1. Introduction
Compounds with a naphthalene moiety are biologically active and are found to show antimicrobial activities [3, 4], anti-inflammatory effect [5], and some naphthalene derivatives are useful as antipsychotic drugs [6] and enzyme inhibitors [1]. The presence of benzofurans in the heterocyclic compounds is worth mentioning and some of them are potentially bioactive and such effects include anti cancer, anti HIV and antimicrobial activities [2,7-16].
Although the synthesis of bioactive heterocycles by metal-annulated cyclization [17-23] procedure is well known in the literature, iodonium assisted heterocyclization is also an efficient synthetic route. The iodinated organic molecules may act as useful intermediates as the iodine can be replaced by other nucleophiles. This is an attractive alternative to organometallic chemistry.
To our knowledge, there is no report of doubly fused [5,6] heterocyclic compounds from the corresponding diallyl-dihydroxy naphthalene precursors involving Niodosuccinimide as the reagent and also by molecular iodinemediated 5-exo-trig cyclization in aqueous micelle using surfactant. The efficacy of the reagent N-iodosuccinimide and surfactant-catalyzed iodine-mediated cyclization in aqueous media prompted us to undertake a comparative study to develop a useful strategy leading to doubly fused benzofuran ring systems. Herein, we report our results.
2. Results and Discussion
The required precursors 3a, 3c and 3e for this purpose were prepared according to the standard published literature procedures [24,25] from the corresponding 2,3- dihydroxy naphthalene (1a), 2,7-dihydroxy naphthalene (1b) and 1,5-dihydroxy naphthalene (1c) respectively by allylation followed by Claisen rearrangement. The other precursors (3b, 3d and 3f) were also accessed employing a similar protocol and characterised by spectral analyses.
Although there are reports [26-29] in the literature for the synthesis of a variety of heterocyclic compounds using N-iodosuccinimide, none reported the synthesis of bis fused naphthodifurans by this methodology. We, therefore, initiated our investigation with the reaction of 1,4-diallylnaphthalene-2,3-diol (3a) and N-iodosuccinimide in acetonitrile solvent at 0˚C - 5˚C.
The substrate 3a was subjected to react under standard reaction condition with N-iodosuccinimide in acetonitrile solvent at 0˚C - 5˚C for 1h and then at room temperature for 3 h. A solid was isolated in 75% yield. Its spectral data indicated that this could be tentatively assigned the naphthodifuran structure 4a. The 1H-NMR of 4a of showed two signals at δ 3.37 and 3.68 as two doublet of doublets with coupling constants 15.6 and 9.6 Hz due to diastereotropic methylene protons of the dihydrofuran rings. The other four methyene protons directly attached to CH2I appeared as multiplets at δ 3.39 - 3.42 (2H) and δ 3.54 - 3.60 (2H). The two—CH-protons of the dihydrofuran rings directly attached to CH2I appeared as a multiplate at δ 5.12 - 5.18.The reaction under the same condition was extended to other substrates 3(b-f) to give the products 4(b-f) in 72% - 80% yields (Schemes 1-3).
