Antifungal activity of total and fractionated phenolic extracts from two wild edible herbs ()
1. INTRODUCTION
The growing attention of consumers, that require fruit and vegetables free of pesticide residues, the legislative restrictions that aim at increasing food safety, and the need for high quality products, have stimulated research towards the development of alternative defence systems, such as natural pesticides with a lower environmental impact, able to limit the development of alterations on fresh fruit and vegetables in postharvest [1].
The most important postharvest fungal pathogens are Botrytis cinerea, Monilinia spp., Penicillium spp., and Aspergillus spp. [2]. The postharvest control of these pathogens is quite efficiently performed by synthetic chemical fungicides [3]. However, the reduced number of authorized active ingredients and the increased resistance of some postharvest fungal pathogens to the few authorized fungicides have increased efforts to develop alternative or complementary control means [1,4-9]. The use of natural substances extracted from plants with antimicrobial activity is considered a promising new strategy for reducing postharvest diseases, replacing or supporting the use of synthetic fungicides, to ensure an acceptable level of disease control associated with a low environmental impact. The phenolic extracts contain substances with antimicrobial activity; in particular, studies which have been carried out in the last years aimed to broaden the knowledge of the active ingredients present in some wild edible plants for their application as natural pesticides [10,11]. It is well known that in nature there are many systems able to release molecules with cytotoxic or more generally biocidal activity, that are the result of evolutionary processes designed to the self-protection of plants from pathogens, herbivores and competitors [12].
Among wild herbaceous species known in Italy, about 800, belonging to 91 families, are classified as edible [13]. They are interesting from an ethno-botanical point of view, since a lot of them are used both in traditional recipes as raw vegetables [14] and in popular medicine as a source of alternative drugs. In fact, they are known as a rich source of antioxidant, anti-inflammatory, diuretic, antibacterial, and antiviral active substances, with medicinal as well as cosmetic applications [15-18]. Generally, they are wild herbaceous species easily adaptable to growing conditions in open field as well in greenhouse, but not widely known as food plants [14].
Salad burnet (Sanguisorba minor Scop. s.l.), belonging to the Rosaceae family, is found in Europe, Northwest Africa and Western Asia; moreover, it has also spread in North America. It is a perennial herbaceous plant 40 - 90 cm tall, drought-tolerant, growing within the whole year. It is used as an ingredient in both salads and dressings, having a flavor described as “light cucumber”. Typically, the youngest leaves are used, since their degree of bitterness increases with the physiological stage of the plant [14].
Broomrape (Orobanche crenata Forsk.), belonging to the Orobanchaceae family, is a parasitic seed plant commonly known as “bean broomrape”, that penetrates living tissue of host plants to extract nutrients and water via haustoria. It is the most important parasite of faba bean (Vicia faba). It spreads in the Mediterranean basin and North and East Africa. It lacks in chlorophyll; the host phloem and xylem are its principal sources of carbohydrates and minerals, respectively. It has also some interest as edible plant because of the high content of antioxidant phenolics of its tender shoots, the edible portion of the plant [14].
The antimicrobial activity of phenolic extracts obtained from wild edible species against postharvest fungal pathogens has been little explored [11,19]. In this work, the in vitro antifungal activity of phenolic extracts obtained from S. minor and O. crenata against important postharvest fungal pathogens was evaluated. Moreover, phenolic fractions and purified phenolic compounds obtained from these extracts were tested as antifungal agents against the same phytopathogenic fungi.
2. MATERIALS AND METHODS
2.1. Plant Material and Chemical Reagents
Plants were collected from Murgia plateau (Apulia region, Southern Italy) in springtime, transferred to the laboratory, and processed within 3 hours from the picking. Only the edible part of each plant was selected for trials, being stems for O. crenata and leaves for S. minor. A representative and randomized amount of fresh plant material (at least 2 Kg per species) was dried in a ventilated oven at 40˚C until constant weight (36 - 48 h), for the dry matter content evaluation.
