The Most Primitive Extant Ancestor of Organisms and Discovery of Definitive Evolutionary Equations Based on Complete Genome Structures ()
1. Introduction
Primitive organisms might appear after long periods of chemical evolution, during which various organic compounds were accumulated. Recently, it was reported that vesicles consisting of lipids and polynucleotides spontaneously replicated under experimental conditions [1]. These pseudo-organisms were the first living things to appear on Earth; however, we cannot trace their origins because the original assembly of chemical compounds was not stable. Only those primitive organisms that established a method of self-replication could survive and continue to evolve. Therefore, it is difficult to identify the true primitive ancestors of life on Earth, as their very make-up would probably be unstable in the current environmental conditions. Although fossilized traces of very early organisms have been found in sedimentary rocks dating from 3.1 - 3.7 billion years ago [2 - 5], they do not appear to be the origin of life. It is likely that only primitive organisms that had cell walls could leave a fossil record, which may rule out finding any traces of simpler organisms.
Several key eukaryotic organelles originated from symbioses between separate single-celled organisms [6 , 7]. For example, mitochondria developed from the proteobacterium Rickettsia or its relatives [8 , 9]. The Reclinomonas americana (Protist) mitochondrion (~70 kb), consisting of 97 genes, is thought to be an ancestral mitochondrial DNA (mtDNA), while vertebrate mtDNA (~16 kb), consisting of 37 genes, seems to be constructed with only essential genes for respiration reactions. Based on 13 respiratory genes, the amino acid composition and gene patterns within complete mitochondrial genomes are almost identical among all animal species, except Amoebazoa [10]. Irreversible evolutionary divergence accompanied by increasing G and C content means that the G and C content of descendants should be identical to or higher than that of the ancestor. However, it was shown that the G and C values for R. americana (G: 0.148, C: 0.114) mtDNA were higher than those of Monosiga brevicollis (G: 0.081, C: 0.059), which were the lowest among the samples examined [11]. In addition, nucleotide regression lines for the two species differed from each other [11 , 12]. Thus, we concluded that mitochondria might derive directly from primitive organisms [13]. Our present results based on genome evolutionary rules deny that the Reclinomonas americana mitochondrion is thought to be an ancestral mitochondrial DNA [8 , 9]. However, there is no scientific evidence that mitochondria are the most primitive extant ancestor of all life. Based on Charles Darwin’s theory of natural selection, all organisms have a single origin and common ancestor, therefore, we predict that the amino acid compositions of their complete genomes should naturally reflect their evolution from bacteria to Homo sapiens [14 , 15].
The two nucleotide relationships were expressed by linear regression lines, which crossed at a single point [11 , 12]. Thus, we concluded that the origin of life was single and pluripotent. Assuming this conclusion, at the crossing point representing the origin of life, (G ≈ C) and (T ≈ A) to satisfy Chargaff’s parity rules [16 , 17]: (G - C) ≈ 0 and (T - A) ≈ 0. The two regression lines representing high and low C/G animal mitochondria must satisfy (G ≈ C) and (T ≈ A) at the crossing point for the line based on Chargaff’s parity rule. Therefore, differences in (G - C) and (T - A) reflect mitochondrial evolution. In addition, to satisfy Chargaff’s second parity rule [17], the nucleotide content of the first half of the DNA strand should be equal to that of the second half [18], providing a symmetry between the 5’ and 3’ ends. As animal mitochondrial genomes deviate from Chargaff’s second parity rule [17], the divergence in animal mitochondria increases the unsymmetrical nucleotide content. To detect intragenomic alterations based on increases in the unsymmetrical nucleotide content, a method that investigates nucleotide content differences, i.e. (G - C), (G - A), (G - T), (C - A), (C - T) and (A - T) in the sequentially divided genomes, was developed in the present study. These exciting results could not have been obtained from sequence analyses.
2. MATERIALS AND METHODS
All genome sequences were obtained from the National Center for Biotechnology Information GenBank database (http://www.genome.jp/ja). The nucleotide contents of all genomes, which are listed in Extended Data Figs, were normalized (G + C + A + T = 1) because normalized values are independent of species and genome sizes [19 , 20]. A whole genome was divided into several fragments to evaluate intra-genome alterations due to evolution, and the differences between the two nucleotide contents of six pairs ((G - C), (G - T), (G - A), (C - T), (C - A), (T - A)) were calculated for all fragments. In addition, mitochondria were divided into three equal-size fragments. To easily understand evolution at a glance, these six nucleotide differences were graphically expressed with a single pattern. This appears to be a novel approach. Calculations were carried out with a personal computer, TOSHIBA dynabook T552 (Tokyo, Japan), Windows 10 installed.
