Synthesis and Structure of Dimeric Copper (I) Complex from Bis[(2,2’)-dimethyl 2,2’-(1,10-phenanthroline-2,9-diyl) bis(methan-1-yl-1-ylidene)-bis(hydrazinecarbo dithioate)] ()
1. Introduction
Schiff bases bearing bidentate N,N’ chelating agents such as 1,10-phenanthroline (phen) have been used extensively as a ligand in both analytical and preparative coordination chemistry as well as in the preparation of many mixed-ligand complexes [1]. These complexes have interesting physiochemical properties and have high potential as antibacterial, antifungal and chemotherapeutic agents [2]. Metal complexes derived from 1,10-phenanthroline have also been found to be widely used in areas such as molecular catalysis, solar energy conversion, calorimetric analysis, herbicides, molecular recognition, self-assembly, antineoplastic agents and nucleic acid probes [3]. The key feature of 1,10-phenanthroline is π-electron deficient nitrogen which stabilizes various metal complexes in lowered oxidation states [4]. The copper/phen complexes are able to interact and cleave the DNA/RNA backbone [5,6]. Recently, it has been found that copper(II) complexes of ferrocene(Fc)-conjugated reduced Schiff base of L-tyrosine are cytotoxic in HeLa (human cervical cancer) and MCF-7 (human breast cancer) [7]. 1,10-phenanthroline and its copper (I and II) complexes showed interesting magnetic, electronic and redox process [8].
Schiff base containing amino thiol group (HN2-NHCS-R) and their metal complexes exhibit various biological activities. Thiosemicarbazone containing compounds was first used as drugs against tuberculosis, leprosy [9], small pox [10] and tumour [11]. Recently, Triapine (3-amino pyridine-2-carboxaldehyde thiosemicarba zone) has been developed as an anticancer drug and has completed clinical phase II on several cancer types [12, 13].
Schiff bases have been made by reacting 1,10-phenanthroline-2,9-dicarboxaldehydehemi hydrate with sulfur containing amines and studied their antibacterial properties [14]. However, the synthesis of metal complexes of these Schiff bases has not been studied. We report the synthesis & characterization of an interesting dimeric copper (Cu) complex by using one of these Schiff bases.
2. Materials and Methods
2.1. Materials
The CuCl2 salt and the solvents were commercially available and used without further purification. The Schiff base (2,2’)-dimethyl 2,2’-(1,10-phenanthroline-2,9-diyl) bis(methan-1-yl-1-ylidene)-bis(hydrazinecarbodithioate) (1) was prepared according to a published procedure [15]. Infrared spectra were recorded on a Thermo Nicolet iS10 FTIR spectrometer equipped with ATR accessory. NMR spectra were recorded on a Bruker 400 MHz spectrometer. Diffraction study was done on a Rigaku XtaLab mini bench-top instrument. Data collection and data reduction was done with the software of the instrument, “Crystal Clear” [16]. The radiation source was MoKα (λ = 0.71075 Å).
2.2. Synthesis
Copper (II) chloride (0.0134 g, 0.1 mmol) dissolved in methanol (2 ml) was mixed dropwise with boiling solution of the Schiff base 1 (0.044 g, 0.1 mmol) in hot methanol (20 ml). The mixture was refluxed for 1 h and then left to stand overnight (Scheme 1). This resulted in precipitate of the copper complex 2, which was then filtered off and kept in desiccator over silica gel. Then the precipitate was dissolved in DMSO for recrystallization. After 15 days, black crystals of X-ray quality was formed (% yield = 30%). IR: ν (cm−1). 3100, 2900 (C-H aromatic and aliphatic), 1570 (C=N), 1500, 1600 (C=C, aromatic), 1030 (C=S). 1H-NMR (DMSO-d6, ppm): δ H 13.75 (s, 2 NH), 8.65 (d, J = 8.7 Hz, 2H), 8.30 (d, J = 8.7 Hz, 2H), 8.60 (s, 2H), 8.07 (s, 2H), 2.01 (s, 6H).
