1. Introduction
Agro-industrial activities have resulted in the accumulation of lignocellulosic residues and the fungi have an important function in the decomposition of this organic matter. The use of these residues as carbon and energy source depends on the capacity of the fungus to produce and excrete lignocellulolytic enzymes [1].
Lentinula edodes (shiitake) is a saprotrophic fungus that degrades lignocellulosic residues and produces lignocellulolytic enzymes as laccase, manganese peroxidase, cellulase and xylanase [2-6]. Besides of the lignocellulolytic enzymes production of industrial interest, L. edodes is capable of converting lignocellulosic residues, e.g. eucalypt logs and sawdust, coffee husks, and corncobs, in mushrooms that can be used in the human and animal feeding [1,7,8].
In addition to be considered as delicacies, the edible mushrooms are a versatile food, low in energy and fat but containing considerable amounts of protein, dietary fibers, vitamins and mineral. They have effective mechanisms for absorption and accumulation of some elements, such potassium (K), iron (Fe), copper (Cu), zinc (Zn), selenium (Se), lithium and other metalloids [9-12], and depending on the fungal growth conditions, these mushrooms can be enriched with minerals which present low availability in the soil or in the foods [9,12].
L. edodes has been showed as an alternative for production of Se-enriched food [11,13]. However, the productivity of mushrooms can be influenced by the activity of respiratory and lignocellulolytic enzymes, which has not been investigated. In Pleurotus ostreatus, high level of Se in the substrate, decreases the productivity and alters the morphology of the mushrooms [12].
Selenium is a metalloid naturally found in the soil and part of the structure of enzymes like glutathione peroxidase, deiodinase, thioredoxin redutase and selenoprotein P, which were involved in the oxidative metabolism [14,15]. Furthermore, the selenate and selenite reduction to elementary selenium can be catalyzed by microbial enzymes [16].
Thus, the objective of this study was to evaluate the effect of Se in the respiratory and enzymatic activity of L. edodes growing in lignocellulosic residue.
2. Material and Methods
2.1. Microorganism
In this study, UFV 11, UFV 16 and UFV 53 L. edodes (Berk.) Pegler isolates from the collection of the Department of Microbiology of the Federal University of Viçosa (MG, Brazil) were used. These isolates were obtained from basidiocarps of commercial mushrooms that were produced in Moji das Cruzes, São Paulo, Brazil. The stock cultures were maintained on potato dextrose agar (PDA, Merck). Mycelium of each culture was grown at 25˚C on the surface PDA agar in Petri dishes.
2.2. Growth Conditions and Analyses of Respiratory Activity
Three L. edodes isolates were cultivated in 20 g of substrate based on eucalypt sawdust (78% eucalypt sawdust, 20% rice bran, 0.4% CaCO3 and 1.6% CaSO4) with addition of 0.32, 0.64, 0.96 or 1.28 mmol∙L−1 of sodium selenite (Na2SeO3) [11]. Tap water was added to reach 65% moisture content.
The substrates were placed in glass flasks (150 mL), autoclaved at 121˚C for 20 min and, after cooling, each flask was inoculated with one agar disc (8 mm diameter) containing fungal mycelium grown in PDA for 15 d. These flasks were incubated at 25˚C for 21 d. The control treatment was the eucalypt sawdust without addition of Na2SeO3 and incubated in the same experimental conditions.
For determine respiratory activity the flasks were coupled to the respirometer of continuous flow with detector of infrared (TR-RM8 Respirometer Multiplexer—Sable systems). CO2 production measurement was carried out every seven hours [17] and bottles remained coupled to the respirometer for seven days.
2.3. Enzymatic Assay and Protein Quantification
Every seven incubation days substrate samples with fungal growth were collected for analyses of laccase (EC 1.10.3.2), cellulase (EC 3.2.1.4), xylanase (EC 3.2.1.8/ EC 3.2.1.32/EC 3.2.1.136) specific activities and for soluble protein determination.
For protein extraction and preparation of enzyme extracts (EE), 3 g of the substrate colonized by the fungi were put in Erlenmeyer (125 mL) containing 10 mL of sodium acetate buffer (0.1 mol∙L−1 and pH 4.8) and agitated at 150 rpm for 30 mim [3,18]. Next, the suspension was filtered in Millipore membranes (Whatman 1).