3. RESULTS AND DISCUSSION
3.1. Primitive Ancestor
Chargaff’s parity rules, where (G = C) and (T = A), were the original rules for inter- [16] and intra- [17] molecular relationships of DNA, and are applicable to chromosomal DNA [21 - 23]. The normalized nucleotide contents (C, T and A), predicted from complete genomes, are expressed by the content of the fourth nucleotide (G) using linear regression lines in chromosomal [21], non-animal mitochondrial and chloroplast DNA [22 , 23]. Classifying animal mitochondria into high and low C/G groups, the content of the first three nucleotides could be similarly expressed by the forth nucleotide content [23], although animal mitochondria deviated from Chargaff’s second parity rule [24 , 25] (Figure 1(a)). Vertebrate mitochondria overlapped with the high C/G invertebrates, which have high C content (Figure 1(a)), indicating that both groups were descended from the same origin [12 , 13], and that more highly evolved organisms seem to have a greater cytosine content [11]. Non-animal mitochondria and chloroplasts obeyed Chargaff’s rules [23]. C contents in cellular organelles and chromosomal DNA vs G contents were expressed with linear regression lines, which crossed at a single point (Figure 1(a)). This is consistent with our previous findings [11 , 12]. In addition, we found that nucleotide content relationships in viral DNA (Extended Data Table 1) were heteroskedastic (Figure 1(b)).
Based on the normalization of the four nucleotides (G + C + T + A = 1), the GC content, (G + C), is expressed by {1 - (A + T)}, where GC content (G + C) and AT content (A + T) are completely linear, not only in chromosomal, non-animal mitochondrial, and chloroplast DNA, but also in animal mitochondrial DNA (Figure 1(c)). The same result was obtained for viral DNA (Figure 1(d)). Assuming irreversible divergence, it is generally thought that GC content increases along with biological evolution. Thus, the organelles that have the lowest GC content might be the most primitive. In the current study, the GC content of the mtDNA of the choanozoan M. brevicollis was the lowest among the samples examined. The GC content of the bacterium Streptomyces coelicolor was the highest of all the samples, although this organism was not the most evolved. Amongst the viruses examined, the GC content of Melanoplus sanguinipes Entomopoxvirus was the lowest, while that of Papio ursinus Cytomegalovirus was the highest (Figure 1(d)).
A previous study [11] stated that the C content of complete mitochondrial genomes reflects biological evolution better than the GC content. Based on the normalization of the four nucleotides, G + C + A + T = 1, C = 1 - (G + A + T). Thus, C content and (G + A + T) are linear. In the current study, the lowest C content was observed in M. brevicollis mtDNA, while the highest C content was found in mtDNA from avian species Gallus gallus and Taeniopygia guttata, and primates H. sapiens, Pan paniscus, Pan troglodytes, and Gorilla gorilla (Figure 1(e)). Amongst the viral genomes, the C content of Mollivirus sibericum was the lowest, while that of De-Brazza’s monkey virus was the highest (Figure 1(f)).
3.2. Genome Evolution
The complete genome is represented by four nucleotide contents based on more than a certain amount of randomly chosen fragments, as well as on completely linear fragments [26 , 27]. Therefore, when the whole genome of bacterium Ureaplasma urealyticum (G; 0.131, C; 0.127) was sequentially divided into nine equal fragments, the amounts of the four nucleotides in each fragment were quite similar (Figure 2(a)). This is consistent with our previous results, which indicated that a whole genome may be constructed from small units with similar amino acid compositions [26 , 27]. Nucleotide content differences (i.e. (G - C) and (A - T) were reversed from positive to negative values in fragments 5 and 6 of the U. urealyticum genome (Figure 2(b) and Figure 2(c)). The ratios of (G - C)/(G + C) and
![]()
Figure 1. Regression lines. Left and right panels represent cellular organelles and viruses, respectively. Upper panels (a and b): horizontal and vertical axes represent G and C contents, respectively. Upper left panel: C content plotted against G content in vertebrate mitochondria (asterisk), high C/G invertebrate mitochondria (triangle), low C/G invertebrate mitochondria (cross), bacteria (circle), non-animal mitochondria, chloroplasts (diamond), and chromosomes (square). Red square represents Monosiga brevicollis mitochondrion. As the p-value based on the two regression lies representing high and low C/G animal mitochondria is 0.000, these two lines are significantly different each other. There is no-significant difference among chromosomal, non-animal mitochondrial and chloroplast DNA, while these lines differ from those representing animal mitochondrial DNA. Right upper panel (a): C content plotted against G content in viruses. Middle panels (c and d): Horizontal and vertical axes represent (G + C) and (A + T) contents, respectively. Lower panels (e and f): Horizontal and vertical axes represent (C) and (G + A + T) contents, respectively.
![]()
Figure 2. Nucleotide content was sequentially divided into nine fragments. Left side: Ureaplasma urealyticum mitochondrion, right side: Monosiga brevicollis mitochondrion. G: blue, C: red, A: black, and T: green. Vertical axis represents nucleotide content.