High performance liquid chromatography (HPLC) grade water was obtained by a Milli-Q system (Millipore, Bedford, MA, USA). Methanol (HPLC grade) was obtained from Carlo Erba Reagents (Milan, Italy). HPLC standards verbascoside and isoverbascoside were purchased from Phytolab GmbH & Co. KG (Vestenbergsgreuth, Germany); quercetin-3-glucoside and kaempferol-3-glucoside were from Extrasynthèse (Genay, France). All HPLC standards had a chromatographic purity > 95%. Potato Dextrose Agar (PDA) and Potato Dextrose Broth (PDB) were purchased from Oxoid Ltd. (Berkshire, UK).
2.2. Preparation of Plant Extracts
About 50 g of fresh plant tissue of the two species were extracted twice with refluxing 80% aqueous methanol (1:5 w/v) for 1 h. After extraction, the methanolic extracts were filtered through Whatman filter paper and evaporated to dryness under reduced pressure at 35˚C, using a rotary evaporator. The residue was dissolved in 50 mL of potassium phosphate buffer (KH2PO4-K2HPO4, 0.1 M, pH = 5.5); the resulting solution was centrifuged at 10,000 × g; the supernatant was filtered through a sterile 0.22 µm pore size membrane filter (Millipore, Bedford, MA, USA), and stored at −20˚C until use for in vitro activity assays.
Besides the methanolic extracts, aqueous extracts of the two species were also prepared, under the same operating conditions described for methanolic extracts, except for the use of boiling water instead of 80% aqueous methanol as solvent.
2.3. HPLC Analysis of Phenolic Compounds
The HPLC analysis of phenolic compounds in the extracts was performed as previously described (Gatto et al., 2011). Briefly, an Agilent 1100 Series liquid chromatograph (Agilent Technologies Inc., Santa Clara, CA, USA) equipped with binary gradient pump (Agilent P/N G1312A) and spectrophotometric photodiode array detector (DAD) (Agilent P/N G1328A) was used as chromatographic system. The Agilent ChemStation (Rev. A. 06.03) software was used for spectra and data processing. An analytical Phenomenex (Torrance, California, USA) Luna C18 5 µm (250 × 4.6 mm) column at 35˚C in thermostatic oven (Agilent P/N G1316A) was used for peak separation. A binary gradient elution at a flow rate of 1 mL min−1 with methanol (solvent A) and 5% (v/v) acetic acid in deionized water (solvent B) was used. The elution profile was as follows: 0 - 25 min = 15% - 40% A in B; 25 - 30 min = isocratic 40% A in B; 30 - 45 min = 40% - 63% A in B; 45 - 47 min = isocratic 63% A in B; 47 - 52 min = 63% - 100% A in B; 52 - 56 min = isocratic 100% A, and then back to the equilibrium conditions (15% A in B).
2.4. Fractionation of Phenolic Extracts
A method to fractionate phenolic extracts by Flash Liquid Chromatography (FLC) has been developed. A chromatographic Reveleris® system (Grace Davison Discovery Sciences) equipped with both spectrophotometric UV detector and Evaporative Light Scattering Detector (ELSD) was used. A preparative reversed-phase C18 column Reveleris® 12 gr (Grace P/N 5152103) was used for peak separation. Binary gradient elution profiles at a flow rate of 30 mL∙min−1 with methanol (solvent A) and deionized water (solvent B) were performed. The different elution profiles used to fractionate phenolic extracts of the two species allowed to obtain five and four fractions from S. minor and O. crenata extracts, respectively.
2.5. Fungal Pathogens
Extracts and FLC fractions were individually tested in vitro against 8 fungal pathogens: B. cinerea, the causal agent of grey mould; M. laxa and M. fruticola, inducing brown rot of stone fruit; P. digitatum, causing green mould of citrus fruit; P. italicum, causing blue mould of citrus fruit; P. expansum, causing blue mould of apple; A. niger and A. carbonarius, the causal agents of black mould. The pathogens were isolated from infected fruit and vegetables collected in local markets (Figure 1) and cultured on PDA (39 g∙L−1 in distilled water) at 25˚C ± 1˚C for 5 days. Pure cultures were maintained at 4˚C in PDA tubes. Spores were harvested from 2-week-old PDA fungal cultures grown at 25˚C ± 1˚C, by adding 3 - 5 millilitres of water in each Petri dish. Suspensions were filtered through two layers of cheesecloth, and the concentration was adjusted to 104 spores mL−1.
2.6. In Vitro Assay of the Antifungal Activity of Plant Extracts
Both total plant extracts and FLC fractions were tested