Laccase activity was determined by oxidation of the 2,2’-azino-bis-3-ethil benzothiazole-6-sulfonic acid (ABTS) at 37˚C [19]. The enzymatic reaction was prepared with 0.6 mL of sodium acetate buffer (0.1 mol∙L−1 and pH 5.0), 0.3 mL of EE and 0.1 mL of ABTS (1 mol∙L−1). This reaction was incubated in a water bath at 37˚C for 10 min and, subsequently, absorbance was determined at 420 nm. One unit of laccase/enzyme activity was defined as the amount of enzymes required for oxidation of 1 µmol of ABTS per min at 37˚C.
Cellulase and xylanase activities were determined by the amount of released reducing sugars from filter paper (Whatman 1) and xylan, respectively [3,20,21]. The filter paper and xylan were used as enzymatic substrate. One units of these hydrolases activity was defined as the amount of enzymes catalyzing the release of 1 mmol∙L−1 of glucose or xylose per min at 50˚C.
Reducing sugars were determined by dinitrosalicylic acid method (99.5% dinitrosalicylic acid, 0.4% phenol and 0.14% sodium metabisulfite) [3,22].
Soluble protein was determined by the colorimetric method described by Bradford [23] and used for calculation of enzymes specific activity.
2.4. Statistical Analysis
Factorial delineation was used with three fungal isolates (UFV 11, UFV 16 and UFV 53), five Se concentrations (0, 0.32, 0.64, 0.96 and 1.28 mmol∙L−1), three incubation time (7, 14 and 21) and three replicates. The data were subjected to analysis of variance and regression (p < 0.05) using Saeg software (version 9.1, Federal University of Viçosa).
3. Results
The respiratory activity of L. edodes was different among isolates (p < 0.05) and decreased in function of sodium selenite concentration in substrate. In isolate UFV53, this reduction was higher than 40% and, for each isolate a specific regression model was obtained (p < 0.05, Figure 1). These models are important to determine the sodium selenite dose that should be added in the substrate for a better fungal growth.
Se enhanced the laccase specific activity (p < 0.05) when compared with the treatment without addition of this element (Figure 2). The activity of this enzyme was different among the isolates (p < 0.05) with higher value obtained by UFV11, at of 0.96 and 1.28 mmol∙L−1 concentrations (Figure 2).
Cellulase specific activity was low and presented significant difference (p < 0.05) between sodium selenite
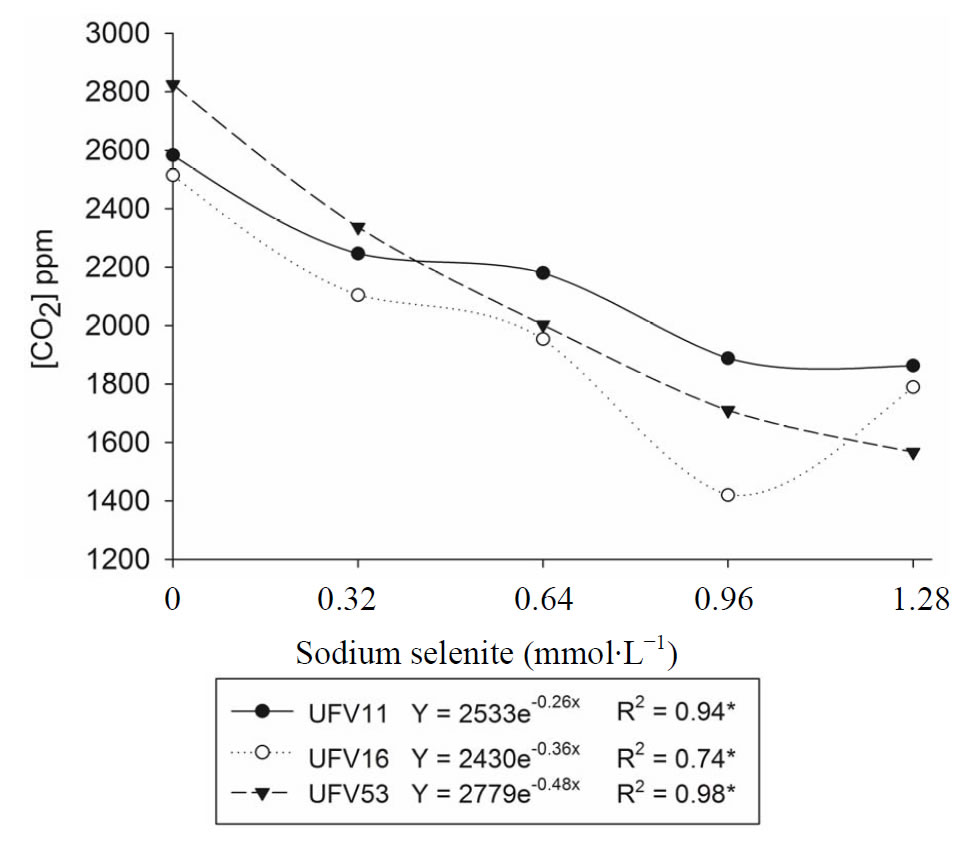
Figure 1. Lentinula edodes respiratory activity, UFV11, UFV16 and UFV53 isolates, cultivated in the eucalypt sawdust containing or not different sodium selenite doses, during 21 days at 25˚C. *Significant regression model (p < 0.05).