(A - T)/(A + T) are called the GC and AT skew, respectively [28]. The skew seems to be based on differences in replication processes between the leading and lagging strands [29]. In particular, replication of the lagging strand increases the probability of mutations as a result of the deamination of cytosine, and the inversion of nucleotide content differences reflects biological divergence. Similar phenomena are observed in mitochondria, which consist of heavy (H) and light (L) chains [30 - 32]. Plotting the GC skew vs. G content was used to classify animal mitochondria into two groups: high and low C/G [11]. In M. brevicollis mitochondria, the nine DNA fragments showed almost the same nucleotide contents (Figure 2(d)), as was observed in U. urealyticum (Figure 2(a)). However, GC and AT content difference inversions were not observed in M. brevicollis mtDNA (G: 0.081, C: 0.059) (Figure 2(e) and Figure 2(f)). Thus, the M. brevicollis mitochondrion might be more primitive than the U. urealyticum chromosome. These results clearly indicate that nucleotide content differences such as (G - C) and (A - T) reflect biological evolution. Therefore, the other nucleotide content differences, (G - A, G - T, C - A and C - T), were examined to determine whether or not these values reflect biological evolution.
3.3. Organelle Evolution
Complete mitochondrial genomes were investigated (Figure 3, left panel). To allow simple visual comparison of inter- and intra-species genome structures, genomes were sequential divided into three
![]()
Figure 3. Nucleotide content differences in complete mitochondrial genomes (left side) and the three sequential fragments of each mitochondrial genome from 1 to 9 (right side). Left to right: (G - C), (G - T), (G - A), (C - T), (C - A) and (T - A).
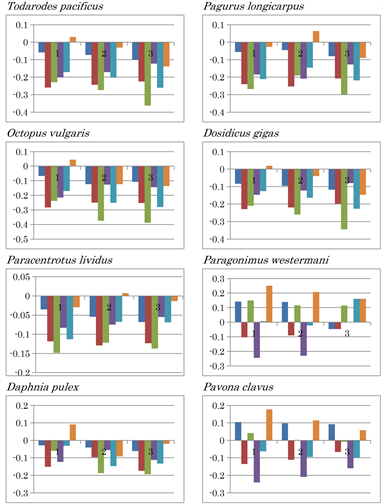
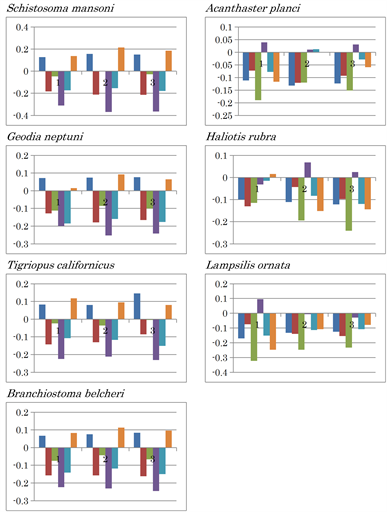
fragments throughout subsequent analyses, from which three separate patterns emerged. Six nucleotide content differences were observed among the mitochondria of the four species (M. brevicollis, P. pallidum, D. discoidium and R. americana) (Figure 3, right panels). The six nucleotide content patterns were conserved within the three fragments among the four species. No inversion of nucleotide content differences was observed in the mtDNA of M. brevicollis (G: 0.081, C: 0.059), the mycetozoan Polysphondylium pallidum (G: 0.143, C: 0.085), or Dictyostelium discoideum (G: 0.171, C: 0.104) (Figure 3), although differences in (G - C) and (T - A) values for M. brevicollis mtDNA were the lowest amongst these species. Based on genome sequencing, choanoflagellates are most closely related to animals [33]. As the nucleotide content difference patterns of the three fragments were almost identical for these three species, their nucleotide distributions were judged to be homogeneous, indicating nucleotide content symmetry. Thus, these mitochondria are likely to be primitive. Consistent results were obtained from Ward’s clustering analysis using amino acid compositions predicted from complete mitochondrial genomes as traits [11]. These findings indicate that the M. brevicollis mitochondrion is the most primitive among the three. In contrast, AT inversion was observed in the third fragment of Reclinomonas americana mtDNA (G: 0.148, C: 0.114), which has previously been proposed as a mitochondrial ancestor [8]. However, differences in (G - C) and (T - A) values in R. americana mtDNA were smaller than those in the mtDNA of the previous three organisms. Nucleotide content inversion causes significant differences in nucleotide content patterns as a result of unsymmetrical nucleotide content. Thus, the R. americana mitochondrion is probably more evolved than the former three mitochondria. In addition, AT inversion occurred in the following more highly evolved organisms: Mollusca species, squid (Todarodes pacificus), octopus (Octopus vulgaris), Echinodermata species, sea urchin (Paracentrotus lividus), water flea (Daphnia pulex), hermit crab (Pagurus longicarpus), and Humboldt squid (Dosidicus gigas) (Extended Data Figure 1). In addition, large positive (G - A) values in the three fragments were observed in Paragonimus westermani, while large positive (G - C) and (A - T) values in the three fragments were observed for the mtDNA of representatives of the following phyla: Cnidaria (Pavona clavus), Platyhelminthes (Schistosoma mansoni), Porifera (Geodia neptuni), Arthropoda (Tigriopus californicus), and Chordata (Branchiostoma belcheri) (Extended Data Figure 1).
