Effect of the Cation Size Disorder at the A-Site on the Structural Properties of SrAFeTiO6 Double Perovskites (A = La, Pr or Nd) ()
1. Introduction
Double perovskite oxides, A2BB'O6 or AA'BB'O6, where A and A' are alkaline―earth and/or rare―earth metals and B and B' are transition metals, have been investigated [1] . These materials are especially interesting in view of the possibility that B and B' cations could be ordered at the octahedral sites, giving rise to novel electrical and magnetic properties due to B - O - B' interaction [2] . The study of these oxides was initiated in the 1950s and since then several hundred of these compounds have been produced and studied because they exhibit quite interesting structural, electronic as well as magnetic properties [2] . One can anticipate many more of these types of compounds to be realized given the diversity of the ions that can occupy both the A- and B-sites. The last few years ordered double perovskite oxides have been received an enormous amount of attention because certain members of this family of oxides exhibit spectacular physical phenomena such as colossal magnetoresistance and half metallicity in the ferrimagnetic Sr2FeMoO6 [3] and La2VRuO6 [4] compounds.
The physical properties of double perovskite DP are generally determined by the cations occupying the B, B' sites. Thus, special attention has been given to the factors that impact the B-sites arrangement, namely the charge, size and electronic configuration of B cations as well as the A/B size ratio. Anderson et al. [2] have concluded that an ordered structure is most likely to be formed if the charge difference between B and B' is two or higher. An exception to this condition is the case of LaCaMnCoO6 [5] ; this compound was ordered even though the B - B' charge difference is one. The ideal perovskite has a primitive cubic structure with the formula ABO3, where the B-ions are at the corners of the cubic cell and the A-ions at the cubic octahedral (12 coordinated) site. There is a variety of ordered structures known in perovskite oxides; among them the double perovskite (A2BB'O6 or AA'BB'O6) and the triple perovskite (A3BB'2O9) structures are the most popular.
These ordered structures can be obtained mainly by substituting other cations at their A or B sites of the ABO3 structure and occur due to the charge difference. Many of these ordered perovskite cations Ti, Fe and Mn as the B-cations, are interesting due to their optimum dielectric characteristics for applications in electronic industry [6] - [8] .
2. Experimental Details
The double perovskite SrAFeTiO6 samples where A is (La, Pr, Nd) respectively, were prepared.
Most of the chemicals used in the preparation were Alfa Acer of purity 99.9%. The method used for preparation of these double perovskite is in accordance with the well known procedures [9] [10] . The X-ray diffraction and FTIR measurements were carried out for the samples under investigation.
SrLaFeTiO6, SrPrFeTiO6 and SrNdFeTiO6 were synthesized by both conventional and precursor solid state reaction from the compounds SrCO3, La2O3, Fe2O3, TiO2, Pr7O11 and Nd2O3 raw materials with purity more than 99%. The grouped compounds were mixed together and then ground in an agate mortar for 45 minutes at least. After that the compounds were heated into a Carbolite air furnace to 900 degrees of centigrade for 12 hours. Then it was pressed into pellets under 7 MPa pressure for 30 seconds following by preheating in air at 900˚C for 24 hours with intermediate grinding and finally it was calcinated at 1200˚C for 12 hours. Phase analysis and characterization were carried out by X-ray diffraction (XRD) using Cu Kα radiation on both X’Prot Bruker D8 and Schimadzu 6000.
3. Results
Determination of the crystal structure, the lattice parameters and the space group symmetry become of central importance in the study of structural, electrical and magnetic properties of the double perovskite materials [11] .
The results of the XRD for the samples obtained after conventional solid state method is shown in Figures 1(a)-(c) for the samples SrLaFeTiO6, SrPrFeTiO6 and SrNdFeTiO6. Clearly, the quality of the samples is poor whatever grinding times and/or heating temperatures are committed.
By following the precursor method, good quality samples are obtained as shown in Figure 2.
A selection range is given in Figure 3 in order to show the difference in the peak position for each sample. The shift in the reflection positions indicates differences in the lattice parameters and/or the type of the crystal structure.