From the mechanistic point of view two alternative structures are possible for the products of iodocyclization, either the structure 4 by 5-exo-trig or 6 by 6-endo-trig mode. To unambiguously establish the structure of the products we have subjected the product 4a to dehydroiodination condition by treating the product 4a in ethanolic KOH under reflux to afford the product 5a. The 1H NMR of the product 5a is different from that of the cyclized product 4a and much more simplified in 5a showing a singlet at δ 2.56 for two methyl protons of newly formed two methyls by dehydroiodination followed by isomerisation of the newly generated exocyclic double bonds of the two furan rings (Scheme 1). If the product of cyclization of the substrate 3a were 6a then that after dehydroiodination would have afforded 7a. The formation of 5a from 4a, therefore, definitely establishes the structure of the product of iodocyclization of the substrate 3a as naphthodifuran 4a. This was further supported by the failure of the product 4b to undergo dehydroiodination under the aforesaid reaction condition as there is no scope for dehydroiodination in case of 4b. If the structure of the product of iodocyclization was 6b that should have afforded 7b.
The dehydroiodination of the other products 4c,d and 4e,f also afforded the naphthodifurans 5c,d and 5e,f showing the formation of these products 4c,d and 4e,f from the substrates 3c,d and 3e,f exclusively by a 5-exotrig mode of cyclization in preference to 6-endo-trig mode of cyclization (Schemes 2 and 3).
There are only few reports of the iodine-mediated heterocyclization in aqueous media [30,31] and also in organic media using additives [32,33] such as Envirocat EPZ-10R or SnCl4 [34-36]. The limitation of the traditional methods may be overcome by using surfactants as cosolvents. Surfactants contain both hydrophobic groups (their lypophilic tails called micelles) and hydrophobic groups (their heads). A large volume of literature is available on the use of surfactants as cosolvents for carrying out a variety of reactions in aqueous media [37-44]. The surfactants (cationic, anionic and neutral) form micelles in protic sovents especially in water. The micelles act as a micro reactor to increase the reaction rates. Selforganized aggregates, such as aqueous micellar medium bind the insoluable organic substrates accommodating their hydrophobic tail in the mecellar interior and placing their polar head at the water micelle interface. This changes the physical properties, quantum efficiency and the reactivity of the substrates thus accelerates many organic reactions in aqueous micellar media. Recently, Majumdar and co-workers reported [45] the synthesis of angularly fused furano pyrone/coumarin and quinolone derivatives by using surfactants in water and this is the first report described in literature. We, therefore, turned our attention to investigate the electrophilic iodocyclization of the precursors 3(a-f) by molecular iodine-mediated reaction in the presence of CTAB as surfactant to evaluate application of this methodology. Our initial attempt to obtain
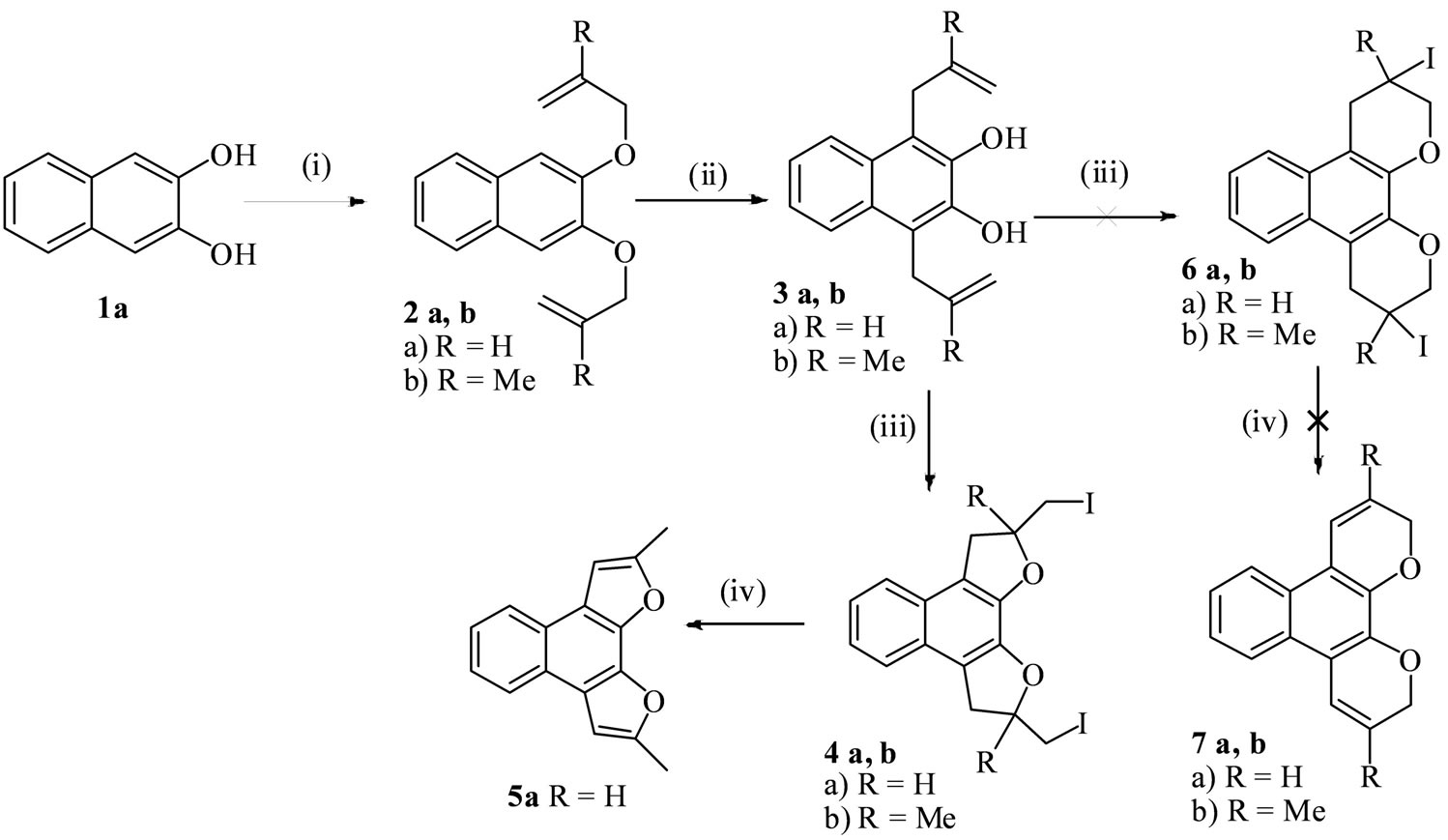
Scheme 1. Reagents and conditions: (i) Allyl bromide or 3-chloro-2-methyl-1-propene, acetone, K2CO3, NaI, reflux, 12 - 15 h; (ii) o-DCB, reflux, 8 - 10 h; (iii) Condition A: substrates 3a,b (1.0 mmol), NIS (2.0 mmol), MeCN, 0˚C-r.t, 4 h; Condition B: substrates 3a,b (0.5 mmol), I2 (1.5 equiv.), CTAB (0.30 mmol), H2O (10 ml.), r.t, 8 h; (iv) KOH (2.0 mmol), EtOH, reflux, 2 - 3 h.
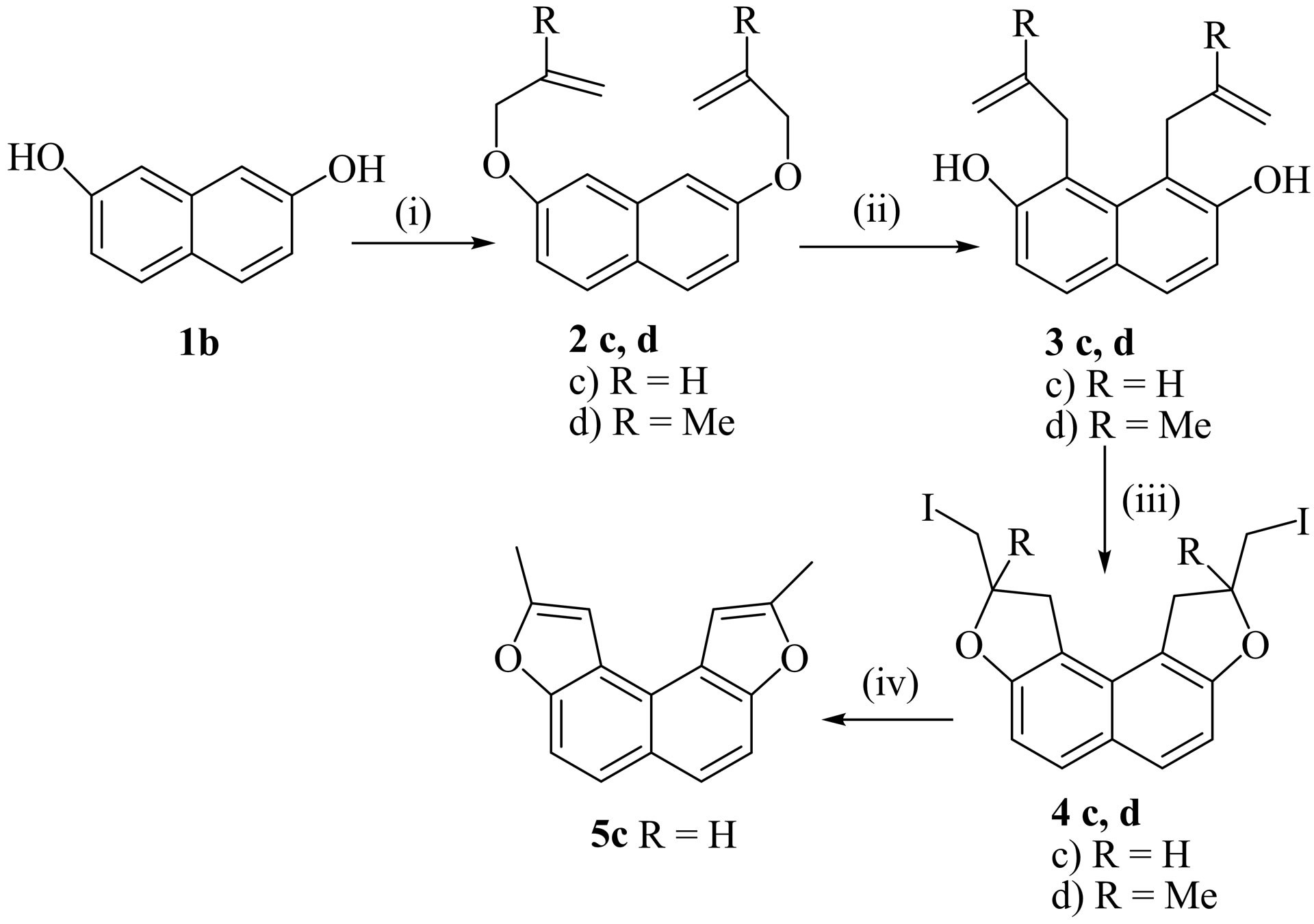
Scheme 2. Reagents and conditions: (i) Allyl bromide or 3-chloro-2-methyl-1-propene, acetone, K2CO3, NaI, reflux, 12 - 15 h; (ii) o-DCB, reflux, 8 - 10 h; (iii) Condition A: substrates 3c,d (1.0 mmol), NIS (2.0 mmol), MeCN, 0˚C-r.t, 4 h; Condition B: substrates 3c,d (0.5 mmol), I2 (1.5 equiv.), CTAB (0.30 mmol), H2O (10 ml.), r.t, 8 h; (iv) KOH (2.0 mmol), EtOH, reflux, 2 - 3 h.