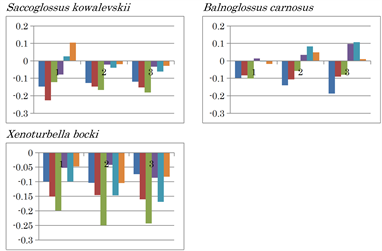
A positive (C - T) value was characteristically observed in the three fragments of Echinodermata species Acanthaster planci, the second and third fragments of Mollusca species Haliotis rubra, and in the first fragment of Mollusca species Lampsilis ornata. CT inversion occurred in H. rubra and L. ornata mtDNA (Extended Data Figure 1). The nucleotide content difference patterns of the mtDNA of hemichordates Saccoglossus kowalevskii and Balanoglossus carnosus differed from each other. Both AT and CT inversions occurred in the first mtDNA fragment of S. kowalevskii, while large positive (C - T) and (C - A) value differences occurred in the second and third fragments. AC and AT inversions were observed in B. carnosus mtDNA (Extended Data Figure 1). Neither nucleotide inversion nor positive nucleotide differences were observed in the mtDNA of deuterostom, Xenoturbella bocki.
In the mtDNA of primate species H. sapiens, P. troglodytes, G. gorilla, Macaca mulatta, Daubentonia madagascariensis, Nycticebus coucang, and Tupaia belangeri, nucleotide content difference patterns were quite similar in the first four species, and large positive increases in (C - T) differences in the three fragments clearly indicated evolutionary divergence (Figure 4). The positive (C - T) differences in all three fragments were characteristic of these four primate mitochondria, while positive increases in (C - T) values were only observed in the third fragment of N. coucang and T. belangeri mtDNA. In contrast, nucleotide content difference patterns of the prosimian Lemur catta completely differed from those of the primates, although TA inversion was observed in the second fragment. The primate mtDNA nucleotide content patterns were also completely different from that of hemichordate B. carnosus, although their C contents were the highest among all organisms examined [11]. This finding indicates that mitochondrial structures respect epigenomic evolutionary functions.
The mitochondria of other vertebrates (Extended Data Text.), rodents (Extended Data Figure 2(a)), ocean-dwelling mammals, cetaceans, aves (Extended Data Figure 2(b)), amphibians (Extended Data Figure 2(c)), reptiles (Extended Data Figure 2(d)), and fishes (Extended Data Figure 2(e) and Extended Data Figure 2(f)) were also examined. In these organelles, differences in (G - C) and (A - T) or in other nucleotide inversions, as well as GC and AT inversions, increased along with evolution. Consistent results were also obtained from non-animal mitochondria and chloroplasts (Extended Data Figure 3), as well as prokaryotes (Extended Data Figure 4). In the microsporidian protozoan Encephalitozoon cuniculi, GC and AT inversions, as well as other nucleotide content inversions, were observed (Extended Data Figure 5).
3.4. Virus Evolution
The M. sanguinipes Entomopoxvirus genome had the lowest G content among the viruses examined (Figure 1), and AT inversion was also observed (Extended Data Figure 6). In P. ursinus Cytomegalovirus,
![]()
Figure 4. Nucleotide differences in the three fragments of primate mitochondrial genome from 1 to 3. Left to right: (G - C), (G - T), (G - A), (C - T), (C - A) and (T - A).
whose GC content was the highest, both GC and AT inversions occurred. Mollivirus sibercum, which had the lowest C content, showed both GC and AT inversions, while DeBrazza’s monkey virus 1 showed both GA and GT inversions.
Ebola haemorrhagic fever, caused by the Ebola virus, can be fatal in humans. The Reston, Sudan, and Zaire strains did not show any nucleotide inversion in the three genome fragments (Figure 5). In the Tai Forest and Bundibugyo strains, however, CT inversion was clearly observed in the first fragment, and was accompanied by a decrease in GT content difference. These nucleotide content differences corresponded to the GC contents of the strains: Reston (G: 0.198, C: 0.210) [34], Sudan (G: 0.198, C: 0.216) [35], Zaire (G: 0.198, C: 0.213) [36], Tai Forest (G: 0.192, C: 0.231) [37], and Bundibugyo (G: 0.192, C: 0.228) [37]. An increase in C content and decrease in G content were observed in the Tai Forest and Bundibugyo strains. The calculated GC contents for the strains were: 0.406 (Reston), 0.411 (Zaire), 0.414 (Sudan), 0.420 (Bundibugyo), and 0.423 (Tai Forest). These results may indicate that Ebola virus evolution occurred over a short period of time.
![]()
Figure 5. Nucleotide content differences in the three fragments of Ebola virus genomes from 1 to 3. Left to right: (G - C), (G - T), (G - A), (C - T), (C - A) and (T - A).