concentrations added to the substrate and also between the analyzed strains (Figure 2). The highest activities of this enzyme were observed in the cultivation of isolates UFV11 and UFV16 in substrate with 1.28 mmol∙L−1 of sodium selenite (Figure 2).
The xylanase specific activity was also low, varied in function of the addition of sodium selenite in substrate and the incubation time (Figure 2) and, no difference among the isolates of L. edodes was observed (p < 0.05).
4. Discussion
The reduction of the respiratory activity (p < 0.05) shows that Se in concentration greater than 0.96 mmol∙L−1 can be toxic to the fungus (Figure 1). This toxicity had also been observed by Nunes [24] once that the mycelial growth of L. edodes in Petri dish, containing PDA or eucalypt sawdust, decreased as the sodium selenite dose in the culture medium was increased. According to this author, the inhibitory effect of mycelial growth was observed regardless the composition of the cultivation medium and, in the concentrations greater than 1 mmol∙L−1 the colony diameter was halved. The Se concentration higher than 12.8 mg∙kg−1 also decreased the mycelial growth and altered the shape of the P. ostreatus mushroom [12]. The toxicity of high doses of other metals such as cobalt, cadmium (Cd) and magnesium have also been shown in the growth of Psathyrella atroumbonata [25] and Paxillus involutus [26]. Furthermore, the fungus stress induced by toxicity of certain metals depends on nutrients availability, as observed in Trichoderma viride and Rhizopus arrhizus, cultivated in culture medium with low nutritional level or supplemented with Cd, Cu and Zn [27]. To avoid the inhibitory effect of high doses of Se on the growth of L. edodes, Nunes et al. [11] suggested the addition of up to 1.28 mmol∙L−1 of sodium selenite in the water cold shock that is used to induce fructification of mushroom after mycelial growth. The inhibitory effect maybe avoided due to the smallest time of exposition of the fungus to the high doses of sodium selenite.
No correlation between the reduction of the respiratory activity with increase of the sodium selenite concentration (Figure 1) and lignocellulolytic enzymes activity was observed (Figure 2). This fact can be due to factors such as 1) greater synthesis of extracellular enzymes to keep fungal growth under conditions of stress, 2) the presence of the selenium in the substrate do not inhibiting the lignocellulolytic enzymes activity or 3) this element have an activator effect on the activity of these enzymes. The last two factors seem to be most probable, because enzymatic activity was greater in the presence of the selenium (Figure 2). Although reports have not been found in the literature on the effect of the Se in the microbial enzyme activity, several studies have showed that the laccase activity is inhibited in presence of certain metals like iron, mercury and silver [28], Cd, lead (Pb) and mercury [29]. Cadmium, Cu, Pb, manganese (Mn) and Zn also affected cellulase and xylanase activity of P. ostreatus cultivated in lignocellulosic residues [30,31]. Nevertheless, the presence of minerals such as Cu, Mn and Zn increased ligninases activity of different Basidiomycetes [3,30,32]. Hence, the selenium might have been an activator of laccase in this study.
A high specific activity of the laccase was also observed during P. ostreatus growth in different lignocellulosic residues [18] and L. edodes in eucalypt bark [3]. In contrast with the present study, Cavallazzi, Brito, Oliveira, Villas-Bôas and Kasuya [3] showed that of this enzyme declines with incubation time. Ohga and Royse [7] noticed that the laccase activity of L. edodes was higher during the mycelial growth, decreasing quickly in the beginning of the fructification period. According to the last authors [7] this behavior is due to the fact that, the expression of the laccase genes, measured as mRNA levels, is maximum during the substrate colonization and decrease during fructification. So, the activity of this enzyme can also be used for monitored of the fungal development in a specific substrates or in substrate enriched with mineral or still to determine the inhibitory effect of a element on the fungal growth.
