Pregnancy with and without birth trauma modulates the gene expression of proteins involved in elastic fiber homeostasis in the rat vagina ()
1. INTRODUCTION
Pelvic organ prolapse (POP) is a major health and quality of life problem that affects women in their reproductive and menopausal years [ 1 ]. The lifetime risk of having surgery for POP is as high as 19%, while the annual cost to surgically manage POP in the U.S. is estimated to be $1 billion [2,3]. The causes of POP common to most women include pregnancy and parturition, and aging. With respect to the former, increasing vaginal parity, and operative vaginal delivery have been identified as the exposures most associated with higher rates of POP; yet, most women who have born children do not develop POP and when they do so it is well after the associated exposure [ 4-7 ]. Our inability to explicate this discrepancy, in part, relates to our limited understanding of the pathophysiology of POP.
In recent years a significant advancement in our understanding of the pathophysiology of POP came to light when an important physiologic link between elastic fiber homeostasis and POP was observed in two murine knockout (KO) models (Lysyl Oxidase Like-1 [LOXL-1] and Fibulin 5 [FBLN 5]) that develop POP due to abnormalities in elastic fiber formation [8,9]. Elastic fibers consist of two distinct components: elastin and microfibrils. Elastin is an insoluble polymer that consists of crosslinked tropoelastin monomers, while microfibrils are made up of a complex array of macromolecules [10]. LOXL-1 is thought to play a role in the cross-linking of tropoelastin monomers (elastin’s precursor) to create elastin polymers, while FBLN 5 is thought to play a role in the transfer of elastin aggregates to microfibrils, the microfibrils acting as the scaffolding of elastic fibers [ 11 ]. Elastic fibers are found throughout the extracellular matrix of the reproductive tract and endow these tissues with the characteristic of elastic recoil [12]. While elastic fibers in most tissues undergo little change during adult life, those of the reproductive tract differ by undergoing significant remodeling as a result of pregnancy and parturition [ 13 ]. Recent studies have illuminated this process. Drewes and colleagues studied the regulation of elastic fiber synthesis and assembly in the vaginal wall and pubocaudalis tissues of wild type mice. They found a burst in the protein expression of FBLN 5, tropoelastin and desmosine (desmosine is a marker of mature elastic fibers), during the immediate postpartum period. This led them to hypothesize that this burst in elastic fiber synthesis and assembly was critical to the recovery of pelvic floor support after vaginal delivery, and its absence or derangement may play a primary role in the pathogenesis of POP. To test this hypothesis, Drewes and colleagues injected the posterior vaginal wall of wild type mice with elastase immediately postpartum, and found POP developed in the elastase injected mice, but not in saline or inactivated elastase treated animals [ 14 ].
To test the hypothesis posited by Drewes, Rahn and colleagues exposed nulliparous FBLN 5 KO, FBLN 5 heterogeneous, and wild type mice to vaginal distention (to simulate birth trauma) at 6 weeks of age (prior to the spontaneous onset of prolapse in FBLN 5 KO mice) and found that within days of the exposure only the FBLN 5 KOs developed pelvic organ prolapse [ 15 ].
In light of these findings, our group, using a rat model, has examined the effect of pregnancy and mode of delivery on the gene expression of proteins involved in elastic fiber synthesis and assembly.
2. MATERIALS AND METHODS
All chemicals were purchased from Sigma Chemicals (St. Louis, MO, USA) except as noted below. TRIzol reagent, oligo (dT) and the SuperScript II First-Strand Synthesis System for reverse transcriptase-PCR (RT-PCR) were from Invitrogen Corporation (Carlsbad, CA, USA). SYBR® Green PCR Master Mix and 96-well PCR plates/ covers were from Applied Biosystems Inc. (Foster City, CA, USA). Primers were synthesized by Invitrogen Corporation and the sequences for the individual molecular targets are listed in Table 1.
All study protocols were approved by the Animal Institute Committee of the Albert Einstein College of Medicine. The study used 38 female Sprague-Dawley rats. 10 virgin rats underwent simulated vaginal delivery (SiVD) as follows: under anesthesia a 10 French Foley catheter was inserted into the vagina, the Foley balloon inflated with ~2.5 cc of water, and the catheter placed to gravity off the edge of the lab bench for 3 hours, then removed while still inflated. 10 pregnant rats underwent spontaneous vaginal delivery (SpVD). 11 pregnant rats were submitted to cesarean delivery (CD) on gestation day 21 as follows: under anesthesia, a small vertical incision was made in the lower ventral abdominal wall allowing for delivery of the gravid uterine horns. Through longitudinal incisions, rat pups and their placentas were removed; the horns were returned to the abdominal cavity and the ventral incision closed with permanent suture. Experimental animals were sacrificed on day 2 or day 14 after their respective mode of delivery and categorized as shown in Figure 1. 7 virgin rats served as controls.