3.5. Interspecies Evolution
Nucleotide content differences were calculated in vertebrate mitochondria (Extended Data Figure 7(a)), invertebrate mitochondria (Extended Data Figure 7(b)), non-animal mitochondria (Extended Data Figure 7(c)), chloroplasts (Extended Data Figure 7(d)) and nuclear DNA (Extended Data Figure 7(e)).
3.6. Definitive Universal Equation
Plotting (X - Y)/(X + Y) against (X - Y), the following linear relationship was obtained in mitochondria, chloroplasts, and chromosomes (Figure 6(a)), and viruses (Figure 6(b)): (X - Y)/(X + Y) = a (X - Y) + b, where X and Y are nucleotide contents, and (a) and (b) are constants. As (b) was almost null and (a) was ~2.0, (X - Y)/(X + Y) ≈ 2.0 (X - Y). In these genome analyses, which are independent of Chargaff’s parity rules (Extended Data Figure 8, left panels), the values of (a) for (G, C), (G, A), (G, T), (C, T), (C, A) and (A, T) were 2.5858, 1.85558, 1.9908, 1.9771, 1.9968 and 1.5689, respectively. Based on these results, (G + C), (G + A), (G + T), (C + A), (C + T) and (A + T) were 0.39, 0.54, 0.50, 0.51, 0.50 and 0.64, respectively. In virus genome analyses (Extended Data Figure 8, right panels), the constant values for (a) were 1.9 - 2.1, and the values for (X + Y) were 0.47 - 0.53. In contrast, in the normalization of nucleotide contents (G + C + A + T = 1), as (G = C) and (A = T) based on Chargaff’s parity rules, (2G + 2A = 1) is obtained. This equation is altered to (G + A = 0.5). This value is consistent with the value obtained above from genome analyses. Similarly, (G + T = 0.5), (C + A = 0.5) and (C + T = 0.5), although (G + C) and (A + T) cannot be determined. Therefore, the four nucleotide contents are expressed by the following regression lines,
![]()
Figure 6. Universal rules. Left side: relationship between (G - C) and (G - C)/(G + C), expressed by a linear regression, and right side: relationship between (G/C) and (G - C)/(G + C), expressed by a logarithmic function. The random numbers were not normalized.
plotted against G content: A = 0.5 - G, T = 0.5 - G, C = G and (G = G). Lines G and C overlap, as do lines A and T, and the former line is symmetrical to the latter against line (y = 0.25). The intercepts of lines G and C are close to the origin, while those of lines A and T are close to 0.5 at the vertical and horizontal axes. All organisms from bacteria to H. sapiens are located on the diagonal lines of a 0.5 square, termed the “Diagonal Genome Universe”, using the normalized values that obey Chargaff’s parity rules [27]. These relationships lead to (G or C) + (A or T) = 0.5. The present results indicate that a linear regression line equation, (X - Y)/(X + Y) = a (X - Y) + b, universally represents all normalised values, including the values deviating from Chargaff’s parity rules. This newly discovered equation clearly reflects not only Chargaff’s parity rules, based on hydrogen bonding between two nucleotides, but also natural rule.
A linear regression line was not obtained when using randomly chosen value (Figure 6(c)). Furthermore, plotting (X - Y)/(X + Y) against (X/Y), the following logarithmic function was obtained for all tested genomes as well as when using randomly chosen values (Figure 6 (a’)-(c’)): (X - Y)/(X + Y) = aln(X/Y) + b. As (b) was almost null and (a) was ~0.5, (X - Y)/(X + Y) ≈ 0.5 ln(X/Y). The ratio between two values, (X/Y), can be expressed by a logarithmic function, ~0.5 ln (X/Y) ≈ (X - Y)/(X + Y). Plotting the GC skew vs. G content, animal mitochondria were classified into two groups: high and low C/G [11]. This fact indicates that the ratio C/G and the GC skew are evolutionarily related to each other. Any change can be expressed universally by a definitive logarithmic function, (X - Y)/(X + Y) = a ln(X/Y) + b. The present results indicate that cellular organelle evolution is strictly controlled under these characteristic rules,
![]()
Figure 7. Universal rule governing genome evolution from viruses to H. sapiens. Upper pane: cellular organelles, middle panel: viruses, and lower panel: cellular organelles plus viruses.
although non-animal mitochondria, chloroplasts, and chromosomes are controlled under Chargaff’s parity rule [17]. The present study clearly shows that biological evolution, which seems to be based on complicated processes, is governed by simple universal equations. In fact, the codon [22] and genome evolution [23] were expressed in complete genomes by linear formulas, and it has been shown that a whole genome is constructed with gene assembly of small units with a similar amino acid composition [26], as shown in Figure 2 and Extended Data Figure 5.