2.3. Single Crystal X-ray Analysis
Crystals of the complex 2 were grown from DMSO at room temperature. A crystal of 0.17 × 0.13 × 0.09 mm dimension was mounted on a MiTeGen loop with Apiezon grease at ambient temperature. Diffraction study was done on a Rigaku XtaLab mini bench-top instrument. Data collection and data reduction was done with the software of the instrument “Crystal Clear” [17]. The radiation source was MoKα (λ = 0.71075 Å). The compound crystallized in a triclinic crystal system with P-1 space group. Unit cell dimensions: a = 10.006(3) Å, b = 13.272(3) Å, c = 22.123(6) Å, a = 85.656(6)˚, b = 81.656(6)˚, g = 73.097(5)˚ and volume = 2779.5(13) Å3.
Structure Solution and Refinement: The structure was solved by direct methods [16] and expanded using Fourier techniques. The non-hydrogen atoms were refined anisotropically. Hydrogen atoms were refined using the riding model. The final cycle of full-matrix least-squares refinement on F2 was based on 6595 observed reflections and 607 variable parameters. Which converged with unweighted and weighted agreement factors of: R1 = 0.0823 and wR2 = 0.2222. This was conducted using the program suite WINGX [18]. The compound crystallized as solvates. Disordered DMSO molecules were present in the lattice that could not be completely modeled. One of the DMSO molecules was located close enough to be within the range of hydrogen bonding interaction. The crystal data and structure refinement details are listed in Table 1.
3. Results and Discussion
The coordination sphere around each copper center reveals that copper in the complex was reduced. The ligand in this reaction had only neutral donor atoms as starting material. Hence in order to satisfy the charge neutrality of the complex there needs to be two chloride ions. However, no chloride ion was detected in the structure. This could be rationalized by the fact that thionic sulfur can readily be oxidized to a disulfide form [19,20]. In the process of oxidation of the ligand the copper (II) becomes reduced to copper (I). The C-S bond distance (1.712) of the coordinating sulfur atom in the complex suggests that. this distance is longer that the distance of C=S (1.651)
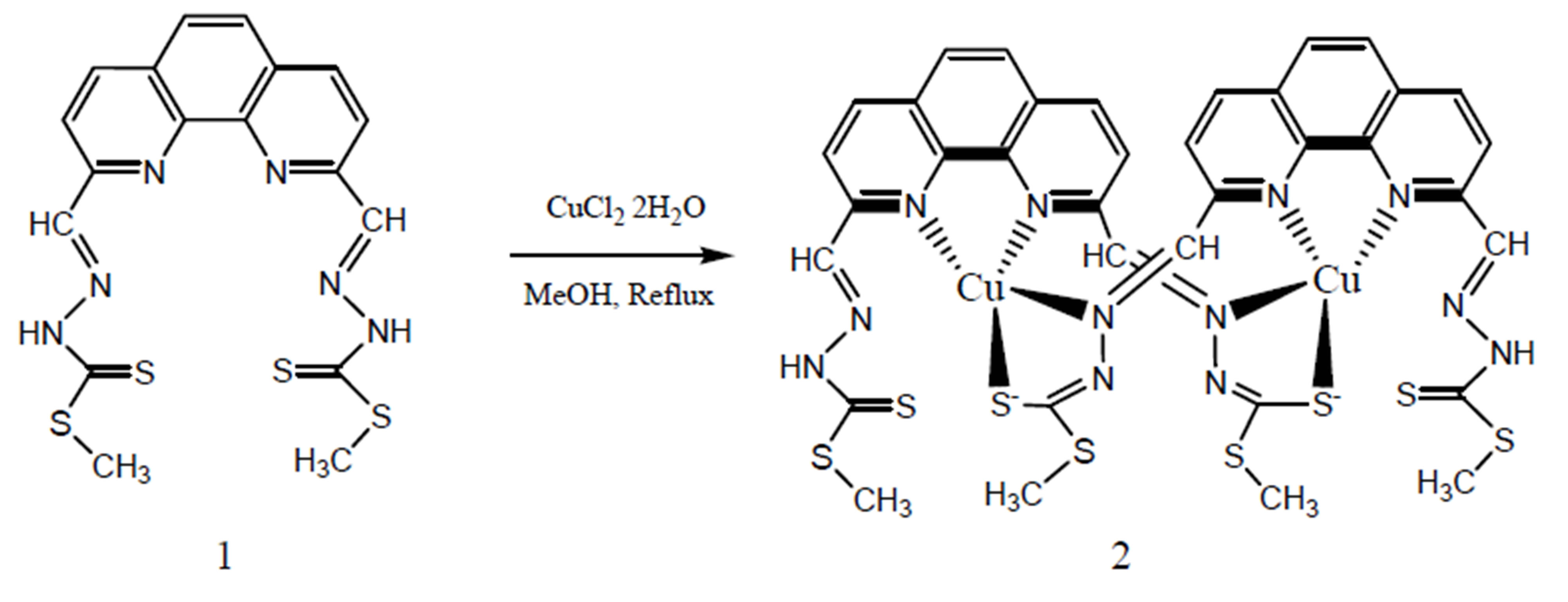
Scheme 1. The synthetic route for the preparation of dimeric copper (II) complex of Schiff base 1.