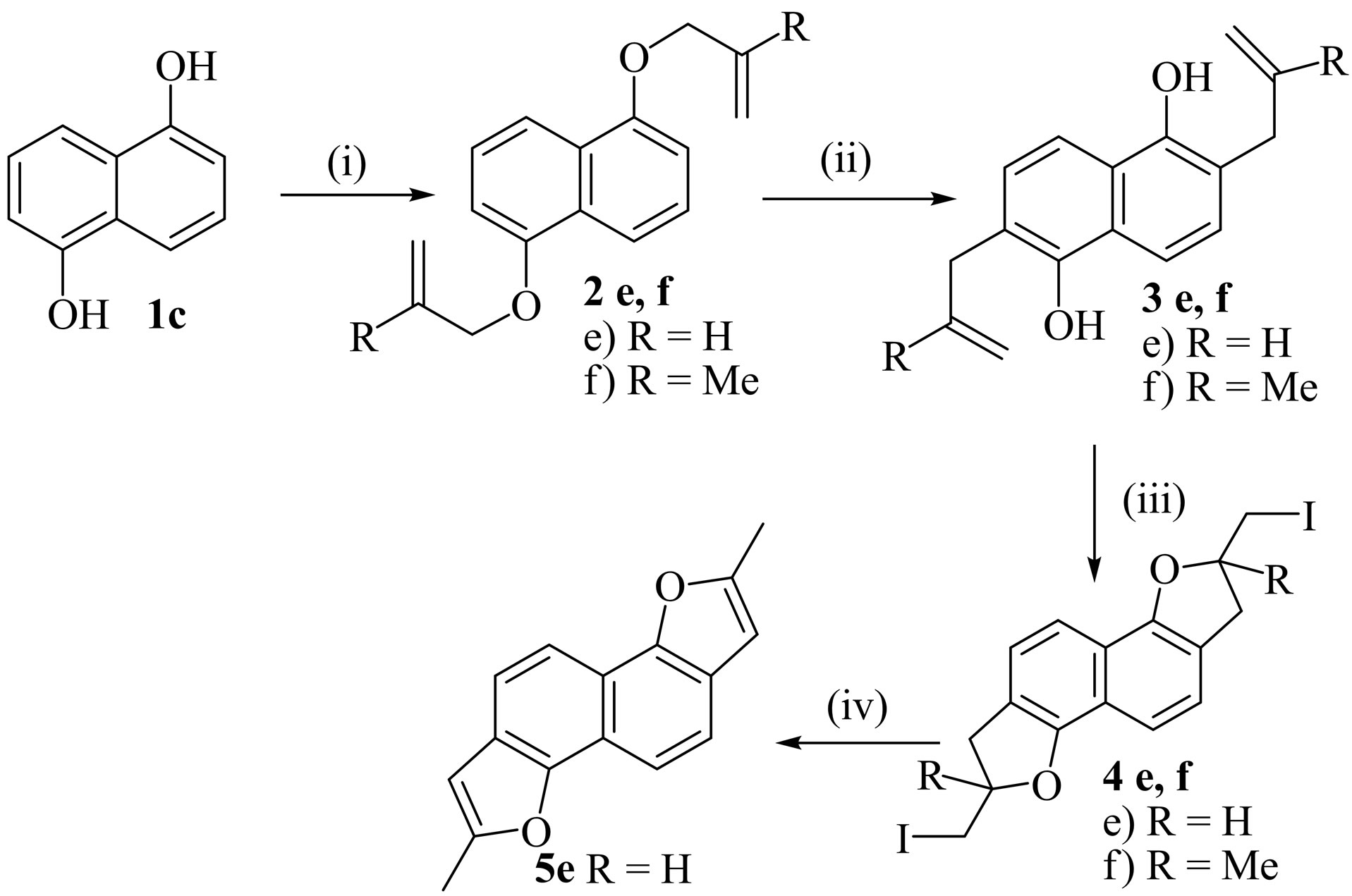
Scheme 3.Reagents and conditions: (i) Allyl bromide or 3-chloro-2-methyl-1-propene, acetone, K2CO3, NaI, reflux, 12 - 15 h; (ii) o-DCB, reflux, 8 - 10 h; (iii) Condition A: substrates 3e,f (1.0 mmol), NIS (2.0 mmol), MeCN, 0˚C-r.t, 4 h; Condition B: substrates 3e,f (0.5 mmol), I2 (1.5 equiv.), CTAB (0.30 mmol), H2O (10 ml.), r.t, 8 h; (iv) KOH (2.0 mmol), EtOH, reflux, 2 - 3 h.
cyclized product in aqueous medium failed by using even upto 1.5 equivalents of molecular iodine for 24 h at room temperature. One of the problems encountered in such a reaction in aqueous media was the insolubility of the di-allyldihydroxy naphthalene precursors in water.