All rats were killed humanely by placing them in a

Table 1. Summary of target genes and the primer sequences used to amplify them in real-time PCR.

Figure 1. Group allocation based on mode of delivery and timing of postpartum sacrifice *7 virgin rats were used as controls.
carbon dioxide chamber. Immediately after sacrifice, vaginal tissue was harvested and divided into an upper segment (from the mid-vagina to the level of the vaginal fornices) and lower segment (from the mid-vagina to the level of the perineal body), and the tissue flash frozen.
Total RNA was isolated from the frozen vaginal tissue of each rat using TRIzol reagent according to the manufacturer’s protocol. RNA concentration and purity was determined using a ND-1000 spectrophotometer (NanoDrop Technologies, Wilmington, DE, USA). For each sample, 1 µg of RNA was converted to cDNA using RT via the SuperScript II First-Strand Synthesis System according to the manufacturer’s protocol. Primer pairs were designed using published cDNA sequences obtained via Entrez Nucleotide of the National Center for Biotechnology Information and the Primer Express program (Applied Biosystems). Amplicons were deliberately kept between 50 and 100 bp for all primer pairs to enable equal transcriptional efficiency. For RT-PCR, equal amounts of cDNA template and primer pairs were combined with SYBR Green Master Mix to final concentrations of 0.04 - 0.4 ng/µL and 50 - 900 nm, respectively, depending on the molecular target, and real time RT-PCR performed as previously described [ 16 ]. The following targets were amplified using SYBR Green for amplicon detection: LOXL-1, Tropoelastin, FBLN 5. For relative quantification the efficiency of amplification for each individual primer pair was determined using cDNA target and the 2−ΔΔct method [17] in conjunction with the RQ Study Software version 1.2.3 (Applied Biosystems). Gene expression was normalized to expression of the beta actin reference gene.
3. RESULTS
On postpartum day two the relative mRNA expression of all three target genes in upper and lower vaginal tissue was found to be up-regulated when compared to controls (see Figures 2 and 3). By postpartum day fourteen the relative mRNA expression of the target genes in lower vaginal tissue had uniformly decreased from their day two levels (see Figure 3). In upper vaginal tissue this trend remained true for the SiVD cohort. However, in both pregnancy cohorts (SpVD and CD), the relative mRNA expression of our target genes on day fourteen remained similar to day two expression levels, and in the case of LOXL-1 and FBLN 5, in the CD cohort, actually increased (see Figure 2). Finally, though our experimental methods were not designed to detect statistical difference between tissue origin, we observed that the relative mRNA expression of our target genes in the SiVD cohort was notably more up-regulated in lower vaginal tissue compared to upper vaginal tissue at postpartum day two (see Figures 2 and 3).
