Mitochondrial Dysregulation in Skeletal Muscle from Patients Diagnosed with Alzheimer’s Disease and Sporadic Inclusion Body Myositis ()
1. Introduction
Mitochondria are dynamic organelles that play a pivotal role in cellular function, not only as a major site of ATP production, but also as an organelle regulating energy metabolism, protein turnover, cellular proliferation, and apoptosis [1] [2] . Not surprisingly, defects in this organelle can have a profound impact on cellular function and are implicated in the development of metabolic and neurological disease [3] -[5] . Mitochondrial function is dependent on a number of factors including mitochondrial biogenesis, mitochondrial autophagy, and dynamics [6] [7] . Mitochondrial dynamics is a concept that includes mitochondrial movement within the cell and mitochondrial interactions controlled by fusion/fission events [8] . The importance of these events has recently become evident with the identification of genes responsible for fusion (mitofusin 1 and 2, optic atrophy protein 1) and fission (dynamin Related Protein 1 and fission 1) [9] -[12] . Pharmacologic and/or genetic manipulation of these genes can result in gross mitochondrial abnormalities including, altered substrate metabolism, reduced oxygen consumption, a decline in ATP synthesis, and mtDNA nucleotide dyshomeostasis [8] . Furthermore, impairment in mitochondrial function is detrimental to myofiber health and can result in fiber death and muscle atrophy [2] .
Disruption of mitochondrial dynamic pathways has been documented in the brains of Alzheimer’s patients and occurred in conjunction with mitochondrial network fragmentation, increased reactive oxygen species (ROS) production, and accumulation of pathological protein fragments including amyloid β and tau [13] -[16] , although it is not known if other tissues are affected. Much less is known about the role of the mitochondria in the development of sporadic Inclusion Body Myositis (sIBM), however, mtDNA dyshomeostas along with structural and functional abnormalities have been observed in patients diagnosed with the disease [17] -[19] . Furthermore, Boncompagni, et al. [20] observed structural and functional alterations in mitochondria from MCK-βAPP mice, which over express amyloid b precursor protein (AβPP) in skeletal muscle and display characteristics of the disease. Importantly, these alterations are observed preceding the reported appearance of histopathological and clinical features and may represent a key early event in disease pathology [17] [20] [21] .
Both AD and sIBM are characterized by the accumulation of protein fragments such as amyloid β (Aβ), a peptide fragment processed from the AβPP, in the brain and skeletal muscle, respectively [22] [23] . In fact, the presence of Aβ in both diseases originally suggested a common pathology. However, while the role of Aβ in the development of AD has been established, whether Aβ accumulation in skeletal muscle from sIBM patients is a cause or secondary side effect is not known [24] -[26] .
Recent work has demonstrated that AβPP can interact with both the outer and inner mitochondrial membrane import channels and prevent import of de novo synthesized nuclear-encoded mitochondrial proteins [27] -[29] . Furthermore, intramitochondrial amyloid β may directly impair mitochondrial function by disrupting mitochondrial dynamic pathways [16] [30] . This provides a potential mechanism for the observed impairments in mitochondrial function and suggests a role for mitochondrial dynamic pathways in the development of sIBM. The purpose of the current paper was to examine pathways regulating mitochondrial fusion and fission as well as regulators of mitochondrial biogenesis and autophagy in skeletal muscle from patients diagnosed with AD and sIBM compared with healthy, elderly individuals.
2. Methods
Human Samples. Human skeletal muscle samples were obtained from the National Register Disease Interchange (New York, New York). Samples were obtained from patients diagnosed with AD (n = 5; age = 79 ± 3; 2 males and 3 females), sIBM (n = 2; age = 69 ± 4; all males), and elderly controls with no evidence of muscle disease (n = 6; age = 66 ± 4; all males). Samples were collected from the vastus lateralis muscle within 12 hours post mortem. Samples were shipped overnight on dry ice and processed for assessment of mRNA, protein, and enzyme activity.