Table 1. Crystal data and structure refinement for compound 2.
bond in the ligand.
3.1. Bond Lengths and Angles
The ORTEP drawing of the complex is given in Figure 1. Cu atom has distorted tetrahedral coordination with sulfur and nitrogen atoms. The bond angles for standard tetrahedral is 109.5˚ but from the Figure 1 it has been shown that the angle between S(5)-Cu(1)-N(42) is 86.5 (2)˚ whereas the angle between S(5)-Cu(1)-N(38) is 131.66 (19)˚. The other two angles for central Cu(1) atom are 81.8 (3)˚ and 100.3 (3)˚ for angles N(38)- Cu(1)-N(39) and N(39)-Cu(1)-N(42) respectively. However, the Cu(1)-N(38), Cu(1)-N(39), Cu(1)-N(42) and Cu(1)-S(5) distances are 2.038 (6), 2.086 (7), 2.066 (7), and 2.228 (3) Å respectively.
The methyl group attached to S(9) was refined at two positions with site occupancy factors of 0.61 and 0.39 for major and minor conformations, respectively. The methyl group at S(7) atom apparently did not have such freedom probably due to the solvent molecules nested in the lattice around this sulfur atom limiting the free rotation around the single bond. The bond angles and bond distances for other copper center, Cu(2) resembles to that of the first copper center (Table 2). Here overall geometry of the complex is cliplike. The clip appearance is provided by semi parallel phen moiety. Although the geometry is cliplike, there is not much clearance for another molecule to fit in due to steric hindrance (Figure 2).
The average values of bond distances and angles in different atoms are in agreement with the literature values [18]. Selected bond distances and bond angles of molecule 2 are shown in Table 2.
Hydrogen Bonds
The bond distance between hydrogen and oxygen, hydrogen and noted noted X-H (Å) and the possible intramolecular interactions by hydrogen bonds noted X-H (Å) of molecule 2 is shown in Table 3. Tables 4 and 5 provide isotropic displacement parameters and Anisotropic displacement parameters of compound 2. These different hydrogen bonds showed in Figure 3 are responsible of the molecular packing in the unit cell.
