Stability Studies of Lysine Acetylsalicylate (Aspirin Derivative): Mechanisms of Hydrolysis ()
1. Introduction
The LAS compound is an active ingredient known to Aspegic. It is a molecule that is part of the family of salicylates including aspirin, which represents one of the most commonly, used drugs in the world. Aspegic is an acetyl derivative of salicylic acid that is a white, crystalline, weakly acidic substance. Acetylsalicylic acid decomposes rapidly in solutions of ammonium acetate or of the acetates, carbonates, citrates, phosphates or hydroxides of the alkali metals. Acetylsalicylic acid is stable in dry air, but gradually hydrolyses in contact with moisture to acetic and salicylic acids. In solution with alkalis, the hydrolysis proceeds rapidly and the clear solutions formed may consist entirely of acetate and salicylate. The principal cause of the hydrolysis of this family of the salicylic acid acyl esters (e.g. aspirin), in aqueous solution about pH 3, is the attack of these substances by water ions (H3O+ or HO–) [1-6].
Since Edwards has shown that the rate of hydrolysis of aspirin is independent of pH between pH 4 and 8, the reaction has been the subject of a number of studies, particularly by Garrett [1,3-5]. Edwards suggested that in this pH range, the hydrolysis reaction resulting from the attack by a water molecule on the anion of aspirin (Scheme 1), but several authors have pointed out that this mechanism is not consistent with Edwards’ own demonstration, showing that the hydrolysis reaction is not catalyzed by acetate ion, a considerably more powerful nucleophile than water.
They considered that the facts point rather to intramolecular nucleophilic catalysis by the ionized carboxyl group [7,8]. This mechanism has been set out in its most acceptable form by Bender (Scheme 1) [9]. This hypothesis was supported by the same authors [10], which showed that a small amount of labelled oxygen from solvent  appears in the salicylic acid produced. Only Garrett has
appears in the salicylic acid produced. Only Garrett has
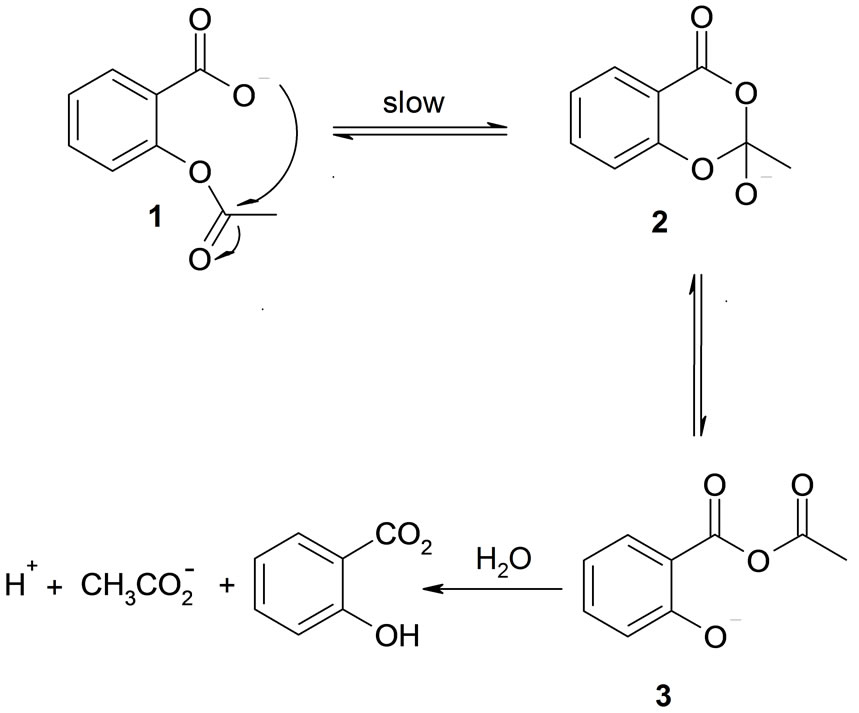
Scheme 1. Hydrolysis reaction of the LAS reagent.