Figure 1. Colonies of Monilinia, Penicillium, and Alternaria spp. in Petri dish.
for in vitro antifungal activity. Assays on 96-well 100 µL volume plates were set up; each well contained 10 µL of liquid substrate (PDB), 2 µL of conidial suspension 108 spores mL−1, and 88 µL of the extract to be tested in 4 replicates. On each plate, in addition to the control, there were also: 1) in the case of S. minor, the total extract together with five fractions obtained from the latter by FLC; 2) in the case of O. crenata, the total extract and four fractions obtained from the latter by FLC, i.e. F1 (front), F2 (verbascoside), F3 (isoverbascoside), F4 (washing). Three plates were used for each fungus; plates were incubated at 25˚C for 7 days.
The two extracts, fractions and compounds separated by FLC were also tested setting up micro-assays on the slide that allowed the qualitative analysis by optical microscopy, according to a previously developed protocol [11].
2.7. Statistical Analysis
Data were subjected to analysis of variance (ANOVA) using Statistica Software (version 6.0; StatSoft Inc., Tulsa, OK, USA). Duncan’s Multiple Range Test (DMRT; P ≤ 0.01) was used to compare the means [20].
3. RESULTS AND DISCUSSION
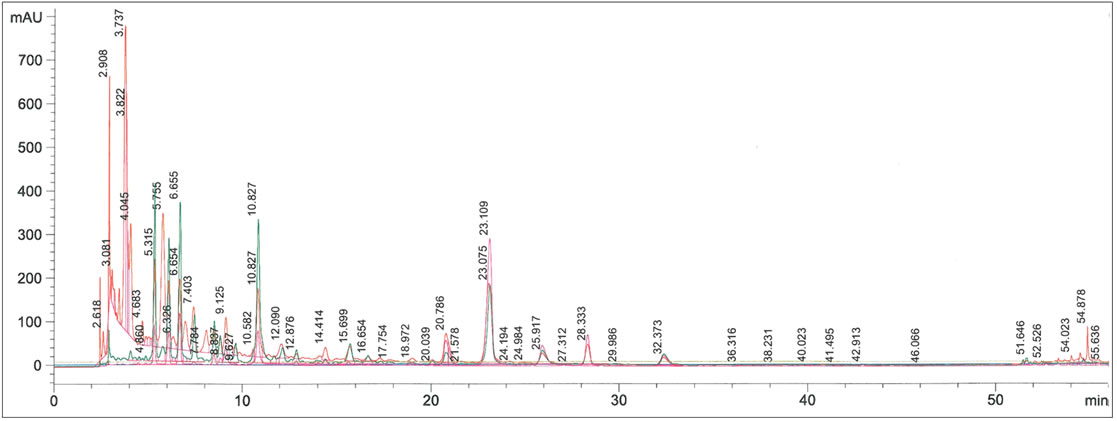
S. minor extract showed a complex HPLC chromatogram (Figure 2), with several peaks not well separated and difficult to be identified. For this reason, a fractionation method of the extract by FLC has been developed (Figure 3).
The obtained fractions were assayed for their in vitro antifungal activity against B. cinerea, P. digitatum, P. italicum, P. expansum, M. laxa, M. fructicola and A. carbonarius. The total extract showed an excellent antifungal activity (>80% inhibition, P ≤ 0.01) against M. laxa, M. fructicola, P. expansum and P. italicum (Figure 4); it showed a good efficacy (between 50% and 80% inhibition, P ≤ 0.01) also against B. cinerea and A. carbonarius, while it resulted inactive against P. digitatum (Table 1). As regards the different fractions, F3 and F4 showed to be particularly active, completely inhibiting P. italicum and B. cinerea, respectively. In particular, F4 proved to be very active against B. cinerea, unlike the total extract which caused a weak inhibition of the growth of this fungus (Table 1). This fraction showed the presence of three highly concentrated flavonoids (Figure 5(b)), two of which were identified (for comparison of retention times and UV spectra obtained by DAD with those of commercial standards) as quercetin-3-glucoside and kempferolo-3-glucoside. Given the high activity of this fraction, it could be interesting to proceed to further fractionation to achieve the separation and identification of the individual compounds and to test their biological