Gene Expression. RNA was extracted using an RNeasy Mini Kit (Qiagen) and DNase I treatment (Qiagen, Valencia, CA), according to the manufacturer’s instructions. qRT-PCR was performed using an ABI 7900 Sequence Detection System instrument and Taq Man Universal PCR Master Mix according to the manufacturer’s specifications (Applied Biosystems, Foster City, CA) and as previously described [31] . mRNA of uncoupling protein three (UCP3), manganese superoxide dismutase (SOD2), catalase, beclin, peroxisome proliferator-activated receptor gamma coactivator 1-alpha, myosin heavy chain 7B, myosin heavy chain 2, and myosin heavy chain 4 were assessed. Primers and 5# FAM-labeled TaqMan probes were purchased as prevalidated assays (Applied Biosystems, Foster City, CA). Relative quantification of target genes was calculated using the 2−ΔCT method. Derivation of the 2−ΔCT equation has been described in Applied Biosystems User Bulletin No. 2 (P/N 4303859). Target gene expression was normalized against GAPDH.
Protein Content. Western analysis was performed as previously described using cell lysates harvested in Mammalian Cell Lysis Buffer (Sigma Aldrich) [31] . Proteins (30 μg) were separated using a 10% CriterionTris·HCl gel (Bio-Rad, Hercules, CA) and subsequently transferred to a polyvinylidene difluoride membrane (Bio-Rad). Blots were probed with primary antibodies against GAPDH (1:1000; Santa Cruz Biotechnology, Santa Cruz, CA), peroxisome proliferator-activated receptor (PPARα) (1:1000; Abcam, Cambridge, MA), mitofusin 2 (1:1000; Cell Signaling, Danvers, MA), optic atrophy protein 1 (1:1000; Abcam, Cambridge, MA), dynamin related protein 1 (1:1000; Abcam, Cambridge, MA) phosphorylated 5’-AMP-activated protein kinase (AMPK) and total AMPK (1:1000; both Abcam, Cambridge, MA) followed by anti-rabbit, mouse, or goat secondary antibodies (1:10000; Jackson Immuno Research Laboratories, West Grove, PA). Proteins were visualized using Super-Signal Chemiluminescent Substrate (Pierce, Rockville, IL) and a ChemiDoc XRS Imaging System (Bio-Rad). Protein content was normalized to GAPDH and phosphorylated AMPK was adjusted for total AMPK and normalized to GAPDH.
Enzyme Activity. Enzyme activities were assessed in muscle homogenates (20-fold dilution). Sample buffer consisted of 0.1 mol/l KH2PO4/Na2PHO4 and 2 mmol/l EDTA, pH 7.2. Phosphofructokinase (PFK), citrate synthase (CS), malate dehydrogenase (MDH) and beta hydroxyacyl-CoAdehydrogenase (β-HAD) activities were determined spectrophotometrically as previously described [31] [32] .
Citrate synthase catalyzes the formation of citrate and coenzyme A (CoASH) from acetyl-CoA and oxaloactetate. CoASH reduces DTNB and CS activity was determined from the reduction of DTMB over time. Ten microliters of a 1:5 diluted muscle homogenate was added, in triplicate, to 170 μl of a solution containing Tris buffer (0.1 M, pH 8.3), DNTB (1 mM, in 0.1 M in Tris buffer) and oxaloacetate (0.01 M, in 0.1 M Tris buffer). Following a 2-minute background reading, the spectrophotometer (SPECTRAmax ME, Molecular Devices Corporation, Sunnyvale California) was calibrated and 30 μl of 3 mM acetyl CoA was added to initiate the reaction. Absorbance was measured at 405 nm at 37˚C every 12 seconds for 7 minutes. Maximum CS activity was calculated and reported as μmol/min/mg.
For the determination of β-hydroxyacyl-CoA dehydrogenase, oxidation of NADH to NAD was measured. In triplicate, 35 μl of whole muscle homogenate was added to 190 μl of a buffer containing 0.1 M liquid triethanolamine, 5 mM EDTA tetrasodium salt dihydrate, and 0.45 mM NADH. The spectrophotometer (SPECTRAmax PLUS 384, Molecular Devices Corporation, Sunnyvale California) was calibrated and 15 μl of 2 mM acetoacetyl CoA was added to initiate the reaction. Absorbance was measured at 340 nm every 12 seconds for 6 minutes at 37C. Maximum BHAD activity was calculated and reported as μmol/min/mg.
Malate dehydrogenase reversibly catalyzes the oxidation of malate to oxaloacetate using the reduction of NAD+ to NADH. The rate of the disappearance of NADH was measured spectrophotometrically at 340 nm at 37˚C. Briefly, 10ul of sample were pipetted in triplicate in wells. Then, 290 ul of reaction media (0.1 M potassium phosphate buffer, pH 7.4 plus 0.006 M oxaloacetic acid, prepared in potassium phosphate buffer plus 0.00375 M NADH, prepared in potassium phosphate buffer) was added to the wells and samples were read for 5 minutes at 340 nm. The rate of disappearance of NADH was analyzed and expressed relative to protein content. Data is expressed as means ± S.E.M.