The highest reduction of the respiratory activity and lowest laccase activity observed in UFV53 may indicate the greatest sensibility of this isolate to the Se (Figures 1 and 2) or the physiological characteristic of this isolate, because, it presented high manganese peroxidase activity and low laccase activity in eucalypt bark without addition of Se [3,18]. Therefore, may have not occurred an

Figure 2. Specific activity of lignocellulolytic enzymes (laccase, cellulase and xylanase) of Lentinula edodes, UFV11, UFV16 and UFV53 isolates, cultivated in eucalypt sawdust containing or not different sodium selenite doses, during 21 days at 25˚C.
increasing in the expression of extracellular laccase to compensate the reduction of respiratory activity. And, in this case, Se also is not inhibiting the enzymatic activity, unlike, it can have activated the laccase activity (Figure 2).
Although low cellulase and xylanase activities were observed, selenium did not inhibit the activity of these hydrolases that are responsible for cellulose and hemicellulose degradation (Figure 2). This low cellulase activity and the variation of the activity of this enzyme among the isolates of L. edodes (Figure 2) were also shown by Cavallazzi, Brito, Oliveira, Villas-Bôas and Kasuya [3] and Philippoussis et al. [5]. According to Leatham [2] L edodes is a moderately cellulolytic fungus, with low exo-1,4-D-glucanase activity. In contrast, Silva, Machuca and Milagres [5] observed high cellulase activity of L. edodes that was quite uniform among the evaluated isolates. The difference between this study and that carried out by Silva, Machuca and Milagres [5] may be due to the substrate composition and cultivation conditions used, once; the biosynthetic potential of Basidiomycetes is highly dependent of the growing conditions [5]. Thus, the low cellulase activity (Figure 2) can be in function of the chemical composition of eucalypt sawdust, which presents higher lignin levels than cellulose and hemicelluloses and, not necessarily, due to the addition of sodium selenite to the substrate.
The low xylanase activity and the reduction of this activity along of incubation time can also be related to the substrate composition (Figure 2) because, the main factors to an efficient production of xylanase by microorganisms are the appropriate choice of the substrate and the optimization of the composition of the culture medium [33].
Cellulase and xylanase activities were higher at days seven and fourteen of cultivation, while laccase activity was higher at day twenty-one (Figure 2). Considering that during white rot fungus growth in lignocellulosic substrate, the laccase acts firstly in the complex polymers degradation releasing cellulose and hemicelluloses imprisoned in the lignin layers, it was expected that hydrolytic enzymes activities would happen in opposite order as observed (Figure 2). In this case, it is possible to suppose that there was a partial acid hydrolysis of the lignin during the substrate thermal treatment, since pH was 4.5. During acid hydrolysis of lignocellulosic materials, the acid molecule penetrates into the lignin layers, accelerating the hydrolysis of hemicellulose and noncrystalline areas of cellulose [34]. In this hydrolysis, the polysaccharides react with the water releasing dimers and monomer that are used to initial fungal growth [35]. Furthermore, the low initiate laccase activity can also be justified by addition of rice bran to the substrate (Figure 2), which according to Cavallazzi, Brito, Oliveira, VillasBôas and Kasuya [3], is a carbon source immediately used by fungi for mycelial growth, promoting a fast and efficient substrate colonization.
The partial acid hydrolysis of lignocellulosic compounds, due to the thermal treatment releasing sugars with higher metabolization rates can also explain the high L. edodes respiratory rate in a short incubation period (Figure 1).
Thus, it was demonstrated that although high doses of Se inhibit the mycelial growth, this mineral can increase the laccase activity. The characterization of lignocellulolytic enzymes activity, that was different among the L. edodes isolates, contribute for an appropriate choice of the fungal isolates and the Se concentration, that should be added to the substrate, for a better enzymatic production and, consequently a greatest biological efficiency of Se-enriched mushrooms.
5. Conclusion
Selenium increases laccase activity of L. edodes cultivated in eucalypt sawdust and, depending on the dose, this mineral can be toxic to the fungus, inhibiting the respiratory activity. These results are important to define the ideal Se dose to be added to the substrate for greater lignocellulosic residues degradation and, consequently, guarantee a higher production of Se-enriched mushrooms.
6. Acknowledgements
The authors are very grateful to Brazilian financial Institutions: Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG).
NOTES