3.1. The XRD Analysis
For quantitative analysis, the XRD patterns are analyzed by the standard Rietveld refinement method. Three
![]() (a)
(a)![]() (b)
(b)![]() (c)
(c)
Figure 1. (a) The XRD results of the SrLaFeTiO6 sample prepared by the conventional solid state reaction; (b) The XRD results of the SrPrFeTiO6 sample prepared by the conventional solid state reaction; (c) The XRD results of the SrNdFeTiO6 sample prepared by the conventional solid state reaction.
![]()
Figure 2. The X-ray patterns for the GI series samples. The green colored is SrNdFeTiO6, the red colored is SrPrFeTiO6 and the blue one is SrLaFeTiO6 sample respectively. Shifts in the intensity are clearly seen above 2θ = 38 to the end of the scan.
![]()
Figure 3. A selected 2θ range to show the difference in the peak positions for each sample. The green colored is SrNdFeTiO6, the red colored is SrPrFeTiO6 and the blue one is SrLaFeTiO6 sample respectively.
types of the crystal structures commonly observed in the double perovskites were selected for the analysis purpose. They are cubic, tetragonal and orthorhombic structures.
3.1.1. The SrLaFeTiO6 Structure
For accurately determination of the crystal structure and its lattice parameters commonly observed among the double perovskites are tested. The XRD data of the SrLaFeTiO6 structure was used along cubic structure with space group Fm3m, Tetragonal with space group I4/m, tetragonal with space group I2/n and orthorhombic with space group Pnma. The criteria used for the judgments are: all peaks must be fitted to certain crystal structure model and if all the peaks are fitted to more than one space group then the lowest Bragg’s factor in the better model is obtained. It was worthy to mention here, that the fitting was started by selecting the highest symmetry structure which is cubic, and subsequently going down to the orthorhombic at the end Figure 4 as well as the corresponding Table 1.
3.1.2. The SrPrFeTiO6 and SrNdFeTiO6 Structures
Similar investigations are carried out for the SrPr/NdFeTiO6 samples. The best fit is obtained for the orthorhom-
![]()
Figure 4. Refined XRD patterns of the SrLaFeTiO6 sample. It shows the orthorhombic structure with (Pnma) space group.
![]()
Table 1. The atom positions, the space groups, the lattice parameter and the refinement reliability factors for Sr/(La, Pr, Nd) FeTiO6 samples.
bic structure by adopting the Pnma space group. The data for these two samples are shown in Figure 5 and Figure 6.
In an ideal perovskite, the A-cation is surrounding by 12 oxygen anions in a regular dodecahedral environment and the B-cation is octahedrally coordinated by 6 oxygen ions, giving rise of 180˚ B-O-B bond angles [12] . By replacing A-cation with smaller one, the BO6 octahedral would be rotated from the cubic crystallographic axis to release the structural stress. The cooperative rotation of the B'O6 octahedra consequently, leads to decrease in the B'-O-B angle and reduction in the coordination number of the A cation [13] .
All the pervious reasons let the tolerance factor play one of the conventional methods used to compare the elements and compounds which represent the structural parameters. Figure 7 shows the relation between the
![]()
Figure 5. Refined XRD patterns of the SrPrFeTiO6 sample. It shows the orthorhombic structure with (Pnma) space group.
![]()
Figure 6. Refined XRD patterns of the SrNdFeTiO6 sample. the orthorhombic structure is shown with (pnma) space group.
![]()
Figure 7. The tolerance factor versus the ionic radius for the three samples SrLaFeTiO6, SrPrFeTiO6 and SrNdFeTiO6 respectively.
![]()
Figure 8. The IR spectra of the three samples SrNdFeTiO6, SrPrFeTiO6 and SrLaFeTiO6 respectively. The depth of the stretching frequencies ~600 cm−1, were increased from SrNdFeTiO6 to SrLaFeTiO6 with broadening in the peak of the SrLaFeTiO6 sample.
ionic radius R and the tolerance factor. Obviously the tolerance factor increase with R.
