1. Introduction
In the synthesis of an optically active compound utilizing a chiral auxiliary, cleaving the chiral auxiliary with concomitant reduction of the chiral center is an important step. Therefore, many methodologies for the cleavage of the chiral auxiliary such as the use of 2-oxazolidinone have been reported. The typical method for N-acyl-2- oxazolidinone cleavage is the treatment with alkoxide or lithium hydroperoxide to convert the substrate into a benzyl ester [1-3], a methyl ester [4], or a carboxylic acid [5-7], respectively (route A, Scheme 1). It is also wellknown that a reductive cleavage reaction using a reductant such as LiAlH4 [8-14] or LiBH4 [15] gives the corresponding alcohol (route B). On the other hand, only few reports of Lewis acid-promoted cleavage reactions of N-acyl-2-oxazolidinones to esters [16] or hydroxamic acid derivatives [17-19] have been published (route C).
We have reported that hydantoin is conveniently derived from an amino acid amide without racemization [20] and can be used as a chiral auxiliary to create a chiral center at the β-position of an acyl group utilizing a conjugate addition system [21]. In order to develop hydantoin as a more useful tool as a chiral auxiliary, a new cleavage reaction of 1-acylhydantoin is required (Scheme 2). We report here that treatment of 1-acylhydantoin with an amine at an elevated temperature provides the corresponding amide and hydantoin in high yields. The use of a catalytic amount of diethylaluminum chloride accelerated the reaction rate.
2. Results and Discussion
The initial examinations began with treatment of N-(3- phenylpropionyl) hydantoin 1 with propylamine a in several solvents. As shown in Table 1, the cleavage reaction of 1 with 1.2 - 3.0 equimolar amounts of a in several solvents from 0˚C to room temperature proceeded to give the corresponding amide 2a and hydantoin 3 in moderate yields (Entries 1 - 3). The cleavage reaction in THF performed at elevated temperature resulted in improvement of the yields of 2a and 3 (Entry 4). Since propylamine has a low boiling point (<50˚C), the cleavage reaction using an equimolar amount of propylamine gave 2a in lower yield (Entry 5). In the case of benzylamine instead of n-propylamine, the yields of 2b and 3 were improved (Entry 6). Lewis acids are known to be effective additives in the cleavage