Effective Removal of Nitrates Ions from Aqueous Solution Using New Clay as Potential Low-Cost Adsorbent ()
Received 19 September 2015; accepted 30 November 2015; published 3 December 2015


1. Introduction
Nitrate contamination in groundwater has become an ever increasing and serious environmental threat since 1970s [1] . The excessive application of fertilizers in agriculture causes the infiltration of large quantities of this ion into underground and surface water [2] . Nitrate, due to its high water solubility [3] , is possibly the most widespread groundwater contaminant in the world, imposing a serious threat to drinking water supplies and promoting eutrophication [4] [5] . Elevated levels of nitrate in drinking water can cause many health problems such as gastric cancer, which results from the reduction of nitrate to nitrosamines in the stomach [6] . In addition, methemoglobinemia or blue baby syndrome, a serious health risk, occurs when nitrate is converted to nitrite, which then reacts with the hemoglobin to cause blueness of the skin of newborn infants [7] . After ingestion of plants or water high in , acute poisoning may occur within 30 mins to 4 hrs in cattle. Thus, the problem occurs very quickly and often the cattle are observed to be normal one day and dead the next day [8] . Ward et al. [9] reviewed the epidemiologic evidence for the linkages between drinking water
, acute poisoning may occur within 30 mins to 4 hrs in cattle. Thus, the problem occurs very quickly and often the cattle are observed to be normal one day and dead the next day [8] . Ward et al. [9] reviewed the epidemiologic evidence for the linkages between drinking water 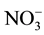 and the risk of specific cancers, adverse reproductive outcomes, and other health outcomes in the context of the current regulatory limit for nitrate in drinking water. Nitrate contaminated water supplies have also been linked to outbreaks of infectious diseases in humans [10] . Literature survey reveals that
and the risk of specific cancers, adverse reproductive outcomes, and other health outcomes in the context of the current regulatory limit for nitrate in drinking water. Nitrate contaminated water supplies have also been linked to outbreaks of infectious diseases in humans [10] . Literature survey reveals that 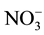 ion also causes diabetes and is a precursor of carcinogen [11] [12] . US Environmental Protection Agency (EPA) has set the maximum contamination level as 44 mg/l of
ion also causes diabetes and is a precursor of carcinogen [11] [12] . US Environmental Protection Agency (EPA) has set the maximum contamination level as 44 mg/l of 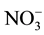 [13] .
[13] .
Therefore, numerous techniques for the removal of nitrate from water samples have been reported. These include biological de-nitrification [14] , chemical reduction [15] , reverse osmosis, electrodialysis [16] , and ion exchange [17] , but adsorption has been shown to be an economical and effective alternative for removing ions from water. Adsorption, in general, is the process of collecting soluble substances that are in solution on a suitable interface. Clays have been widely used as adsorbents for pollution control due to their high uptake ability, non-toxicity and large potential for ion exchange [18] .
The main aim of our study is to determine the adsorption capacity of locally available natural clay materials originated from a dam situated in Morocco (Agadir city) and to remove nitrate from its aqueous solution. Hence, the effect of various parameters on the adsorption process has been investigated: contact time, solution pH, and mass of adsorbent and nitrate concentration. The results are presented and discussed in this study. It is interesting to note that the adsorption of cationic and anionic dyes on this support is examined in our laboratory [19] .
2. Materials and Methods
2.1. Materials
Potassium nitrate used in the present study was of analytical grade and was obtained from Sigma-Aldrich. 1000 mg/l stock solution of nitrate was prepared. The required concentration of nitrate solution was obtained by serial dilution of the stock nitrate solution.
The untreated clay used in this work is crushed then sifted in order to get fractions <80 μm. Afterwards, the support is rinsed many times with distilled water, of pH = 6.5 and of conductivity = 1.1 μS. After every rinse, the clay is put in a sedimentation test, and during one hour of decantation, we recover the two thirds of supernatant volume. Finally, it is placed in an oven between 110˚C and 120˚C during 24 hrs, before being subjected to different adsorptions.