Phosphofructokinase phosphorylates fructose-6-phosphate to fructose-1,6-bisphosphate, and is a key regulatory step in glycolysis. Phosphofructokinase activity was measured spectrophotometrically at 340 nm at 37˚C. Briefly, 30 ul of sample homogenate were pipetted in triplicate. Assay buffer (12 mM MgCl2 , 400 mM KCL, 2 mM AMP, 1 mM ATP, 0.17 mM NADH, 0.0025 mg/mL, Antimycin 0.05 mg/mL Aldolase 0.05 mg/mL GAPDH, in 100 mM Tris buffer, pH = 8.2) was then added to each well. After a 2-minute background reading, 3 mM fructose-6-phosphate was added to each sample well and followed by a 7-minute kinetic reading. Changes in absorbance across time were recorded and expressed relative to protein content. Data is expressed as means ± S.E.M.
Statistical Analysis. Results were analyzed with non-parametric Kruskal-Wallis one-way analysis of variance analysis. Comparisons between groups were assessed using a Mann-Whitney-Wilcoxon (MWW) test. The level of significance was set at P < 0.05.
3. Results
Transcriptional regulation in skeletal muscle from patients diagnosed with sIBM and Alzheimer’s disease. mRNA analysis revealed that AβPP was significantly higher in patients diagnosed with sIBM compared to AD patients and controls (Figure 1(A)). Despite this, mRNA of UCP3 and the antioxidants catalase and SOD2 were all significantly higher in AD patients compared to elderly controls (Figures 1(B)-(D)). While there were no significant differences in UCP3 concentrations between sIBM patients and controls, there were trends for higher and lower catalase and SOD2 mRNA, respectively. Beclin, a regulator of autophagy, was significantly higher in the AD patients with a trend for higher mRNA observed in sIBM patients compared to controls (Figure 1(E)). Peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC1α) expression, a regulator of mitochondrial biogenesis, was significantly higher in both sIBM and AD compared to controls (Figure 2(A)). To determine whether changes in expression patterns were associated with differences in skeletal muscle fiber type,
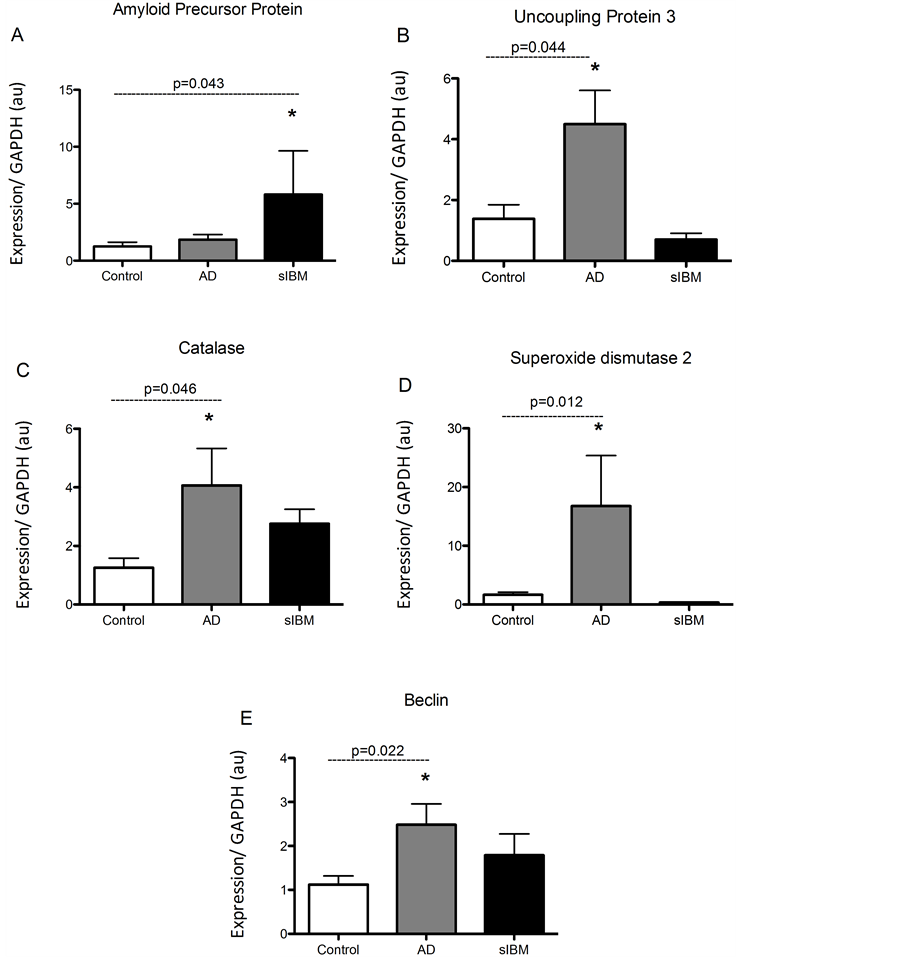
Figure 1. Transcriptional regulation in skeletal muscle from patients diagnosed with AD and sIBM. Relative mRNA content of amyloid precursor protein (APP) (A), uncoupling protein 3 (UCP3) (B), catalase (C), manganese superoxide dismutase (SOD2) (D), and beclin (E) in skeletal muscle from patients diagnosed with AD (n = 5), sIBM (n = 2), and elderly individuals (n = 6). Data are presented as means ± S.E.M. and normalized to GAPDH mRNA, *P < 0.05.