4. Conclusion
Our findings showed that the most primitive extant ancestor of all living organisms is the M. brevicollis mitochondrion. In the normalised genome values, the relationship between the nucleotide difference, (X - Y), and (X - Y)/(X +Y), including biological evolution, can be expressed definitively by a linear regression line: (X - Y)/(X +Y) = a (X - Y) + b, where (a ≈ 2.0) and (b ≈ 0) are constants. (X + Y) = approximately 0.5. The nucleotide difference, (X - Y), is generally expressed by a linear regression line that crosses “0” at (X = Y), representing no evolution. In addition, the relationship between the ratio (X/Y) and (X - Y)/(X +Y) can be expressed definitively by a logarithmic function, (X - Y)/(X + Y) = a ln(X/Y) + b, where (a ≈ 0.5) and (b ≈ 0) are constants. The relationship between the skew “(X - Y)/(X + Y)” and the ratio (Y/X) is universally expressed by a logarithmic line that crosses “1” at (X = Y), representing no change. As the left sides, (X - Y)/(X + Y), are equal in both equations, 2.0 (X - Y) ≈ 0.5 ln(X/Y). Finally, (X - Y) ≈ 0.25 ln(X/Y) using the normalized values from viruses to H. sapiens (Figure 7).
Extended Data Text: Application of newly developed analytical method to various organisms’ mitochondrial genomes
No positive nucleotide content differences were observed in rat (Rattus norvegicus) or mouse (Mus musculus castaneus) mtDNA. The mitochondria of rodents, hedgehog (Erinaceus europaeus), guinea pig (Cavia porcellus), and hamster (Mesocricetus auratus) showed quite similar nucleotide content difference patterns, except for the presence of AT inversion in the second fragment (Extended Data Figure 2(a)). The Laotian rock rat, Laonastes aenigmamus, was described as a new family in 2005 [38], but the classification was changed to Diatomyidae in 2006 [39]. The mtDNA nucleotide content pattern of this rat completely differed from those of the other rodents examined in this study, although it resembled that of the Asian pangolin (Manis pentadactyla) (Extended Data Figure 2(a)). Pangolins are classified into two groups, African (Manis tetradactyla) and Asian, based on morphological characteristics [40]. In African pangolins, AT content inversion was observed in the second mtDNA fragment, while positive (T − C) differences were observed in all three mtDNA fragments of Asian pangolins. The African pangolin pattern resembled that of prosimian L. catta, while the Asian pangolin pattern resembled that of reptile Heteronotiabinoei (Bynoe’s gecko). Although they belong to the same genus, the two pangolins are remarkably separated in terms of genome biological divergence.
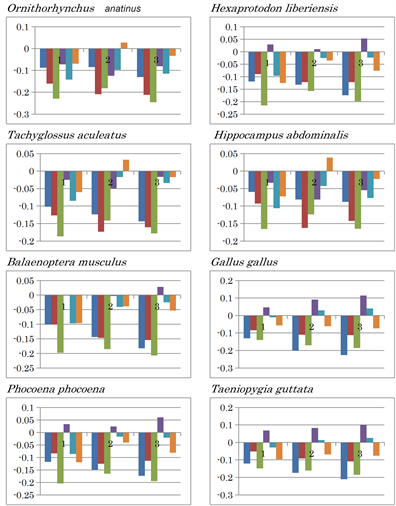
The ocean-dwelling mammal platypus, Ornithorhynchus anatinus, and the short-beaked echidna, Tachyglossus aculeatus, both belong to the order Monotremata, and seem to be the most primitive mammals. TA inversion occurred in the second mtDNA fragment of both these animals (Extended Data Figure 2(b)). Recently, it was reported that whales are related to hippopotamus [41 , 42]. Upon examination in the current study, consistent results were obtained based on Ward’s clustering analyses using amino acid compositions or nucleotide contents predicted from complete mitochondrial genomes as traits [43], or using 16S rRNA sequences [43]. However, nucleotide content difference patterns of mtDNA from whale (Balaenoptera musculus) and dolphin (Phocoena phocoena) species did not resemble that of the pygmy hippopotamus (Hexaprotodon liberiensis) or rhinoceros (Diceros bicornis) (Extended Data Figure 2(b)). In fact, the whale and dolphin patterns resembled those of primate species N. coucang and T. belangeri. This discrepancy should be clarified in the future. Nucleotide content difference patterns of mtDNA from birds (G. gallus and T. guttata) resembled that of primates (Figure 4 and Extended Data Figure 2(b)). This suggests that birds and primates have a similar level of divergence, as described previously [11]. However, a significant difference was observed between the two groups: the (C - T) difference in the second fragment was smaller than that of the first and third fragments in the mtDNA of birds.