From the green chemistry point of view of water is the best solvent of choice as it is environment friendly and also very easily available. It is found that the reactivity of organic reaction in water media can be greatly enhanced by using surfactants [46,47] which actually forms micelle and thus acts as a phase transfer catalyst in water medium. With these facts in our mind we have decided to carry out the iodocyclization of a series of di-allyldihydroxy naphthalene precursors 3a-f in the presence of the surfactant CTAB. So, the precursor 1,4-diallylnaphthalene-2,3-diol 3a (0.5 mmol) was treated with surfacetant CTAB (0.30 mmol) and molecular iodine (1.5 equivalent) in water (10 ml) for 8 h at room temperature. The aforesaid reaction condition (optimized condition) was arrived at by gradually varying either the concentration of iodine or surfactant and also by altering the reaction time (Table 1).
It is important to note that the same spot of 4a (Rf 0.45 in petroleum ether: ethyl acetate 9:1) in TLC was formed by molecular iodine-mediated cyclization in the presence of surfactant CTAB in aqueous medium and as also in the N-iodosuccinimide-mediated reaction. After usual work up and silica gel column chromatrography a solid product was obtained. This was identified as 4a by comparison of spectroscopic data, melting point, mixed melting point and super imposable IR spectroscopy with the product 4a obtained by the other method. The reaction was then performed with other precursors (3b-f) to give the products (4b-f) (Schemes 1-3).
The comparative results of two methodologies are summarized in Table 2.
The mechanism for the N-iodosuccinimide mediated cyclization and molecular iodine-mediated cyclization in water in the presence of CTAB as surfactant is somewhat similar and follows the electrophilic cyclization pathway.
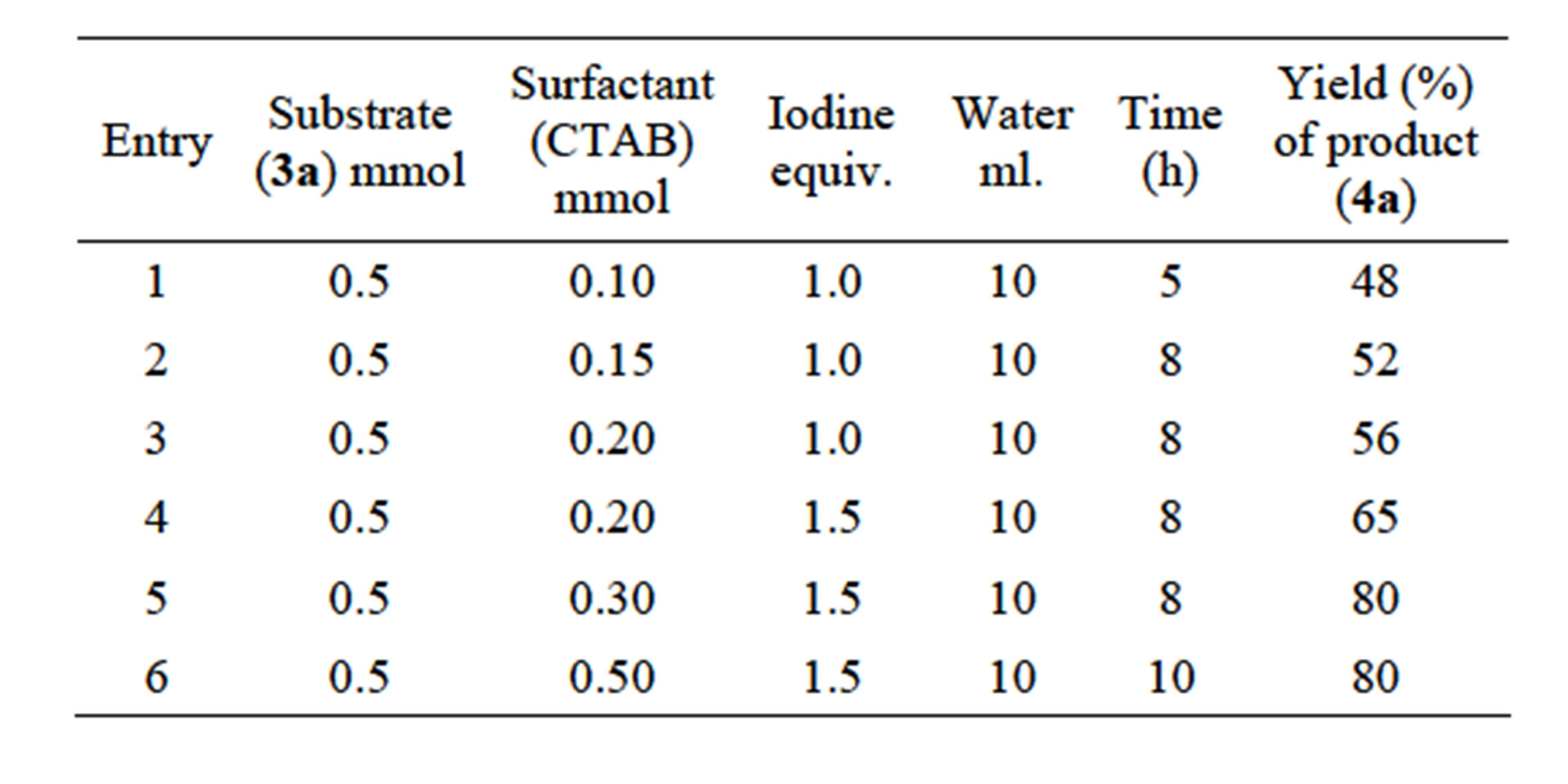
Table 1. Optimized reaction conditions for molecular iodine-mediated cyclization of 3a in aqueous micelle using CTAB as surfactant.