2.2. Nitrate Adsorption Studies
The ability of our new clay mineral (NC) to adsorb nitrate ion was tested at different conditions (Table 1) using a series of batch tests. A number of parameters such as reaction time, concentration of nitrate, adsorbent dose and pH, affecting the removal of nitrate ion have been varied widely in order to optimize the removal process. The experiments were carried out in 200 ml conical flasks by mixing a pre-weighted amount of adsorbent with 100 ml of nitrate solution (300 mg/l). The solution was agitated at 400 rpm on a stirrer during 180 min (adsorption equilibrium), at natural pH (pH = 5.1), at T = 20˚C and with R = 1 g/l. All samples were filtered (through a 0.45 μm Millipore) prior to analysis using disposable syringes in order to minimize interference of the particles fines with the analysis. The nitrate concentrations in liquid phase were determined by a spectrophotometric method (spectrophotometer type JASCO V-630, λmax = 410 nm) based on colourimetric determination using chromotropic acid and sulfuric acid (99%). This method is highly selective, very sensitive, and can be quickly and
easily applied [20] [21] . On calculating the difference between the initial and final concentrations of nitrate in aqueous phase, the amount of adsorbed nitrate at equilibrium (Qe) was calculated (Equation (1)) and the percentage of nitrate removed was also determined (Equation (2)):
 (1)
(1)
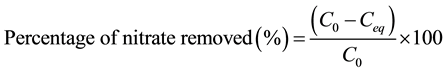 (2)
(2)
with:
Qe: Quantity of nitrate adsorbed per gram of adsorbent (mg/g);
C0: Initial concentration (mg/1);
Ce: Equilibrium concentration (mg/l);
R: Mass of adsorbent per liter of aqueous solution (g/1).
2.3. Characterization of the Adsorbent
X-ray powder diffraction pattern was obtained using the diffractometer XPERT-PRO type PW3064, with copper anticathode. The spectrum of XRD shows that the untreated clay contains a large amount of the Kaolinite (38%) and Illite (24%) followed by Quartz (20%) and Calcite (18%) (Figure 1), the amounts of the phases were calculated by X’PERT Highscore plus software. The BET (Brunauer-Emmett-Teller) surface area of clay was measured by nitrogen (N2) adsorption-desorption isotherms at (77 K) using an automated gas sorption system (Micromeritics, Quantachrome instrument). The determined surface area is equal to 53.47 m2/g.
The morphology of the clay powder particles was observed by scanning electron microscopy (SEM, using Zeiss Ultra Plus) with a coupled energy dispersive spectroscopy (EDS), at 5 kV, after gold coating. The morphological aspects of the clay particles are outlined in Figure 2. The porosity of the surface is clearly visible. The scanning electron micrograph shows the typical regular shapes of the clay particles. The powder is rich in porous and microporous particles that formed noticeable cage-like cavities. These cavities provide suitable binding sites for nitrates ions. On the basis of this fact, it can be concluded that our clay presents an adequate morphology for nitrate adsorption. The chemical compositions (Table 2) have been checked using energy dispersive spectroscopy (EDS) (Figure 3). The point of zero charge (PZC) of our clay was determined by the solid addition method [22] . The pHPZC of NC was determined to be 5.6.
In order to determine the surface functional groups, the Fourier Transform Infrared (FTIR) spectrum of the NC was obtained by using a Bruker Vertex 70 spectrophotometer, at a resolution of 4 cm−1 and averaging over 20 scans, in the range 400 - 4000 cm−1.
The FTIR spectrum of our clay was depicted in Figure 4. The bands at 3695.7 cm−1 and 3620.3 cm−1 correspond to the inner OH stretching vibration, while that at 3435.5 cm−1 represents the outer surface OH stretching vibration. Si-O bending vibration was observed at 1114.2 cm−1, 1031.7 cm−1 and 1008.5 cm−1, while the stretching vibration was observed at 698.5 cm−1 and 796.1 cm−1. Absorption band at 913.0 cm−1 correspond to the Al-O bending vibration and peak intensities recorded at 537.7 cm−1, 469.7 cm−1 and 430.2 cm−1 are due to Al-O- Si skeletal vibrations [23] [24] .
![]()
Figure 1. X-ray diffraction pattern of the NC.
![]()
Figure 2. SEM micrograph of the NC particles.
![]()
Figure 3. Dispersive energy spectrum (EDS) of the NC.
![]()
Table 2. Atomic and mass percentage of the NC constituents.
3. Results and Discussion
3.1. Effect of Contact Time
Adsorption kinetic is an important characteristic for evaluating the efficiency of adsorption. The kinetic behavior of this process was studied at natural pH (Table 1). It appears from Figure 5 that the kinetic of nitrate adsorption consists of two phases, an initial rapid phase when the process is very fast and a second slower phase when it reaches equilibrium. The initial high rate of nitrate uptake is probably due to the greater availability of binding sites near the surface of the clay [25] . The kinetic study indicates that the adsorption equilibrium state is reached after a contact time of 180 min with Qe = 215.69 mg/g since no change in the adsorbed amount is detected afterward.
3.2. Adsorption Kinetics
The kinetics of adsorption data was processed to understand the dynamics of adsorption process in terms of the order of rate constant. Two kinetic models were applied to the adsorption kinetic data in order to investigate the behavior of adsorption process of nitrates onto our clay. These models are the pseudo-first-order and pseudo-second-order models.