3.2. The IR Spectroscopy Results
Since every type of bond has a different natural frequency of vibration, and two of the equivalent types of bond in two unusual compounds are in two slightly diverse environments, no two molecules of dissimilar structure have exactly the same infrared absorption pattern, or spectrum. Although some of the frequencies absorbed in the two cases might be the similar, in no case of two special molecules with their spectra be identical. Thus, the infrared spectrum can be used for molecules such as fingerprint for humans [14] .
It is well known that vibrational spectroscopy is very useful in studying crystal structure of ordered perovskite [15] . The BO6 octahedra present in ABO3 perovskites can be considered as molecules. They have 15 normal modes of vibrations (A1g + Eg + 2T1u + T2g + T2u) when they are in octahedral symmetry [15] . Deviation from this symmetry will result in broadening of lines or even splitting [16] .
4. Discussion and Conclusions
X-ray diffraction patterns measured at room temperature for the samples SrAFeTiO6 (A = La, Pr or Nd) using two different methods and the precursor method gives better pattern than that the conventional method which gives a multi phase (Figures 1(a)-(c)). The best fitting of these compounds could be const as their structure, and the closed ionic radius for all samples illustrated that, they have an orthorhombic structure with a Pnma space group. There are also some differences in the lattice parameter where differ from SrLaFeTiO6 up to SrNdFeTiO6 as follows: a = 5.557654 A˚, b = 7.824075 A˚, c = 5.537659 A˚ for La, and a = 5.52728 A˚, b = 7.79671 A˚, c = 5.51395 A˚ for Pr, and a = 5.51584 A˚, b = 7.79255 A˚, c = 5.51391 A˚ for the Nd. The patterns were subjected to careful analysis by means of the standard Reitveld methods using the FullProf suite [17] . Three structural models were selected instead of the refinement processes for accurate determination of the best crystal structure matching the data. The anti-site effect of the Fe and Ti atoms is considered by their shared occupation between the B and B' sites. The XRD characterization showed that all the samples are of single-phase. The best fitting results are achieved when the anti-site disorder is considered in all the samples. The obtained lattice parameters, atoms positions as well as the reliability factors are shown in Table 1.
The tolerance factor is a measure of the internal pressure for perovskite structures. It is given by [18]
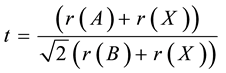 (1)
(1)
where t represents the tolerance factor, r(A), r(B) and r(X) are the ionic radius for A-site, B-site and oxygen respectively. The values of the tolerance factor for SrLaFeTiO6, SrPrFeTiO6, SrNdFeTiO6, are calculated using the SPuDS program [19] and found to be 0.971, 0.965 and 0.959, respectively. The reduction of symmetry in the compounds is related to the small values of the A-site radius (rA) in order to accommodate empty spaces, which by its role leads to tilting the octahedral of the Fe and Ti atoms. A guide for this behavior is a plot of the tolerance factor and the ionic radii of the La, Pr and Nd ions against the tolerance factor. Tilting of the octahedral in SrLaFeTiO6, SrPrFeTiO6 and SrNdFeTiO6 leads to different electronic environment around the Fe ions and impose magnetic moments miss alignment. Therefore, the magnetic properties of these compounds might be influenced by the internal pressure [20] .
Successful preparation of double perovskite oxides of chemical formula SrAFeTiO6 (A = La, Pr or Nd) has been achieved by following the precursor method. The structural characterization analysis proved that it has similar crystal structure (orthorhombic) with a Pnma space group, which found to be consistent with its tolerance factor. The substitution in the A-site has been seen to alter the crystal structure from higher symmetry to lower symmetry as the size of the A-site decreases.
Distortion in the TiO6 octahedra may result from Jahn-Teller splitting due to the ion existence of Ti4+ or Fe3+ or internal chemical pressure. That belongs to the cation size mismatch (σ2) which depends on the average radius of the cations in A-site of the perovskite structure. The FTIR spectra results of the samples under study are given in the Figure 8. This FTIR spectrum resembles the general appearance of the double perovskite structure. A shift in the peak positions in going from La up to Nd sample in the group has been noticed.
NOTES
*Corresponding author.