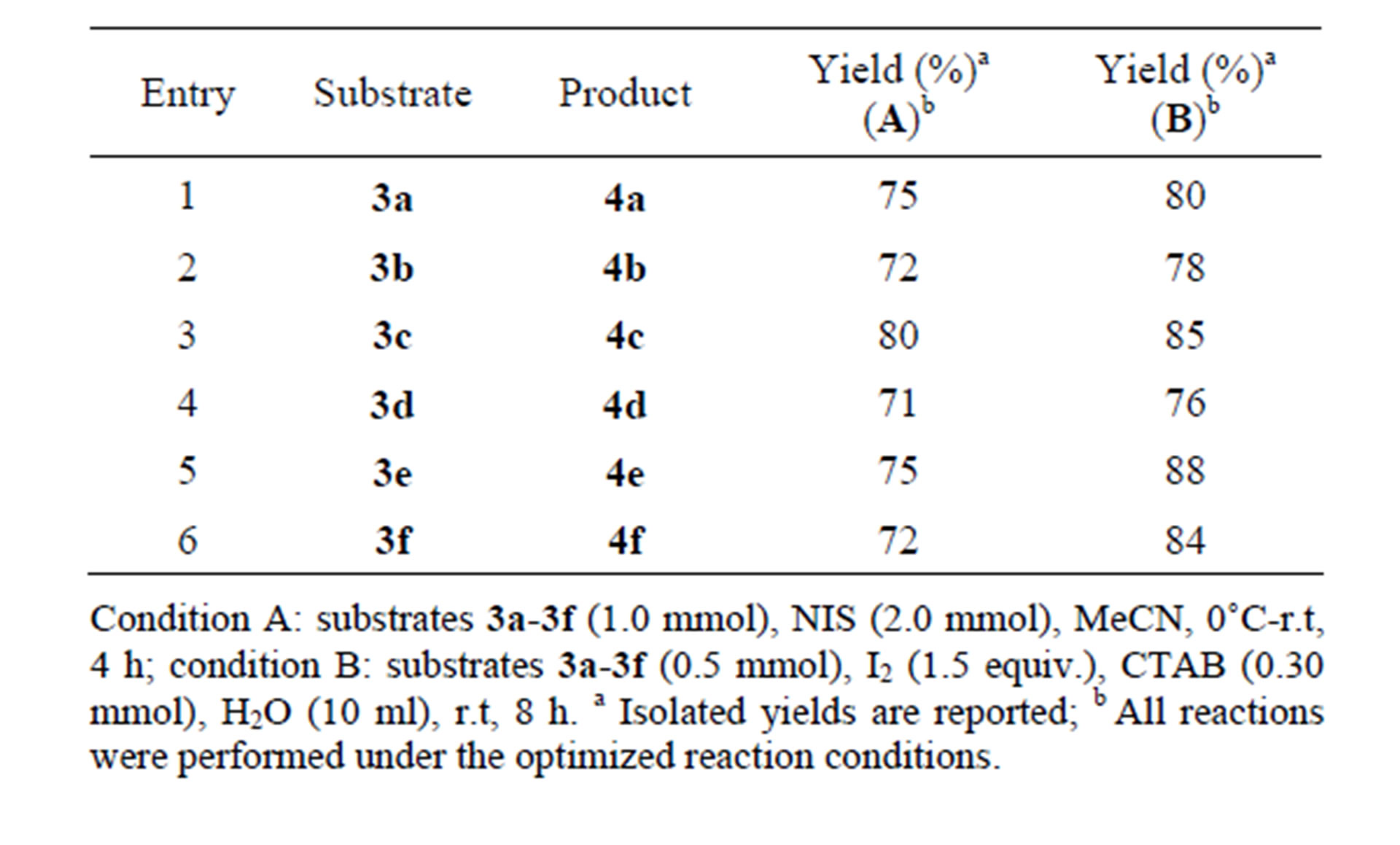
Table 2. Summarized results of the cyclization strategy.
The reaction of solid reagent in water takes place on the surface of water where collision of reacting partners is restricted. However, in the presence of a surfactant e.g. CTAB, the surface tension of water is lowered and allow easier spreading and lowering of interfacial tension between the two reacting partners. Surfactants are usually organic molecules containing both hydrophobic group (lipophilic tails called micelle) and hydrophilic groups (heads), are known to facilitate a number of reactions in aqueous medium. A micelle in a surfactant sticks to the reactant and due to its heavy head settles down into water from the surface and thus increases the collision between the two reacting partners in the intramolecular cyclization and results in more favourable conditions for the reaction compared to reaction on water surface [46,47]. At first the di-allyldi-hydroxy naphthalene precursor 3a may generate the iodonium intermediate 3a, which undergoes 5-exo-trig cyclization to from the cyclized product 4a. The alternative 6-endo-trig pathway to produce the product 6a is not favoured and thus ruled out. The same mechanism may be followed in case of other sub strates (Scheme 4).
3. Conclusions
We have demonstrated that molecular iodine-mediated 5-exo-trig cyclization in aqueous micelle using CTAB as surfactant and N-iodosuccinimide in actonitrile are efficacious strategies for the synthesis of a range of doubly fused [5,6] heterocyclic compounds from a series of bis-allylhydroxy naphthalene precursors. The methodologies described here may be attractive for the synthesis of analogs of some important potentially bioactive heterocycles.
General. All the reagents were obtained from commercial sources and were used as such. Except methanol (which was HPLC-grade), the rest of the solvents were dried and distilled before use. Elemental analyses and Mass spectra (ESI+) were performed at the Indian Institute of Chemical Biology, Kolkata. IR spectra were recorded on KBr discs on a Perkin Elmer L 120-000A apparatus (νmax in cm−1). Routine 1H and 13C{1H} NMR
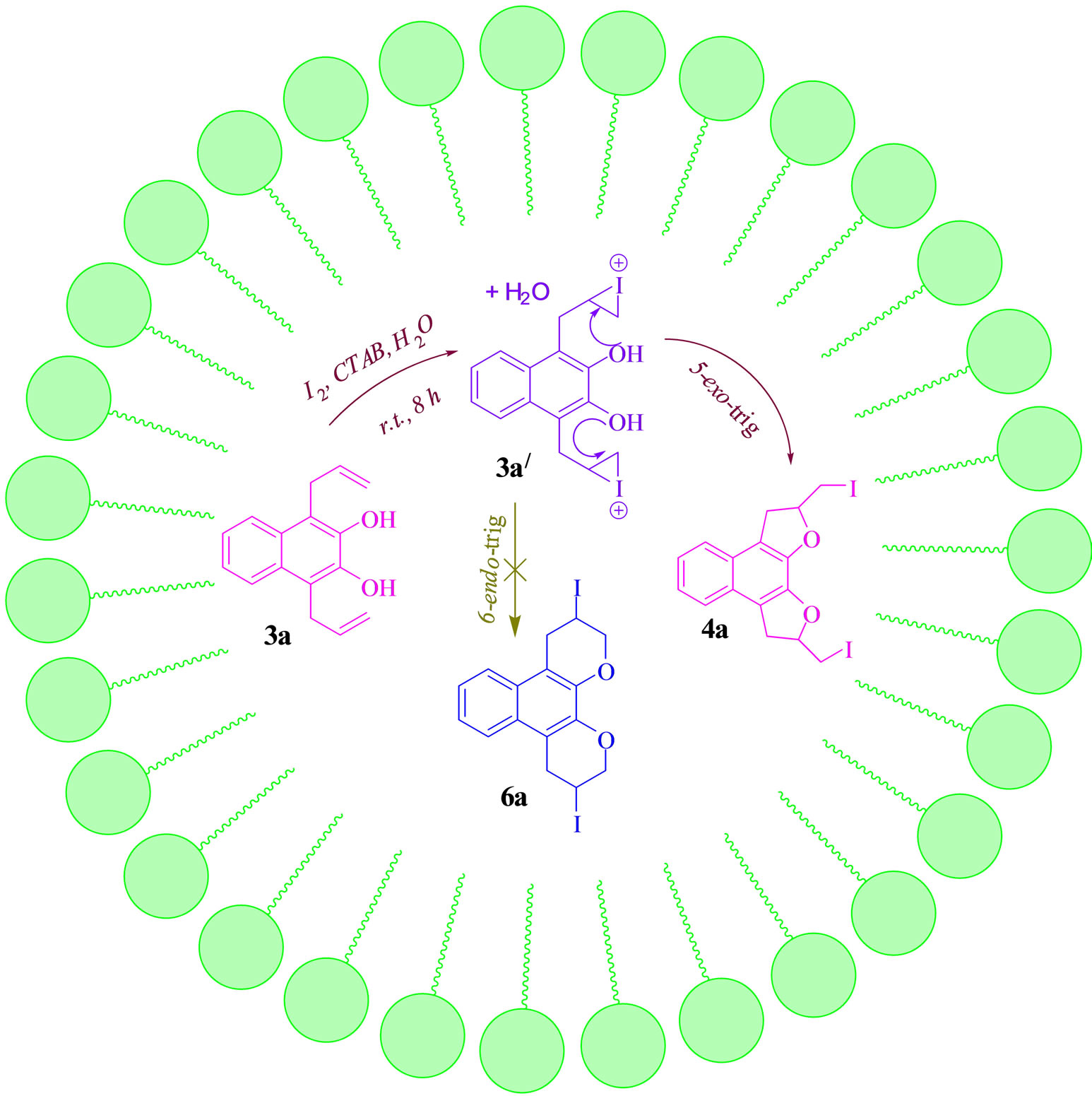
Scheme 4.Probable mechanistic pathway for the formation of 4a.