The pseudo-first-order equation is given by [26] :
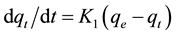 (3)
(3)
where qe and qt are the amounts of nitrate adsorbed (mg/g) at equilibrium and at time t (min), respectively, and k1 (l/min) is the rate constant of first-order adsorption.
The pseudo-second order process can be written as follows [27] :
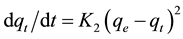 (4)
(4)
where k2 g/(min.mg) is the rate constant of second-order adsorption. The fit of these models was checked by each linear plot of ln(qe − qt) versus t (Figure 6), (t/qt) versus t (Figure 7). The kinetic constants and correlation coefficients of the pseudo-first-order and the pseudo-second-order models are given in Table 3. The results showed that the adsorption system followed the pseudo-first-order model, with correlation coefficient value of 0.998 which is better than the second-order-model (0.928) and the calculated qe,cal value from the model is also in good agreement with the experimental value qe,exp (Table 3).
![]()
Table 3. Kinetic parameters for the adsorption of nitrates ions onto NC.
![]()
Figure 5. Effect of contact time on the adsorption of nitrates ions.
![]()
Figure 6. Pseudo-first-order kinetic plots.
![]()
Figure 7. Pseudo-second-order kinetic plots.
3.3. Effect of Initial Solution pH
The effect of initial solution pH on nitrate removal is illustrated in Figure 8. The initial pH values were adjusted by adding 0.1 M of HCl or NaOH. It is evident from the figure that the percentage removal of nitrate decreased from 92.72% to 38.96% for increase in pH from 2 to 10. The variation of adsorption with pH can be explained by the electrostatic interaction between the adsorbent and adsorbate. With increase of pH values, the surfaces gradually decrease in the extent of positive charging and become negative at pH of about 6 (the isoelectric points of clay is at pH = 5.6). Thus, the high adsorption capacity at low pH is mainly due to the strong electrostatic interaction between the positively charged sites of adsorbent (presence of H+) and the anions. However, lower sorption of the nitrates ions at alkaline pH could be attributed to the abundance of OH− ions which will compete with the pollutant for the same sorption sites [28] .
3.4. Effect of Adsorbent Dose
For studying the effect of mass of adsorbent on nitrate adsorption onto our clay, experiments were performed with varying amounts of adsorbent, ranging from 0.2 to 2 g/l (Figure 9). It is evident from the figure that the removal of nitrate increased from 9.84% (0.2 g/l) to 72.97% (1 g/l). This study revealed that the adsorption capacity of clay increased with increase in adsorbent dose up to a particular region and then reaches an equilibrium level at higher doses. However it is observed that after dosage of 1 g/l, there was no significant change in percentage removal of nitrate. This may be due to the overlapping of active sites at higher dosage. So, there was not any appreciable increase in the effective surface area resulting due to the conglomeration of adsorbent particles [29] . Thus, 1 g/l in 100 ml of nitrate solution was considered as an optimum dose and was used for further study.
3.5. Effect of Initial Nitrate Concentration
The influence of initial nitrate concentration on adsorption percentage was estimated (Figure 10). The initial concentration of nitrate solution was varied from 100 mg/l to 450 mg/l with optimum adsorbent dose (1 g/l). The result obtained is presented in graphical form as percentage removal versus initial nitrate concentration. It is evident from the graph that the percentage removal of nitrate decreased from 97.77% to 53.40% for initial nitrate concentration of 100 mg/l to 450 mg/l. It is due to the limitation of adsorption sites on the clay mineral surface [30] [31] . This can be explained as follows: with increase in the initial nitrate concentration the amount of adsorbate species in the solution increases, but the amount of adsorbent remains constant and hence the percentage removal decreases with increase in initial nitrate concentration. The higher uptake of nitrate at low concentration may be attributed to the availability of more active sites on for lesser number of adsorbate species [32] .
![]()
Figure 8. Influence of pH on the adsorption of nitrates ions.
![]()
Figure 9. Effect of adsorbent dose on the adsorption of nitrates ions.
![]()
Figure 10. Influence of initial concentration on the adsorption of nitrates ions.
3.6. Adsorption Isotherms Studies
Adsorption isotherms indicate distribution of adsorbate between solution and adsorbent at the equilibrium state of the adsorption process [33] . It is expressed by relating the amount of adsorbate taken up per gram of adsorbent, Qe (mg/g), to the equilibrium solution concentration, Ce (mg/l), and at different concentrations of nitrates ions solutions (100, 150, 200, 250, 300, 350, 400 and 450 mg/l), (Figure 11). Monolayer saturation is attained. The type of adsorption isotherm model is very important in order to understand the adsorption behavior for solid-liquid adsorption system [34] . In the present study, Langmuir and Freundlich models were tested to study the adsorption behavior of nitrate as these two are the most commonly used.