reaction of N-acyl-2-oxazolidinone [7]. Therefore, we expected that the addition of a Lewis acid would also be effective for enhancing the present cleavage reaction. The cleavage reaction in the presence of 1.2 equimolar amounts of Et2AlCl was complete in only 1 h (Entry 7). Furthermore, a catalytic amount of Et2AlCl was enough to complete the cleavage reaction, and 2b and 3 were obtained in excellent yields (Entry 8).
In the cleavage reaction of N-acyl-2-oxazolidinone, Narasaka and co-workers reported its tolerance to the attack of primary amines and that the addition of a Cp2ZrHCl (Cp = cyclopentadienyl) catalyst was necessary for the amide conversion [22].
The cleavage reaction using primary amines or aniline also gave 2 and 3 in excellent yields in the presence of
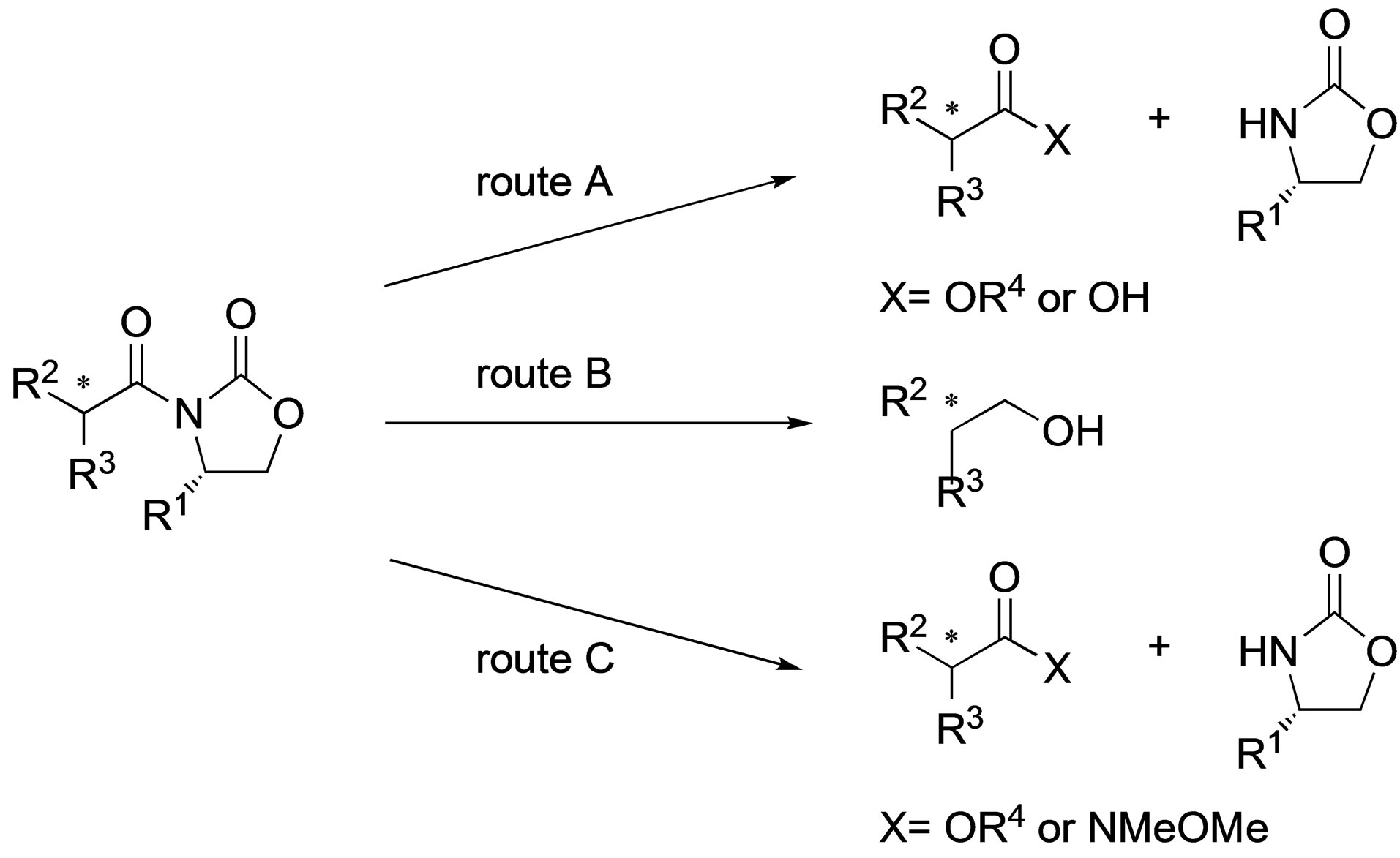
Scheme 1. Conventional cleavage reactions of N-acyl-2-oxazolidinone.


Table 1. Cleavage reaction of 1 with propylamine or benzylamine.
diethylaluminum chloride (Table 2, Entries 1 - 4). In contrast, the reactivity using secondary amines depended on the steric hindrance of the amine (Entries 5 - 8). Thus, the cleavage reaction with the more sterically demanding diisopropylamine failed, whereas that with diethylamine succeeded (Entries 5 and 6). Among secondary amines, cyclic amines such as pyrrolidine or 1,2,3,4-tetrahydroisoquinoline resulted in better yields than acyclic seconddary amines (Entries 7 and 8).
The cleavage reaction of N-(3-phenylpropionyl)-2- oxazolidinone 4 under similar reaction conditions was also examined (Scheme 3). Treatment of 4 with 1.2 equimolar amounts of benzylamine gave 2b in 71% yield. The reaction using hydantoin affords better results than that with 2-oxazolidinone.
3. Experimental
Typical Procedure for Cleavage Reaction of 1: Propylamine and a hexane solution of Et2AlCl were added to a THF solution of 1-acylhydantoin under Ar. After reflux, the reaction was quenched by addition of 1M HCl solution. The organic materials were extracted with CH2Cl2. The extract was washed with saturated NaCl solution and dried over Na2SO4. After removal of solvent under reduced pressure, the residue was separated by preparative TLC (hexane:EtOAc = 1:1) to give the amide and hydantoin.


Table 2. Cleavage reactions of 1 with other amines.

Scheme 3.Conversion of N-dihydrocinnamoyl-2-oxazolidinone to amide.