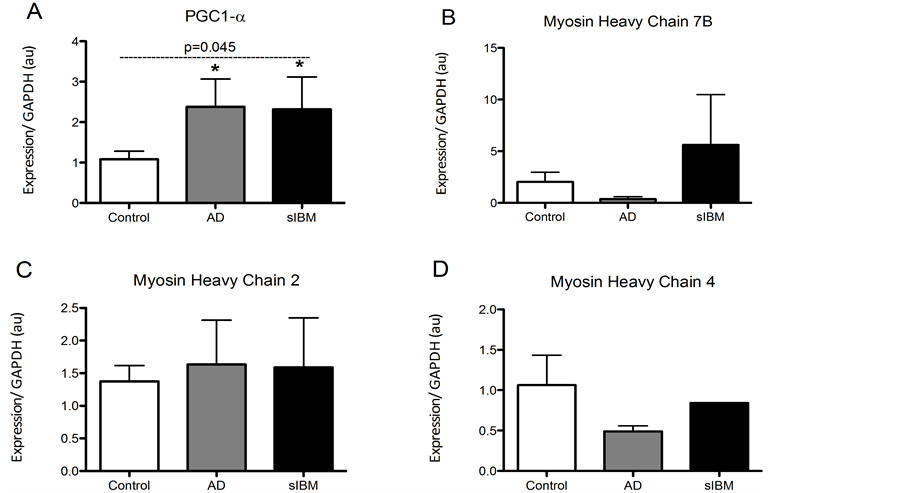
Figure 2. Transcriptional regulation in skeletal muscle from patients diagnosed with AD and sIBM. Relative mRNA content of peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC1a) (A), myosin heavy chain 7B (MHC7B) (B), myosin heavy chain 2 (MHC2) (C), and myosin heavy chain 4 (MHC4) (D) in skeletal muscle from patients diagnosed with AD (n = 5), sIBM (n = 2) for PGC1a, MHC7B, and MHC2 and n = 1 for MHC4), and elderly individuals (n = 6). Data are presented as means ± S.E.M. and normalized to GAPDH mRNA. *P < 0.05, compared to controls.
mRNA of myosin heavy chain 7b, 2, and 4, markers of type 1, type 2A, and type 2B, respectively, were assessed (Figure 2(B)-(D)). There were no differences in expression of genes responsible for fiber type suggesting that observed differences may not be due to differences in fiber type. We were however only able to measure MHC4 in one sIBM sample, and therefore only limited conclusions can be made.
Protein content in skeletal muscle from patients diagnosed with sIBM and Alzheimer’s disease. While there were no significant differences in PGC1a protein content, MFN2, OPA1, and DRP1 were all significantly lower in both patient populations compared to elderly controls (Figure 3(B)-(D)). Additionally, both phosphorylated and total 5’-AMP-activated protein kinase (AMPK) protein content was significantly lower in sIBM patients compared to both AD and controls, respectively (Figure 3(E)).