3.6.1. Langmuir Isotherm
The Langmuir isotherm is based on the assumptions that adsorption takes place at specific homogeneous sites within the adsorbent, there is no significant interaction among adsorbed species, and the adsorbent is saturated after the formation of one layer of adsorbate on the surface of adsorbent [35] . The Langmuir isotherm equation can be written as follows:
![]() (5)
(5)
The linearized form of Equation (5) can be written as Equation (6):
![]() (6)
(6)
where Ce is the equilibrium concentration of the remaining solute in the solution (mg/l), Qe is the amount of the solute adsorbed per mass unit of adsorbent at equilibrium (mg/g), Qm is the amount of adsorbate per mass unit of adsorbent at complete monolayer coverage (mg/g), and KL (l/mg) is a Langmuir constant. In this work, we used the linear form of Langmuir isotherm equation and therefore, a plot 1/Qe versus 1/Ce should indicate a straight line. The Qm and KL values were calculated from the slopes (1/Qm) and intercepts (1/KL∙Qm) of linear plots of 1/Qe versus 1/Ce (Figure 12). The Langmuir isotherms parameters are given in Table 4. The isotherm data of layered clay fits well the Langmuir equation with a correlation coefficient, R2 = 0.992 and shows excellent linearity (Figure 12). The theoretical monolayer saturation capacity Qm,cal is in good agreement with the experimental value Qm, exp (Table 4).
One of the essential characteristics of the Langmuir model could be expressed by a dimensionless constant called equilibrium parameter, RL which is determined as follows [36] [37] :
![]() (7)
(7)
where C0 is the initial nitrate concentration (mg/l). The value of RL indicates the type of isotherm to be irreversible
![]()
Figure 11. Adsorption isotherm of nitrates ions.
![]()
Figure 12. Langmuir isotherm linear plots for the adsorption of nitrates ions.
![]()
Table 4. Langmuir and Freundlich isotherm parameters.
(RL = 0), favorable (0 < RL < 1), linear (RL = 1), or unfavorable (RL > 1). RL values are well within the defined range (Table 4) and indicate that the adsorption process is favorable.
3.6.2. Freundlich Isotherm
The Freundlich isotherm was proposed as the earliest empirical equation and was shown to be consistent with exponential distribution of active centers, characteristic of heterogeneous surfaces [38] . This model states that reactions take place in several sorption sites and as the amount of solute adsorbed rises, the binding surface energy decreases exponentially which means multilayer sorption [39] . The isotherm is expressed by the following equation:
![]() (8)
(8)
Logarithmic form of Equation (8) can be written as Equation (9):
![]() (9)
(9)
where Kf ((mg/g) (1/mg) 1/n) and n are Freundlich adsorption isotherm constants, the values of Kf and 1/n are determined from the intercept and slope of the linear regressions and are presented in Table 4. A plot of ln(Qe) versus ln(Ce) for the studied samples is shown in Figure 13. The value 1/n gives an indication on the validity of the adsorption of adsorbent-adsorbate system. A value 1/n between 0 and 1 that indicates a favorable adsorption [40] . In addition to that, this also indicates that the adsorption capacity increases, and further, adsorption sites appear. When 1/n > 1, the adsorption is not favorable, the adsorption connections become weak and the adsorption capacity decreases. The values of 1/n were less than one (Table 4) revealed favorable adsorption conditions. The correlation coefficient value is lower than Langmuir value. Therefore, adsorption does not follow Freundlich isotherm. Theoretical correlation of the experimental equilibrium adsorption data for the nitrate-clay system was properly explained by the Langmuir isotherm model.
![]()
Figure 13. Freundlich isotherm linear plots for the adsorption of nitrates ions.
4. Conclusion
This study investigated the adsorption characteristics and suitability of our new natural clay mineral (NC) as potential adsorbent for the removal of nitrate from aqueous solutions using batch technique. The results showed that this clay could be used as potential sorbent and it was highly effective as low-cost adsorbent for the removal of nitrates ions from aqueous solutions. The batch study parameters, pH of solution, mass of adsorbent, initial solution concentration and contact time were found to be effective on the adsorption processes. The adsorption equilibrium was attained within 3 hrs. From the studies conducted, it was established that pseudo-first-order kinetic model described the kinetic rate. The percentage removal was found to decrease with increase in pH. The increase in adsorbent dosage increased the percent removal of nitrate due to the increase in adsorbent surface area in adsorbent dosage. The equilibrium data fitted well the Langmuir isotherm equation and this adsorbent showed large uptake capacity of nitrate (Qm, cal = 250 mg/g, Qm, exp = 244.06 mg/g). Our clay was a potential candidate as a highly efficient adsorbent for remediation of nitrate contaminated water owing to its exceptional uptake capacity as well as high selectivity for this anionic contaminant.
NOTES
![]()
*Corresponding author.