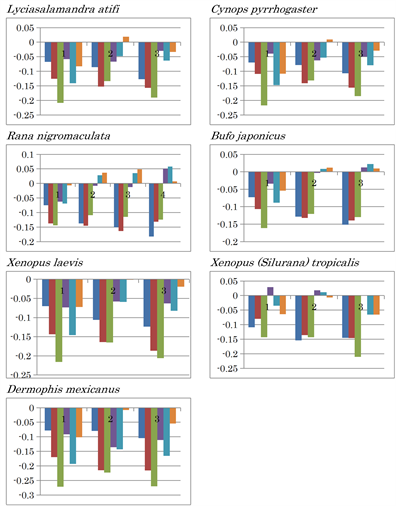
Amphibians are classified into the tailed Caudata (Lyciasalamandra atifi and Cynops pyrrhogaster) and the non-tailed Anua (Xenopus laevis, Rana nigromaculata, and Bufo japonicus) groups. This classification was also confirmed by nucleotide content difference patterns (Extended Data Figure 2(c)). AT, CT, and AC inversions were observed in the latter group, while only AT inversion was observed in the former, except for X. laevis. In X. laevis and Dermophis mexicanus (limbless amphibian) mtDNA, there is no nucleotide content inversion. In contrast, X. tropicalis mitochondria have both CT and CA inversion. Xenopus species, X. laevis [44] and X. tropicalis [45], whose complete genome sequences were analysed, are exciting models for the study of gene duplication that can result from hybridization among species [46]. Their mitochondrial nucleotide content difference patterns are quite different from each other. Eventually, the G and C values for X. tropicalis (G: 0.144, C: 0.281) mtDNA, which is diploid [47], were higher than those of X. laevis (G: 0.135, C: 0.235), which is a pseudo-tetraploid [48]. This fact clearly indicates that the highly evolved X. tropicalis diverged via a hybridized ancestor, such as X. laevis. Thus, X. laevis and X. tropicalis seem to be an excellent model to understand evolution based on gene duplication [49].
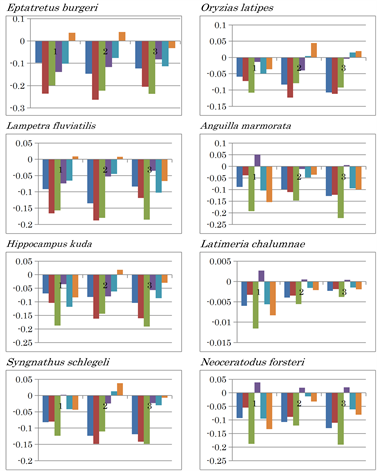
In aquatic vertebrates, the nucleotide content difference pattern of hagfish, Eptatretus burgeri, which has been proposed as an ancestor of vertebrates [50], resembled that of lamprey, Lampetra fluviatilis (Extended Data Figure 2(e)). Among the Gnathostomata, the mtDNA nucleotide content difference pattern of sea horse (Hippocampus kuda) differed from that of seaweed pipefish (Syngnathus schlegeli), although they both belong to the class Actinopterygii. The sea horse pattern resembled that of the Chondrichthyes species thorny skate (Amblyraja radiata) and brownbanded bambooshark (Chiloscyllium punctatum), amphibians (L. atifi and C. pyrrhogaster), and platypus (O. anatinus), which is an ancestor of mammals [51]. The mtDNA nucleotide content difference pattern of the coelacanth Latimeria chalumnae resembled that of eel (Anguilla marmonata) and lung fish (Neoceratodus forsteri), all of which were similar to the mtDNA of reptiles (Extended Data Figure 2(d)). These organisms are characterized by AT inversion in the second fragment. These similarities seem to reflect evolutionary processes from aquatic to terrestrial animals [11].
In non-animals, the mtDNA of fungal species Smittium culisetae showed the lowest GC and C contents, while the highest values were observed in Cycas species Cycas taitungensis mtDNA (Figure 1). GC inversion was observed in the former while both GC and AT inversions were observed in the latter (Extended Data Figure 3). In chloroplasts, no nucleotide inversion was observed in the red alga Cyanidioschyzon merolae, whose GC content was the lowest, while both GC and AT inversions were observed in the green alga Chara vulgaris, which had the highest GC content. Regarding C content, however, the C. vulgaris chloroplast C content was the lowest amongst the samples examined, while the mtDNA of the green alga Nephroselmi solivacea showed the highest C content, and was accompanied by GC and AT inversions.
GC and AT inversions in nucleotide content differences were observed in U. urealyticum, Mycoplasma pulmonis, Rickettsia prowazekii, Staphylococcus aureus, and Escherichia coli, while minor GC inversion was observed in S. coelicolor, which had the highest GC content of all organisms examined (Extended Data Figure 4). Amongst the archaea, both GC and AT inversions were observed in Pyrococcushorikoshii, while only GC inversion was observed in Halobacterium. The inversion patterns based on (G - T), (G - A), (C - T) and (C - A) occurred between low GC content bacteria such as Ureaplasma urealyticum (G: 0.131, C:0.127), Mycoplasma pulmonis (G: 0.133, C: 0.133), Rickettsia prowazekii (G: 0.146, C: 0.144), Staphylococcus aureus (G: 0.167, C: 0.165) and Pyrococcus horikoshii (G: 0.207, C: 0.212), and high GC content bacteria such as Halobacterium (G: 0.339, C: 0.340)and Streptomyces coelicor (G: 0.361, C: 0.359). Escherichia coli (G: 0.254, C: 0.254) showed the intermediate pattern.