Figure 2. HPLC chromatogram of Sanguisorba minor methanolic extract.

Figure 3. FLC chromatogram of Sanguisorba minor methanolic extract.

Figure 4. Activity of Sanguisorba minor extract and fractions against Penicillium italicum conidia growth.
activity as individual active ingredients.
The HPLC analysis of the methanolic extract of O. crenata showed the presence of a predominant caffeic acid derivative identified as verbascoside by comparison of HPLC retention times and UV spectrum [21] with

Table 1. In vitro effect of Ssnguisorba minor total extract and FLC fractions on the growth of the different fungi. Extract activity was tested by 96-well plate assays after 7 days at 25˚C. Symbols: -, +, ++, and +++ indicate that the effect of the compound was: none, light (<50% inhibition), good (between 50% and 80% inhibition), excellent (>80% inhibition), respectively
those of commercial standards (Figure 6). O. crenata has
 (a)
(a) (b)
(b)
Figure 5. HPLC chromatograms from Sanguisorba minor methanolic extract: total extract (a) and F4 fraction (b).
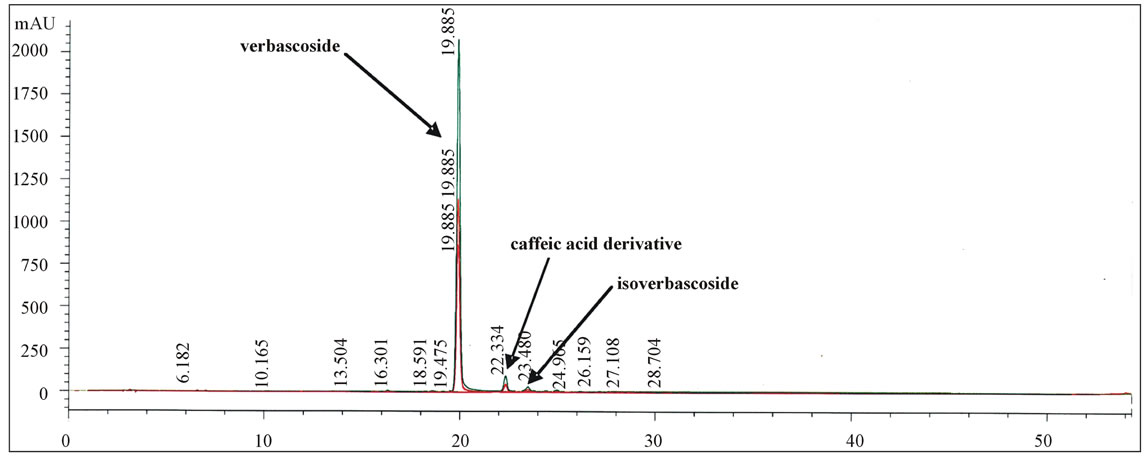
Figure 6. HPLC chromatogram of Orobanche crenata methanolic extract.
also been used to prepare an aqueous extract containing two caffeic derivatives, verbascoside and its isomer isoverbascoside, which also were provisionally identified by comparison with the commercial standards. A FLC separation method of the two compounds present in the aqueous extract was developed, that allowed to obtain the two compounds with chromatographic purity > 98%. The two compounds were lyophilized, purified, and their chemical structure was confirmed by mass spectrometry and nuclear magnetic resonance (NMR) spectroscopy (Figures 7 and 8).
Verbascoside and isoverbascoside were tested for their

Figure 7. 1H-NMR spectrum of verbascoside.