Enzyme activity in skeletal muscle from patients diagnosed with sIBM and AD. Due to lack of sample, we were only able to measure enzyme activity in one patient diagnosed with sIBM, although we were able to measure enzyme activity in all of the AD and control samples. While there were no significant differences in enzyme activity of phosphofructokinase, malate dehydrogenase, or citrate synthase (Figure 4(A), Figure 4(B) and Figure 4(D)), there was a trend for reduced activity of beta hydroxyacyl CoA dehydrogenase activity, the primary regulator of beta-oxidation in AD patients compared to elderly controls (Figure 4(C)).
4. Discussion
The current study supports previous findings and provides a potential mechanism of mitochondrial dysfunction by demonstrating reduced protein content of MFN2 and OPA1 in patients diagnosed with AD and sIBM [33] . Additionally, there was a similar decline in DRP1 protein in sIBM patients, although it did not reach statistical significance in AD patients. Mitochondrial dynamics, the repetitive cycles of fusion and fission, have been recognized as a critical process in the maintenance of mitochondrial homeostasis. These opposing processes determine the architecture of the entire mitochondrial population of the cell with fission events segregating dysfunctional mitochondria from the network to be processed for autophagy and fusion events allowing for equilibration of matrix metabolites and membrane components [7] [34] [35] . A down regulation of the primary regulators of
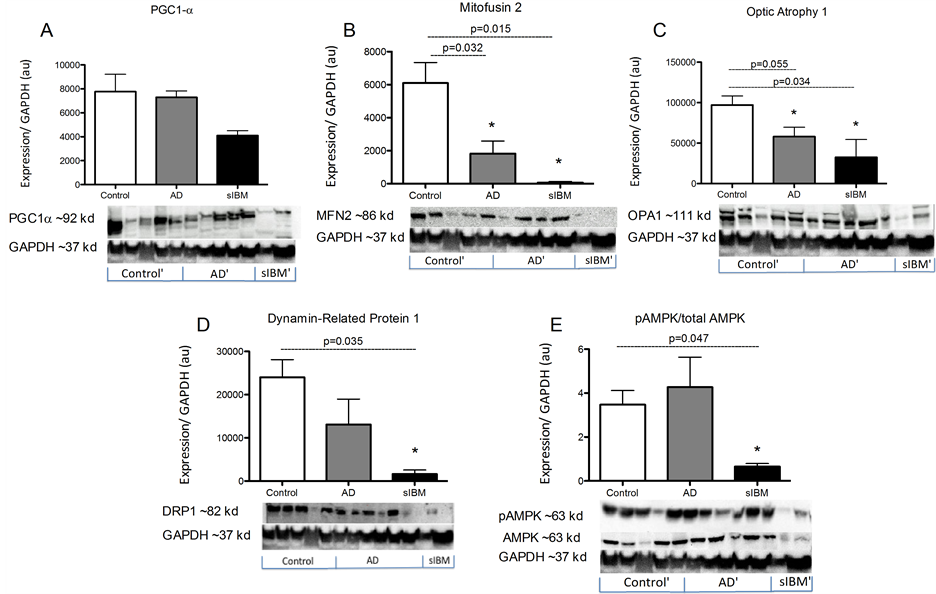
Figure 3. Metabolic enzyme activity in skeletal muscle from patients diagnosed with AD and sIBM. Protein content of peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC-1α) (A), mitofusin 2 (B), optic atrophy protein 1 (C), dynamin related protein 1 (D), and phosphorylated 5’-AMP-activated protein kinase (AMPK) and total AMPK (E) in skeletal muscle from patients diagnosed with AD (n = 5), sIBM (n = 2), and elderly individuals (n = 6). Data are presented as means ± S.E.M. and normalized to GAPDH protein content. *P < 0.05, compared to controls.