The microsporidian protozoan Encephalitozoon cuniculi is a special eukaryote that lacks mitochondria, although it contains 11 chromosomes. Interestingly, the nucleotide contents of the chromosomes were almost identical (Extended Data Figure 5). GC and AT inversions, as well as other nucleotide content inversions, were observed among the 11 chromosomes of E. cuniculi, and nucleotide content difference patterns differed among the chromosomes (Extended Data Figure 5).
(a)
(b)
(c)
Extended Data Figure 1. Nucleotide content differences in the three fragments of the mitochondrial genomes of invertebrates. 1, 2, and 3 represent the sequentially divided first, second, and third fragments, respectively. Left to right: (G - C), (G - T), (G - A), (C - T), (C - A), and (T - A).

(a)
(b)
(c)
(d)
(e)
(f)
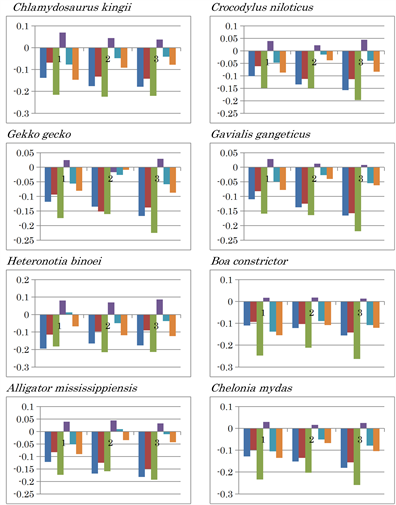
Extended Data Figure 2. Nucleotide content differences in the three fragments of the mitochondrial genomes of vertebrates. 1, 2, and 3 represent the sequentially divided first, second, and third fragments, respectively. Left to right: (G - C), (G - T), (G - A), (C - T), (C - A), and (T - A).
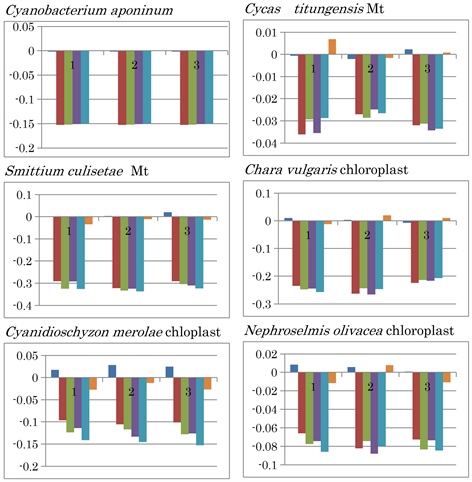
Extended Data Figure 3. Nucleotide content differences in the three fragments of the mitochondrial genomes of non-animal mitochondria and chloroplasts. 1, 2, and 3 represent the sequentially divided first, second, and third fragments, respectively. Left to right: (G - C), (G - T), (G - A), (C - T), (C - A), and (T - A).
![]()
Extended Data Figure 4. Nucleotide content differences in the three fragments of bacterial genomes. 1, 2, and 3 represent the sequentially divided first, second, and third fragments, respectively. Left to right: (G - C), (G - T), (G - A), (C - T), (C - A), and (T - A).
![]()
![]()
Extended Data Figure 5. Nucleotide content differences in the three fragments of the chromosome of microsporidian protozoan species Encephalitozoo ncuniculi. Left to right: (G - C), (G - T), (G - A), (C - T), (C - A), and (T - A).
![]()
Extended Data Figure 6. Nucleotide content differences in the three fragments of viral genomes. 1, 2, and 3 represent the sequentially divided first, second, and third fragments, respectively. Left to right: (G - C), (G - T), (G - A), (C - T), (C - A), and (T - A).
![]()
![]()
(a)
![]()
![]()
(b)
![]()
![]()
(c)
![]()
![]()
(d)
![]()
![]()
(e)
Extended Data Figure 7. Nucleotide differences. (a) Mitochondria of aquatic vertebrates (blue arrow) and terrestrial vertebrates (red arrow). (b) Mitochondria of high C/G invertebrates (black arrow) and low C/G invertebrates (red arrow). (c) Non-animal mitochondria of fungi (blue arrow) and plants (green arrow). (d) Chloroplasts. (e) Chromosomes of prokaryotes (blue arrow), archaea (green arrow), and eukaryotes (red arrow).
![]()
![]()
(a) (b)
Extended Data Figure 8. Universal rules. Left side: relationship between (X - Y) and (X - Y)/(X + Y), and right side: relationship between (X/Y) and (X - Y)/(X + Y). (a): organelles and (b): viruses.
Extended Data Table 1. List of viruses used.
NOTES
*This study has been initially posted on BioRxiv: http://biorxiv.org/cgi/content/short/371575v1.