spectra were recorded with a Bruker DPX-400 MHz instrument at 298 K. The chemical shifts (δ) were given in ppm and the coupling constants (J) in Hz. CDCl3 were used as solvent and SiMe4 as the reference standard for recording of spectra. Silica gel [(100 - 200, 230 - 400 mesh), SRL, India] was used for chromatographic separation. Silica gel G [E-Marck (India)] and Silica gel 60F 254 [E-Marck (Germany)] were used for TLC. Petroleum ether refers to the fraction boiling between 60˚C and 80˚C.
General procedure for the preparation of 2,3-bis (2-methylallyloxy) naphthalene (2b), 2,7-bis (2-methylallyloxy) naphthalene (2d), and 1,5-bis (2-methyllallyloxy) naphthalene (2f).
A mixture of the compound 1b or 1d or 1f (1 mmol), 3-chloro-2-methyl-1-propene (2.5 mmol) and anhydrous K2CO3 (4.5 g), NaI (100 mg) were refluxed in anhydrous acetone (75 mL) for 12 - 15 h. The mixture was cooled, filtered, and the solvent was evaporated. The residual mass was extracted with CH2Cl2 (2 × 25 mL), washed with water (2 × 25 mL), and dried (Na2SO4). The solvent (CH2Cl2) was removed and the residual gummy mass was subjected to column chromatography over silica gel using petroleum ether-ethyl acetate (9:1) as eluent to produce the desired diallyl derivatives 2b or 2d or 2f.
The simple diallyl derivatives 2a, 2c and 2e were synthesized according to the literature procedure [24,25].
2b: Reddish yellow solid, 82% yield; mp 46˚C - 48˚C; LRMS (70 eV, EI): m/z = 268 (M+); IR (KBr): ν/cm−1 = 3055, 2916, 1587, 1513, 1482, 1262, 1171, 1112, 1003, 857, 745; 1H NMR (400 MHz, CDCl3): δ = 1.86 (s, 6H, CH3), 4.60 (s, 4H, OCH2), 5.05 (s, 2H, =CH2), 5.13 (s, 2H, =CH2), 7.10 (s, 2H, ArH), 7.28 - 7.33 (m, 4H, ArH). Analysis Calcd for C18H20O2 (268.15): C, 80.56; H, 7.51%. Found: C, 80.49, H, 7.58%.
2d: Yellow solid, 74% yield; mp 166˚C - 168˚C; HRMS (EI): m/z = 268.1469 (M+); IR (KBr): ν/cm−1 = 3031, 2925, 1625, 1606, 1551, 1453, 1210, 1169, 1021, 904, 835; 1H NMR (300 MHz, CDCl3): δ = 1.86 (s, 6H, CH3), 4.52 (s, 4H, OCH2), 5.01 (s, 2H, =CH2), 5.15 (s, 2H, =CH2), 7.00 - 7.04 (m, 4H, ArH), 7.65 (d, J 7.8 Hz, 2H, ArH); 13C NMR (100 MHz, CDCl3): δ = 19.4, 71.7, 106.6, 112.2, 116.4, 124.4, 129.1, 135.8, 140.9, 157.3. Analysis Calcd for C18H20O2 (268.15): C, 80.56; H, 7.51%. Found: C, 80.64, H, 7.62%.
2f: Yellow solid, 78% yield; mp 86˚C - 88˚C; LRMS (70 eV, EI): m/z = 268 (M+); IR (KBr): ν/cm−1 = 3076, 2976, 1655, 1592, 1512, 1418, 1264, 1043, 904, 774; 1H NMR (300 MHz, CDCl3): δ = 1.91 (s, 6H, CH3), 4.59 (s, 4H, OCH2), 5.03 (s, 2H, =CH2), 5.20 (s, 2H, =CH2), 6.81 (d, J 7.8 Hz, 2H, ArH), 7.37 (t, J 7.8 Hz, 2H, ArH), 7.91 (d, J 7.8 Hz, 2H, ArH). Analysis Calcd for C18H20O2 (268.15): C, 80.56; H, 7.51%. Found: C, 80.41, H, 7.38%.
General procedure for the preparation of precursors: 1,4-bis(2-methylallyl) naphthalene -2,3-diol (3b), or 1,8-bis(2-methylallyl) naphthalene-2,7-diol (3d), or 2,6-bis (2-methylallyl)naphthalene-1,5-diol (3f):
Compound 2b or 2d or 2f was refluxed in o-dichloro benzene (5 mL) for about 8 - 10 h. The mixture was cooled and directly subjected to column chromatography over solid silica gel. The desired product (3b or 3d or 3f) was obtained in pure form after usual column chromatographic separation using petroleum ether-ethyl acetate (8:2) as eluent.