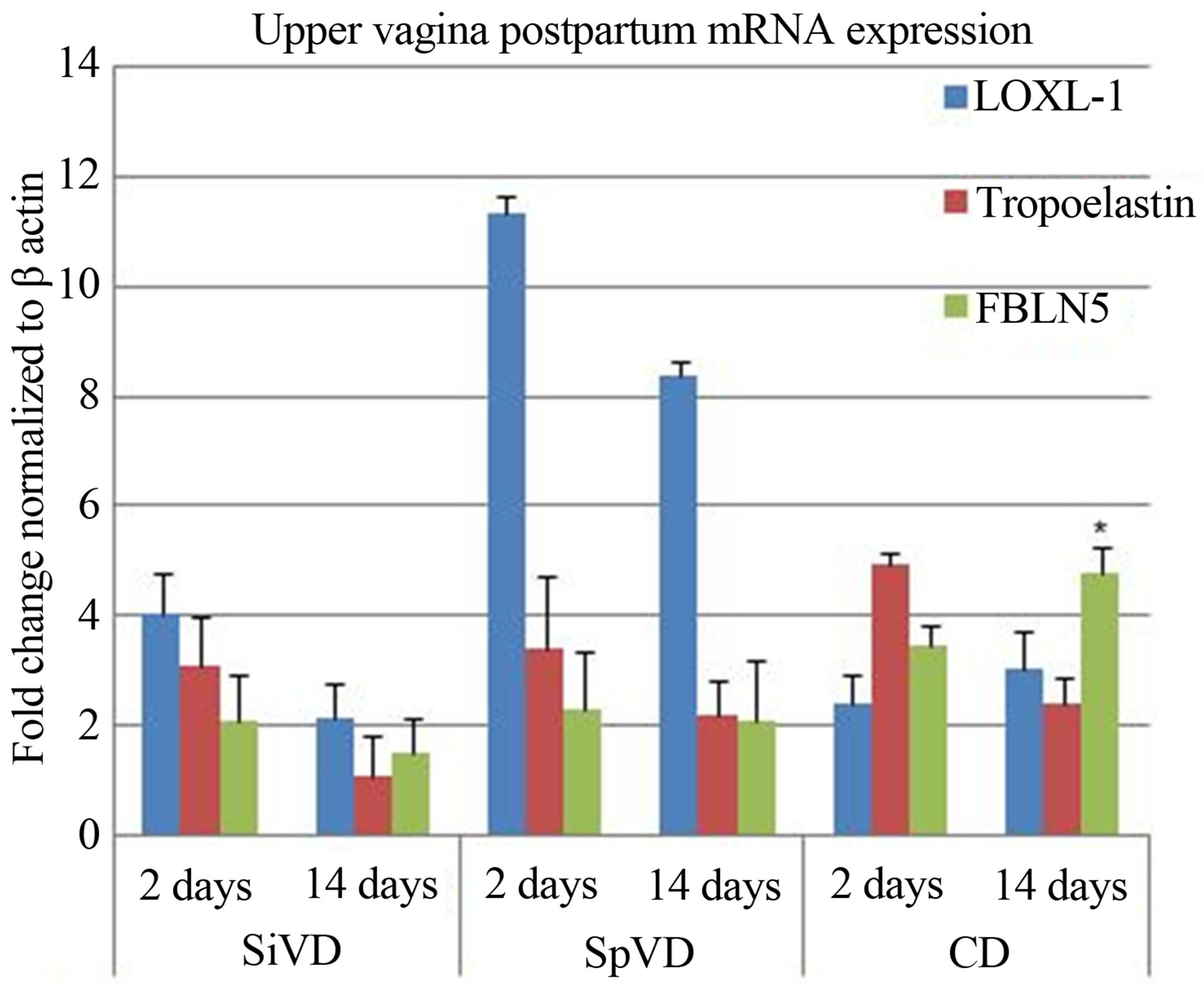
Figure 2. Bar graph representing the relative mRNA expression level of LOXL-1, Tropoelastin and FBLN5 at 2 days and 14 days after SiVD (Simulated Vaginal Delivery), SpVD (Spontaneous Vaginal Delivery) and CD (Cesarean Delivery) in upper vaginal tissue. The error bar represents the standard error measure and the asterisk the statistical significance at P < 0.05 compared to control (Student’s t-test). Results are normalized to β actin and set to a value of 1 for control.

Figure 3. Bar graph representing the relative mRNA expression level of LOXL-1, Tropoelastin and FBLN5 at 2 days and 14 days after SiVD (Simulated Vaginal Delivery), SpVD (Spontaneous Vaginal Delivery) and CD (Cesarean Delivery) in lower vaginal tissue. The error bar represents the standard error measure and the asterisk the statistical significance at P < 0.05 compared to control (Student’s t-test). Results are normalized to β actin and set to a value of 1 for control.
4. DISCUSSION
Our understanding of the pathophysiology of POP and progress in its management has developed slowly over time [18]. Over the last decade, a number of studies have drawn an association between vaginal parity and pelvic floor disorders (PFDs) including POP [5-7,19,20]. These studies and others have caused some leaders in the field to address the question of whether there is a role in twenty-first century obstetrics for elective cesarean delivery for the primary purpose of preventing PFDs [21]. With the cesarean delivery rate over 30% in the United States, this might seem a reasonable option for patients and physicians, particularly in women who are older and desire low parity [ 22 ]. Yet, to the contrary, pregnancy alone has been found to be associated with changes in pelvic floor support and function, and cesarean delivery (even before labor) is not completely protective of PFDs [ 23-25 ]. Further, with advancing age, the rates of some PFDs in nulliparous women rival that of their parous counterparts [ 26 ].
