSSION
4.1. Physico-Chemical Composition of the Waters in Langtang Area
Table 1 shows the physical and chemical composition of the waters of Langtang area. Table 2 shows the mean, minimum and maximum values of the parameters. Temperature of the waters ranges from 26˚C - 27.90˚C with a mean of 27.90˚C as against the 26.5˚C obtained in Crystalline Hydrogeological Province of Nigeria as given by Schoeneich and Garba, 2010 [3]. Hydrogen exponent