The other bis-allylhydroxy naphthalene derivatives 3a, 3c and 3e were synthesized according to the literature procedure [24,25].
3b: White solid, 71% yield; mp 120˚C - 121˚C; LRMS (70 eV, EI): m/z = 268 (M+); IR (KBr): ν/cm−1 = 3377, 1628, 1607, 1517, 1454, 1384, 1355, 1259, 1221, 1031, 966, 845, 742; 1H NMR (400 MHz, CDCl3): δ = 1.84 (s, 6H, CH3), 3.80 (s, 4H, CH2), 4.72 (s, 2H, =CH2), 4.90 (s, 2H, =CH2), 5.83 (s, 2H, OH), 7.30 - 7.36 (m, 2H, ArH), 7.66 (d, J 7.2 Hz, 1H, ArH), 7.82 (d, J 7.2 Hz, 1H, ArH); 13C NMR (100 MHz, CDCl3): δ = 22.7, 34.1, 109.0, 112.0, 118.2, 121.9, 123.3, 124.2, 127.3, 128.8, 129.9, 142.9, 144.3. Analysis Calcd for C18H20O2 (268.15): C, 80.56; H, 7.51%. Found: C, 80.47, H, 7.68%.
3d: Reddish yellow solid, 76% yield; mp 90˚C - 92˚C; LRMS (70 eV, EI): m/z = 268 (M+); IR (KBr): ν/cm−1 = 3407, 2973, 1627, 1516, 1456, 1355, 1280, 1207, 1018, 833; 1H NMR (400 MHz, CDCl3): δ = 1.86 (s, 6H, CH3), 4.51 (s, 4H, CH2), 5.01 (s, 2H, =CH2), 5.14 (s, 2H, =CH2), 5.34 (s, 2H, OH), 6.91-7.03 (m, 2H, ArH), 7.65 (d, J 8.8 Hz, 2H, ArH). Analysis Calcd for C18H20O2 (268.15): C, 80.56; H, 7.51%. Found: C, 80.72, H, 7.60%.
3f: Yellow gummy mass, 74% yield; LRMS (70 eV, EI): m/z = 268 (M+); IR (KBr): ν/cm−1 = 3481, 2917, 1653, 1509, 1408, 1257, 1072, 1048, 898, 794; 1H NMR (400 MHz, CDCl3): δ = 1.76 (s, 6H, CH3), 3.44 (s, 2H, CH2), 3.54 (s, 2H, CH2), 4.71 (s, 2H, =CH2), 4.92 (s, 2H, =CH2), 5.76 (s, 2H, OH), 7.76 (d, J 8.4 Hz, 2H, ArH), 7.86 (d, J 8.4 Hz, 2H, ArH); 13C NMR (100 MHz, CDCl3): δ = 22.4, 37.2, 112.5, 114.6, 118.4, 125.2, 128.1, 143.0, 149.9. Analysis Calcd for C18H20O2 (268.15): C, 80.56; H, 7.51%. Found: C, 80.42, H, 7.34%.
General procedure for the preparation of 4(a-f) using N-Iodosuccinimide: (Condition A).
N-iodosuccinimide (0.450 g, 2 mmol) was added to a dry and distilled acetonitrile (20 mL) solution of the compounds 3(a-f) (1 mmol) at 0˚C - 5˚C under nitrogen atmosphere. The reaction mixture was then magnetically stirred for 1h at 0˚C - 5˚C. It was then stirred for an additional period of 3h at room temperature. On completion of the reaction (as monitored by TLC), acetonitrile was removed from the reaction mixture under reduced pressure. The residual mass was then extracted with CH2Cl2 (3 × 25 mL) and was washed with 5% NaHSO3 solution (3 × 25 mL) and water (3 × 25 mL) and dried (Na2SO4). The residual mass after removal of the solvent (CH2Cl2) was subjected to column chromatography over silica gel using petroleum ether-ethyl acetate (9:1) as eluent to give the products 4(a-f) in 71% - 80% yields.
General procedure for the molecular iodine-mediated cyclization of 3(a-f) in aqueous medium in the presence of CTAB: (Condition B).
Di-allyl-dihydroxy naphthalene derivatives 3(a-f) (0.5 mmol), surfactant CTAB (122 mg, 0.30 mmol) and water (10 mL) were mixed in a conical flask (50 mL), and the mixture was stirred at 0˚C - 5˚C for 15 min. Molecular iodine (192 mg, 1.5 equivalent) was added in three portions at an interval of 10 - 15 minutes and the reaction mixture was stirred at 0˚C - 5˚C for 1h and then at r.t. for 7 h. After the reaction was over (as detected by TLC), the reaction mixture was extracted with EtOAc (3 × 15 mL), washed with saturated sodium sulphite solution (2 × 10 mL), and brine (1 × 10 mL), dried over anhydrous sodium sulphate. The solvent was distilled off in a rotary evaporator at r.t. under reduced pressure. The crude mass was purified by chromatography over silica gel using petroleum ether-ethyl acetate (9:1) as eluent to give the pure products 4(a-f) in 76% - 88% yields.
4a: White solid, mp 197˚C - 199˚C; HRMS (EI): m/z = 491.9088 (M+), 514.8431 (M+Na)+. IR (KBr): ν/cm−1 = 3036, 2938, 2918, 1633, 1596, 1526, 1460, 1377, 1285, 1180, 1119, 1027, 982, 951, 855, 744, 622; 1H NMR (400 MHz, CDCl3): δ = 3.37 (dd, J 15.2, 6.8 Hz, 2H, CH2), 3.39-3.43 (m, 2H, CH2), 3.54-3.60 (m, 2H, CH2), 3.68 (dd, J 15.6, 9.6 Hz, 2H, CH2), 5.12-5.18 (m, 2H, CH), 7.37 (dd, J 6.0, 3.2 Hz, 2H, ArH), 7.58 (dd, J 6.0, 3.2 Hz, 2H, ArH); 13C NMR (100 MHz, CDCl3): δ = 8.9, 35.4, 83.6, 119.6, 123.5, 124.1, 126.4, 143.3. Analysis Calcd for C16H14I2O2 (491.91): C, 39.05, H, 2.87%. Found: C, 39.19, H, 3.01%.