Figure 8. 1H-NMR spectrum of isoverbascoside.
antifungal activity against the above mentioned postharvest fungi (Table 2, Figures 9 and 10). Isoverbascoside completely inhibited (P ≤ 0.01) B. cinerea, P. digitatum (Figure 9), P. italicum (Figure 10), and P. expansum; it fairly inhibited M. laxa and A. carbonarius; while it showed to be less effective against M. fructicola (Table 2). Verbascoside showed significant activity (P ≤ 0.01) against P. italicum, fairly inhibited M. laxa, P. expansum, and A. carbonarius, and showed little activity against B. cinerea, M. fructicola and P. digitatum (Table 2).
The total extract of O. crenata showed to be less active than the two purified compounds (Table 2), probably due to the presence of interfering substances in the fraction F1, eliminated as a “front” (e.g. sugars), which may have contributed to reduce the effectiveness of the two active ingredients present in the total extract.
The analyses carried out by means of optical microscopy showed, in all cases in which there has been a strong antifungal activity, a direct action on the pathogen by the total extract or by the single compound. Figure 10

Figure 9. Activity of Orobanche crenata extract and fractions against Penicillium digitatum conidia growth.

Figure 10. Activity of Orobanche crenata extract and fractions against Penicillium italicum conidia growth.
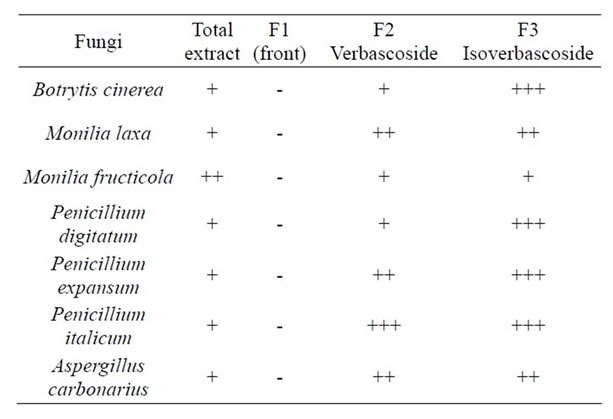
Table 2. In vitro effect of Orobanche crenata total extract and FLC fractions on the growth of the different fungi. Extract activity was tested by 96-well plate assays after 7 days at 25˚C. Symbols: -, +, ++, and +++ indicate that the effect of the compound was: none, light (<50% inhibition), good (between 50% and 80% inhibition), excellent (>80% inhibition), respectively.
shows, for example, the devastating action exerted on P. italicum by both verbascoside and isoverbascoside (fractions F2 and F3, respectively), with complete disruption of the fungal mycelium and leakage of intracellular fluid. However, an induction of host disease resistance cannot be excluded, since it has been demonstrated that phenolic compounds can induce apple resistance to blue mould [22].
4. CONCLUSIONS
The chemical characterization of phenolic extracts of S. minor and O. crenata was performed. Active fractions were separated and characterized and, in the case of O. crenata, two phenolic compounds, verbascoside and isoverbascoside, were quantitatively extracted, purified and characterized. These compounds proved to be effective in reducing the growth of some important fungi and could have a high interest as natural active ingredients against several pathogenic fungi responsible for the postharvest decay of fresh fruit and vegetables. The studied compounds could be reasonably considered harmless from the toxicological point of view, being extracted from edible species and used in concentrations very similar to those present in a normal food ration of the same plant. They could be highly interesting for subsequent studies on their effectiveness in reducing rot incidence on fruit and vegetables after harvest.
The obtained results support the possible use of some wild edible plants as a source of natural phenolic substances for the postharvest biological control of fresh fruit and vegetable rotting. They open new and exciting perspectives of work with the purpose to realize mixtures of natural active substances to be used as antifungal agents in postharvest.
5. ACKNOWLEDGEMENTS
This work has been carried out within the Project “Estrazione e caratterizzazione di composti fenolici con attività biologica da piante spontanee eduli di potenziale interesse per il settore agro-alimentare” co-funded by the “Fondazione Cassa di Risparmio di Puglia” (FCRP), Bari, Italy. The authors thank FCRP for the financial support to the Project.
Furthermore, the authors thank the Project “Rete di Laboratori per l’Innovazione degli Alimenti Funzionali (LAIFF)” (PO Puglia FESR 2007-2013 Asse I, Linea 1.2—PO Puglia FSE 2007-2013 Asse IV) which allowed the Reveleris® FLC system purchasing.