One of the challenges to better understanding the relationship between pregnancy, mode of delivery and PFDs has been the lack of a suitable animal model. Non-human primates have been used to study pelvic floor disorders because of their similarity to humans in pelvic anatomy, the 28-day menstrual cycle, manner of parturition, and the onset of POP. In addition, non-human primates share two of the evolutionary factors associated with PFDs: semi-upright posture and giving birth to infants with large cranial capacity. Nevertheless, non-human primates (e.g., squirrel monkeys), have their limitations such as the inability to take instruction, report symptoms, or inform intentions. Most notably, they have a greater associated cost when compared to the use of murine models, resulting in limited use in published studies [ 27 ].
Although quadrupeds, the development of KO mice to LOXL-1 and FBLN 5 with their abnormalities in elastic fiber homeostasis have become important animal models to aid the study of the complex relationship between pregnancy, parturition, aging and POP. In addition to KO mice, the application of vaginal distention in murine models to mimic the effects of birth trauma on pelvic floor tissues has provided another valuable research tool. Use of these techniques has been best exemplified in the work of Drewes et al. and Rahn et al. [14,15]. Their results have affirmed the link between elastic fibers, vaginal delivery and POP in a murine model. However, there has only been one report analyzing the effect of cesarean delivery compared to vaginal delivery on elastic fiber homeostasis in pelvic floor tissues in a murine model [ 28 ]. In that study by Gustilo-Ashby et al., LOXL-1 KO mice were submitted to either spontaneous or cesarean delivery and had POP assessed at 12 weeks postpartum. They found 59% of LOXL-1 KO mice after spontaneous vaginal delivery and 31% after cesarean delivery developed POP, with those in the vaginal delivery group developing POP approximately three weeks earlier than those in the cesarean delivery group.
In our experiment we determined the relative mRNA expression (using real time RT PCR) of proteins responsible for elastic fiber synthesis and assembly in the early (day two) and late (day fourteen) postpartum period from vaginal tissue taken from Sprague Dawley rats that had been exposed to different modes of delivery. We observed an up-regulation in the relative mRNA expression of LOXL-1, FBLN 5 and Tropoelastin in all groups at postpartum day two. In lower vaginal tissue the up-regulation decreased uniformly from postpartum day two to postpartum day fourteen. However, the relative mRNA expression of the target genes in upper vaginal tissue persisted at postpartum day fourteen in the SpVD and CD cohort. This finding may reflect the influence of pregnancy on tissues of mullerian origin (as opposed to the urogenital sinus derived tissues of the lower vagina) and the postpartum involution process that is unrelated to tissue trauma. In addition, we observed little change in the relative mRNA expression of the target genes in upper vaginal tissue in the SiVD cohort. This may be a function of our experimental model, in which the foley balloon, due to its size and shape, distends and damages tissues of the lower vagina but spares the upper vault. We hope to clarify this finding by determining the extent of elastic fiber damage in the upper and lower vaginal vault after balloon distention by histologic examination.
Finally, to better understand the effect of mode of delivery on elastic fiber homeostasis in our model, we will need to analyze the enzyme activity of elastolytic enzymes such as matrix metalloproteinase 2 and 9 (MMP 2 and MMP 9). These elastolytic enzymes are found in the extracellular matrix of the reproductive tract and have been shown to be affected by pregnancy and parturition [ 29 ]. In an elegant experiment using a double KO mouse to FBLN 5 and MMP-9, Budatha et al., showed that the development of mouse POP not only required aberrant elastic fibers, but also the up-regulation of MMP-9 [ 30 ]. Thus, the effect of mode of delivery on MMP expression could have significant bearing on the development of POP.
5. CONCLUSION
In this study we found that vaginal trauma, whether by balloon distention or vaginal delivery results in the upregulation of genes involved in elastic fiber synthesis and assembly. Further, we found that pregnancy, in the absence of vaginal trauma (i.e., cesarean delivery), also resulted in the up-regulation of these genes. In total, these findings leave open the possibility that alterations in elastic fiber homeostasis occurring during pregnancy, regardless of mode of delivery, may play a role in the pathogenesis of POP and other pelvic floor disorders.
NOTES