N-Propyl-3-phenylpropionamide: 1H NMR (300 MHz, CDCl3) δ = 0.85 (3H, t, J = 7.3 Hz), 1.46 (2H, dq, J = 7.3 and 7.3 Hz), 2.46 (2H, t, J = 6.8 Hz), 2.96 (2H, t, J = 6.8 Hz), 3.16 (2H, q, J = 7.3 Hz), 5.48 (1H, br s), 7.18 - 7.38 (5H, m).
N-Benzyl-3-phenylpropionamide: 1H NMR (300 MHz, CDCl3) δ = 2.51 (2H, t, J = 7.3 Hz), 2.99 (2H, t, J = 7.3 Hz), 4.39 (2H, d, J = 5.7 Hz), 5.64 (1H, br s), 7.10 - 7.30 (5H, m).
N-Butyl-3-phenylpropionamide: 1H NMR (300 MHz, CDCl3) δ = 0.89 (3H, t, J = 7.1 Hz), 1.25 (2H, sext, J = 7.1 Hz), 1.40 (2H, t, J = 7.1 and 5.9 Hz), 2.46 (2H, t, J = 7.8 Hz), 2.97 (2H, t, J = 7.8 Hz), 3.21 (2H, q, J = 5.9 Hz), 5.34 (1H, br s), 7.10 - 7.35 (5H, m).
N-Phenethyl-3-phenylpropionamide: 1H NMR (300 MHz, CDCl3) δ = 2.42 (2H, t, J = 7.3 Hz), 2.73 (2H, t, J = 7.1 Hz), 2.95 (82H, t, J = 7.3 Hz), 3.48 (2H, q, J = 7.1 Hz), 5.34 (1H, br s), 7.00 - 7.35 (10H, m).
N-Cyclohexyl-3-phenylpropionamide: 1H NMR (300 MHz, CDCl3) δ = 0.85 - 1.20 (3H, m), 1.25-1.45 (2H, m), 1.55-1.70 (3H, m), 1.80 - 1.85 (2H, m), 2.43 (2H, t, J = 7.3 Hz), 2.96 (2H, t, J = 7.3 Hz), 3.65 - 3.80 (1H, m), 5.16 (1H, br s), 7.15 - 7.35 (5H, m).
N-Phenyl-3-phenylpropionamide: 1H NMR (300 MHz, CDCl3) δ = 2.64 (2H, t, J = 7.6 Hz), 3.04 (2H, t, J = 7.6 Hz), 7.06 (1H, t, J = 7.8 Hz), 7.10-7.35 (8H, m), 7.43 (2H, d, J = 7.8 Hz).
N-(3-Phenylpropionyl)pyrrolidine: 1H NMR (300 MHz, CDCl3) δ = 1.65 - 1.90 (4H, m), 2.56 (2H, t, J = 7.6 Hz), 2.99 (2H, t, J = 7.6 Hz), 3.29 (2H, t, J = 6.6 Hz), 3.47 (2H, t, J = 6.6 Hz), 7.10 - 7.35 (5H, m).
N,N-Diethyl-3-phenylpropionamide: 1H NMR (300 MHz, CDCl3) δ = 1.10 (3H, t, J = 7.1 Hz), 1.11 (3H, t, J = 7.1 Hz), 2.60 (2H, t, J = 7.6 Hz), 2.99 (2H, t, J = 7.6 Hz), 3.23 (2H, q, J = 7.1 Hz), 3.38 (2H, q, J = 7.1 Hz), 7.15-7.35 (5H, m).
N-(3-Phenylpropionyl)-1,2,3,4-tetrahydroisoquinoline: A mixture of rotamers. 1H NMR (300 MHz, CDCl3) δ = 2.65 - 2.75 (2H, m), 2.80 - 2.90 (2H, m), 2.95 - 3.05 (2H, m), 3.60 and 3.84 (2H, t, J = 5.9 and 5.9 Hz), 5.54 and 4.75 (2H, s and s), 7.00 - 7.35 (8H, m).
4. Conclusion
We have developed the convenient cleavage reaction of 1-acylhydantoin to the corresponding amide. The conversion is successfully performed under mild conditions with a catalytic amount of Et2AlCl. The activation of the imide moiety accelerates the formation of the amide. Other methodologies for new and convenient conversions are now in progress.
NOTES