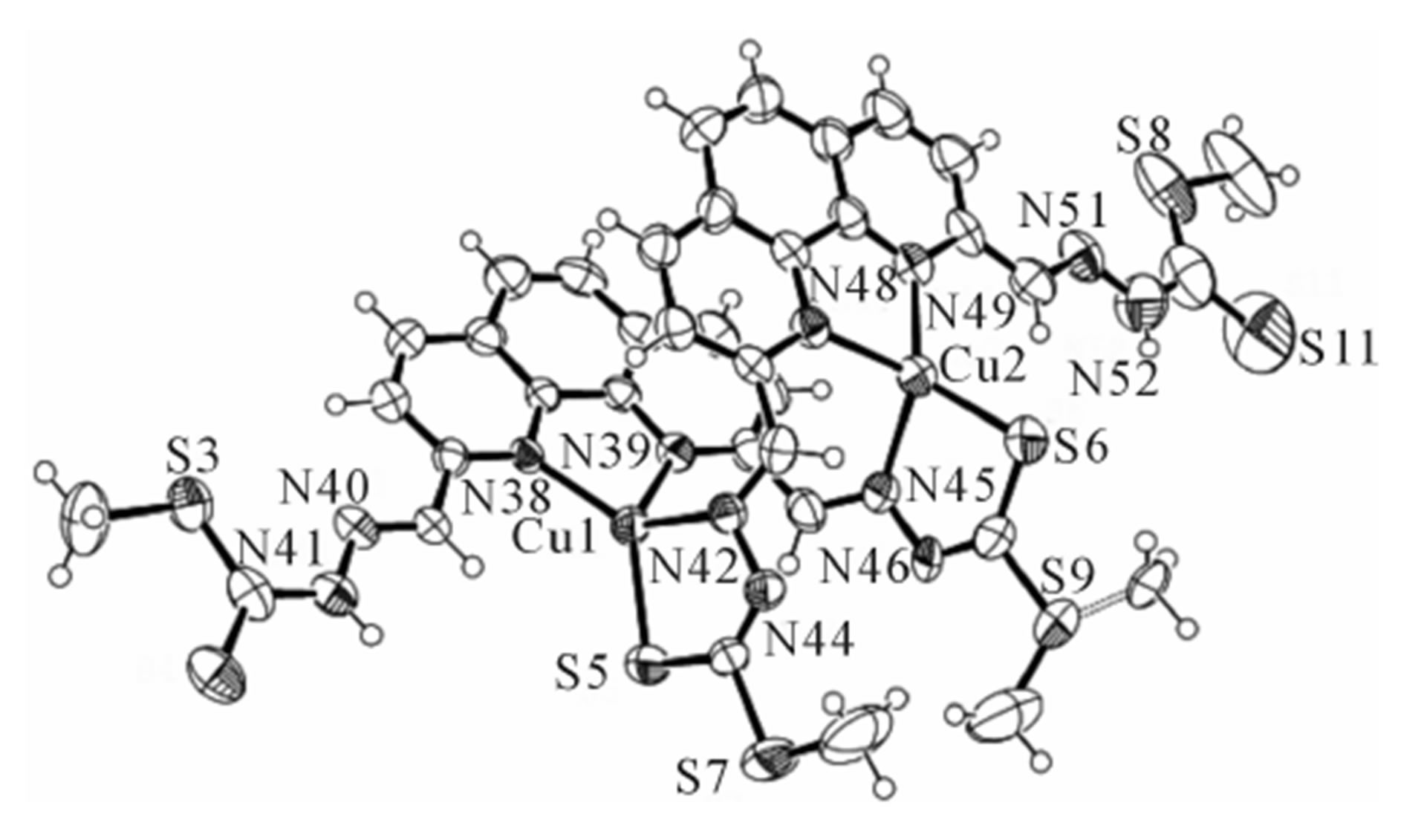
Figure 1. Perspective view of dimer of bis[(2,2’)-dimethyl 2,2’-(1,10-phenanthroline-2,9-diyl)bis(methan-1-yl-1-ylidene)- bis(hydrazinecarbodithioate)] Cu(I) complex showing the labelling scheme.
3.2. Crystal Packing
Hydrogen bonds played crucial roles in the crystal pack ing. Figure 4 shows the presence of two molecules in the unit cell which corresponds to the centrosymmetric triclinic space group P-1 with two general positions (x, y, z) and (−x, −y, −z).
4. Conclusion
The new dimeric coordination complex, bis-[(2,2’)-dimethyl 2,2’-(1,10-Phenanthroline-2,9-diyl) bis(methan-1- yl-1-ylidene)-bis(hydrazinecarbodithioate) copper(I)], was synthesized with a moderate yield and its crystal structure was determined by X-ray diffraction analysis at room temperature. Structural studies reveal the synergistic multicenter coordination by thiosemicarbazide moiety of the Schiff base ligand. The phen moieties of the two ligands are semi-parallel to each other while coordinated to two copper centers. Because of this structural arrangement, the complex looks like a molecular clip. However, the clearance between the two phen moiety is

Figure 2. Space filling model of the compound demonstrating limited clearance for clipping in a molecule.

Figure 3. Hydrogen bonding between oxygen of the DMSO solvent molecule and the hydrogen atom of N-H of the ligand moiety.

Figure 4. The packing diagram (view along a-axis) showing void in the lattice, where disordered solvent molecules are likely to populate.

Table 2. Selected bond angle and bond distances of the compound.
inadequate for the complex to serve as a functional molecular clip.
NOTES