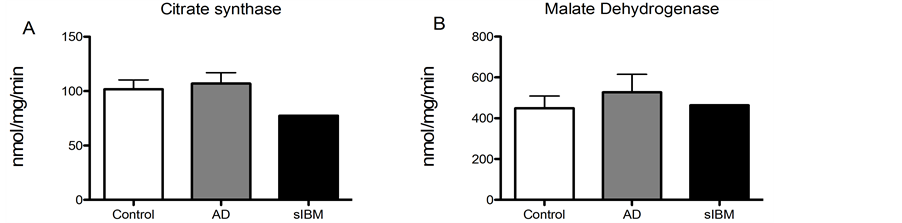
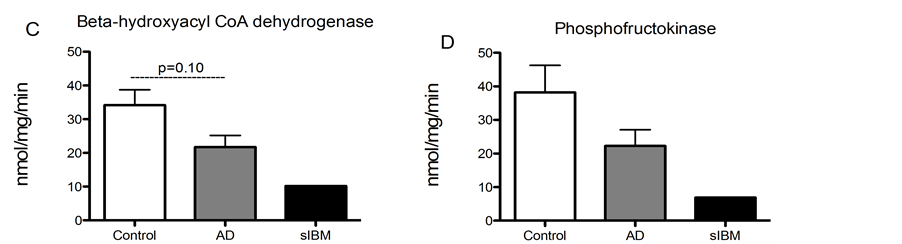
Figure 4. Metabolic enzyme activity was measured in skeletal muscle from patients diagnosed with sIBM, AD and healthy controls. Maximal enzymatic activities of citrate synthase (A), malate dehydrogenase (B), beta hydroxyacyl-Co A dehydrogenase (C), and phosphofructokinase-1 (D) in skeletal musclefrom patients diagnosed with AD (n = 5), sIBM (n = 1), and elderly individuals (n = 6). Data presented at means ± S.E.M. and presented as nmol of mg protein per minute. *P < 0.05, compared to controls.
these pathways could account for the structural and functional alterations previously observed in sIBM as well as demonstrating for the first time, alterations in mitochondrial signaling pathways in skeletal muscle from patients diagnosed with AD.
The above effects are observed in conjunction with a significant increase in PGC1a mRNA in both patient populations, although there were no significant differences in PGC1 protein content. PGC1a is regulated at a number of levels and it has been previously demonstrated that PGC1a mRNA does not reflect protein content [36] [37] . The observed increase in PGC1amRNA along with increased expression of regulators of mitochondrial autophagy, may indicate a compensatory effect to remove and replace damaged, dysfunctional mitochondria in the face of dysregulated mitochondrial dynamic pathways [38] .
While these findings indicate a potential disruption of mitochondrial dynamics and function, the mechanisms underlying the observed similarities between the phenotypes may differ. While AbPP mRNA was higher in sIBM patients, which supports previous findings [39] ; there were no differences between AD patients and elderly controls. The lack of an increase in APP mRNA in AD patients in the current study does not support a link between mitochondrial defects and increased AbPP expression in skeletal muscle in AD patients. Additionally, there was reduced content of both phosphorylated and total AMPK in sIBM patients compared to elderly controls. It has been demonstrated that increased AMPK activity results in an up regulation of MFN2, OPA1, and DRP1 in murine skeletal muscle [40] . Therefore, a down regulation of AMPK may explain the down regulation of MFN2, OPA1, and DRP1 observed in the patients with sIBM. The fact that there was reduced AMPK protein content may also suggest a potential therapeutic strategy for treatment of individuals afflicted with this disease.
While there was reduced phosphorylated and total AMPK content in sIBM patients, there were no significant differences between AD patients and controls. This indicates that something other than AMPK may be responsible for the decline in MFN2 and OPA1 protein content in AD patients. Interestingly, there was an increase in UCP3, SOD2, and Catalase mRNA. This transcriptional up regulation of antioxidant genes may be a protective mechanism in response to an increase in intracellular reactive oxygen species concentrations in skeletal muscle from AD patients [41] [42] . ROS has been demonstrated to regulate expression of both fusion and fission proteins and may therefore also be responsible for the alterations observed in the current work [43] [44] . On the other hand, genetic or pharmaceutical alterations of mitochondrial fusion or fission pathways has been shown to result in increased production of reactive oxygen species [8] . Unfortunately, it is not possible to determine cause and effect from the current study.
There are limitations to the current study. The very low N of our sIBM patients is a concern. However sIBM, although considered the most common myopathy in older individuals, still has a low prevalence rate with only 0.002% of individuals over 50 in the US diagnosed with the disease [45] . Additionally, the AD patients were significantly older than the controls and sIBM patients, making it difficult to exclude an age effect. However, all of the individuals studied were over the age of 60 and the observed similarities along with differences between the two patients populations would suggest that the observations in the current work are not simply due to age.
5. Conclusion
In conclusion, we are reporting for the first time that regulators of mitochondrial fusion and fission are down regulated in skeletal muscle from patients diagnosed with AD and sIBM. These findings suggest a potential for mitochondrial dysfunction in skeletal muscle in both disease states although the underlying causes may differ. This work highlights the need for the future study in an effort to identify new opportunities for prevention or treatment of these life-altering diseases.
NOTES
*Corresponding author.