seriously questioned this mechanism. He found that the addition of ethanol to the solvent increases the rate of solvolysis, and that ethyl acetate is then a product [3-6]. He ruled out the possibility that the rate increase is a generalized solvent effect by showing that the addition of dioxane has very little effect on the rate of hydrolysis of aspirin. Garrett tried to explain his results by proposing a mechanism involving nucleophilic attack by ethanol on the tetrahedral carbon atom of the intermediate (2) (Scheme 1), but this explanation has not been generally accepted [9]. Nevertheless, the demonstration that the addition of ethanol increases the rate of solvolysis does suggest strongly that the question of the involvement of a molecule of solvent in the transition state ought to be reopened. The work of Jencks and Gilchrist [11] shows clearly that ethanol and ethoxide ion are stronger nucleophiles than water and hydroxide ion in reactions at the carbonyl group, and a rate-determining step involving attack by either species would explain Garrett’s results.
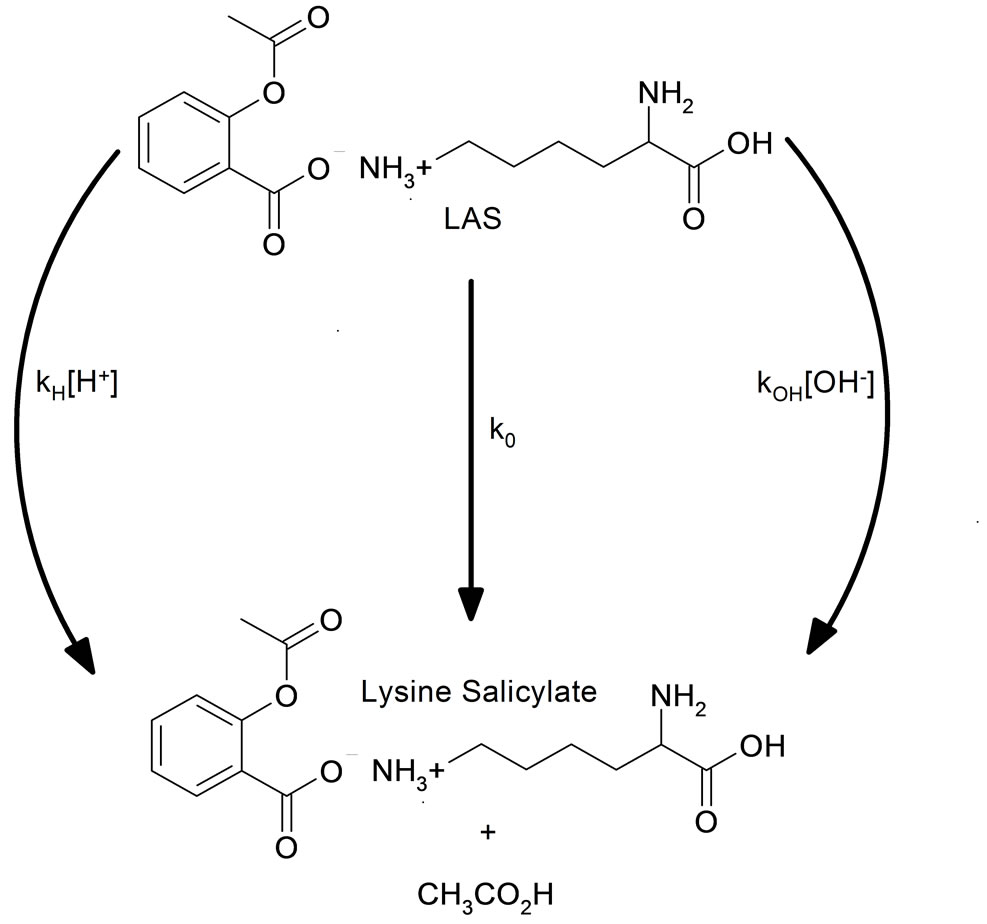
It has been recognized for many years that over a sufficiently wide pH range, three kinetically-distinguishable mechanisms for the hydrolysis of some compounds, such as epoxides and derivatives of acetylsalicylic acid [12-15]. The experimental rate expression for their hydrolysis is given by the following equation:

where kH is the specific second-order rate constant for the hydronium ion-catalyzed reaction, k0 is the specific first-order rate constant for the spontaneous reaction, and kOH is the specific second order rate constant for the hydroxide-catalyzed reaction. This paper reports on the hydrolysis kinetics of the LAS compound (LAS), for pH values between 0.50 and 13. This detailed study for a wide range of pH, allows the one hand, to extract significant results and secondly, to have an overview on the various possible mechanisms for the hydrolysis of this important reagent.