4b: Yellow solid, mp 106˚C - 108˚C; LRMS (70 eV, EI): m/z = 520 (M+); IR (KBr): ν/cm−1 = 3019, 1638, 1606, 1520, 1464, 1385, 1295, 1138, 1055, 965, 893, 761; 1H NMR (300 MHz, CDCl3): δ = 1.76 (s, 6H, CH3), 3.36 (d, J 15.6 Hz, 2H, CH2), 3.52 (s, 4H, CH2), 3.61 (d, J 15.6 Hz, 2H, CH2), 7.24-7.37 (m, 1H, ArH), 7.50 (dd, J 8.4, 0.9 Hz, 2H, ArH), 7.67 (dd, J 8.4, 1.5 Hz, 1H, ArH); 13C NMR (100 MHz, CDCl3): δ = 15.2, 26.4, 40.9, 89.0, 110.6, 123.7, 124.2, 127.3, 141.3. Analysis Calcd for C18H18I2O2 (519.94): C, 41.56, H, 3.49%. Found: C, 41.42, H, 3.31%.
4c: Reddish yellow gummy mass; LRMS (70 eV, EI): m/z = 492 (M+); IR (KBr): ν/cm−1 = 3018, 1632, 1518, 1457, 1373, 1241, 1218, 1061, 994, 933, 827, 772; 1H NMR (400 MHz, CDCl3): δ = 3.19 - 3.27 (m, 2H, CH2), 3.37-3.47 (m, 2H, CH2), 3.48 - 3.53 (m, 2H), 3.62 (dd, J 15.6, 6.4 Hz, 2H, CH2), 5.04-5.07 (m, 2H, CH), 7.58-7.63 (m, 2H, ArH), 7.71 (dd, J 11.6, 8 Hz, 2H, ArH); 13C NMR (100 MHz, CDCl3): δ = 9.3, 39.9, 81.9, 109.4, 117.8, 124.8, 129.0, 132.7, 157.3. Analysis Calcd for C16H14I2O2 (491.91): C, 39.05, H, 2.87%. Found: C, 39.22, H, 2.99%.
4d: Grey gummy mass; LRMS (70 eV, EI): m/z = 520 (M+); IR (KBr): ν/cm−1 = 3076, 2974, 1626, 1512, 1451, 1377, 1275, 1195, 1053, 971, 903, 829; 1H NMR (400 MHz, CDCl3): δ = 1.89 (s, 6H, CH3), 4.61 (s, 4H, CH2), 5.06 (s, 2H, CH2), 5.21 (s, 2H, CH2), 7.10 (d, J 8.8 Hz, 2H, ArH), 7.65 (d, J 8.8 Hz, 2H, ArH); 13C NMR (100 MHz, CDCl3): δ = 19.5, 26.2, 42.1, 85.3, 113.9, 116.7, 124.8, 130.2, 136.3, 158.8. Analysis Calcd for C18H18I2O2 (519.94): C, 41.56, H, 3.49%. Found: C, 41.38, H, 3.58%.
4e: White solid, mp 168˚C - 170˚C; HRMS (EI): m/z = 491.9078 (M+), IR (KBr): ν/cm−1 = 3414, 2899, 1599, 1527, 1400, 1284, 1218, 1174, 1115, 1067, 966, 804; 1H NMR (400 MHz, CDCl3): δ = 3.21 (dd, J 15.6, 6.4 Hz, 2H, CH2), 3.36 - 3.41 (m, 2H, CH2), 3.51 - 3.58 (m, 4H, CH2), 5.03-5.10 (m, 2H, CH), 7.28 (d, J 8.4 Hz, 2H, ArH), 7.49 (d, J 8.0 Hz, 2H, ArH); 13C NMR (100 MHz, CDCl3): δ = 9.3, 36.9, 82.1, 114.2, 118.7, 120.9, 122.4, 154.5. Analysis Calcd for C16H14I2O2 (491.91): C, 39.05, H, 2.87%. Found: C, 38.92, H, 2.66%.
4f: Light yellow solid, mp 72˚C - 74˚C; LRMS (70 eV, EI): m/z = 520 (M+); IR (KBr): ν/cm−1 = 3013, 2971, 2914, 1596, 1518, 1407, 1292, 1162, 1031, 996, 909, 787; 1H NMR (400 MHz, CDCl3): δ = 1.91 (s, 6H, CH3), 3.24 (d, J 15.6 Hz, 2H, CH2), 3.45 - 3.53 (m, 6H, CH2), 7.53 (d, J 8.4 Hz, 2H, ArH), 7.86 (d, J 8.4 Hz, 2H, ArH); 13C NMR (100 MHz, CDCl3): δ = 19.6, 26.3, 42.1, 87.2, 113.9, 122.1, 125.4, 126.2, 154.5. Analysis Calcd for C18H18I2O2 (519.94): C, 41.56, H, 3.49%. Found: C, 41.60, H, 3.34%.
General procedure for the dehydroiodination of compounds 4a, 4c and 4e:
Compound 4a (1 mmol) was dissolved in rectified spirit (5 mL) and potassium hydroxide (0.112 g, 2 mmol) was added and the reaction mixture was refluxed for 2 - 3 h. After the reaction was over (as detected by TLC), rectified spirit was removed and the residue was extracted with dichloromethane (3 × 15 mL) and washed with water (2 × 20 mL) and dried (Na2SO4). The residual mass after removal of the solvent was subjected to column chromatography over silica gel using petroleum etherethyl-acetate (9:1) as eluent to give the product 5a (89%).
Similarly the dehydroiodination of compounds 4c and 4e were carried out to give 5c and 5e, respectively and found to have identical spectral data as that of literature report [24].
5a: Light yellow solid, 89% yield; mp. 146˚C - 147˚C; HRMS (EI): m/z = 237.0918 (M+ H)+. IR (KBr): ν/cm−1 = 2912, 1625, 1595, 1434, 1358, 1192, 926, 809, 754; 1H NMR (400 MHz, CDCl3): δ = 2.56 (s, 6H, CH3), 6.86 (s, 2H, =CH), 7.50 (dd, J 6.0, 3.2 Hz, 2H, ArH), 8.07 (dd, J 6.0, 3.2 Hz, 2H, ArH); 13C NMR (100 MHz, CDCl3): δ = 14.1, 102.5, 122.1, 124.0, 124.3, 124.7, 139.0, 153.6. Analysis Calcd for C16H12O2 (236.08): C, 81.34, H, 5.12%. Found: C, 81.22, H, 5.26%.
4. Acknowledgements
We thank to UGC (New Delhi) for providing financial support [F. PSW-013/ 11 - 12 (ERO)]. One of us (PKB) dedicates this paper in the commemoration of his beloved late father, Sri Bharati Bhusan Basu. AG is grateful to DST (New Delhi) for a research fellowship (SRF). We are extremely grateful to Prof. K.C. Majumdar, University of Kalyani for generous help during the correction of the manuscript. We are thankful to Sri. Latibuddin Thander of our department for help and kind cooperation.
NOTES