2. Experimental Section
Inorganic salts were of analytical grade, and were used without further purification, as well used normadoses solutions of HCl and NaOH were analytical grade. Water was further glass distilled before use. The LAS was obtained commercially. Aqueous solutions of reagent LAS of known concentrations 5 × 10–4 M and 10–3 M were prepared, the desired pH values are determined by adding the quantities: HCl (0.5 M) or NaOH (1 M), respectively for acidic or basic mediums and these pH values are measured with a Hanna 210 microprocessor pH-meter, equipped with a combined glass electrode and calibrated with commercial buffers (pH 4.00 and 7.00). These solutions are maintained at fixed temperature 298 K, and small aliquots were withdrawn at known time intervals. These samples were analyzed by standard Helios g UVvisible spectrometer, using quartz cells of optical path length l = 1 cm. The absorption measurements have been performed at wavelength lmax = 300 nm. Indeed, the UVvisible spectra of Figure 1, clearly show that the salicylate ion formed by hydrolysis of the LAS, has a maximum band of absorption at lmax = 300 nm, and at this maximum wavelength, the reagent LAS has a negligible absorption.
The initial concentration denoted “a” of the reagent has always been very low (5, 10–4 M), so that all the spectrophotometric measurements were carried out under conditions of pseudo-first-order, with higher concentrations of reacting species (H3O+, H2O and HO–), compared to this initial concentration a. All experimental values of optical densities were measured by reference to pure water.
3. Results and Discussion
3.1. Identification of the Catalysis Type for the Hydrolysis Reaction
The hydrolysis reaction of this reagent derivative of aspirin,

Figure 1. UV-Visible spectra for the hydrolysis of the reagent LAS, pH = 11.66, [LAS] = 5 × 10–4 M.
has been studied at 298 K in the pH range of 0.50 to 13 and the kinetics of this hydrolysis phenomenon have proved sensitive to the medium acidity, therefore, there is possibility of the existence of a catalysis phenomenon by ionic species of water (H3O+ and HO–). Generally, the mechanism of hydrolysis may be conducted according to the following Scheme 2.
The rate constants of second-order kH and kOH, are associated with catalytic decomposition process, respectively, in acidic and basic environments. While the rate constant of first-order k0, is involved in the process of hydrolysis under the effect of solvent molecules, however, it must be noted that this constant k0 result of the operation 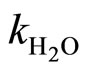 * 55.55 (water concentration), with
* 55.55 (water concentration), with  is the rate constant of second-order corresponding to the process of hydrolysis by an elementary molecule of the water.
is the rate constant of second-order corresponding to the process of hydrolysis by an elementary molecule of the water.
If a is the initial concentration of the reagent LAS, and x the concentration of salicylate ions, formed at time t, and for a fixed acidity of the medium, the rate equation on the hydrolysis reaction is given by the following expression:
 (1)
(1)
After separation of variables, we have the relation:
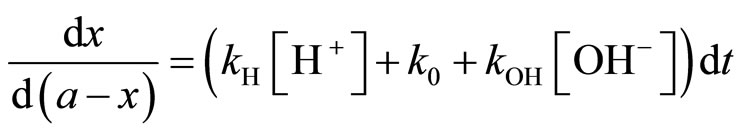 (2)
(2)
The integration of this relationship allows obtaining the following kinetic equation:
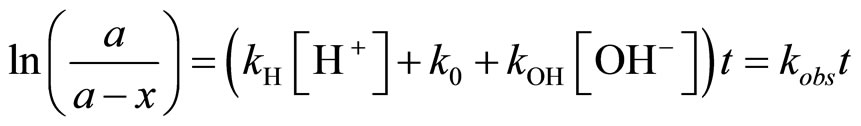 (3)
(3)
The apparent rate constant of pseudo first-order kobs,
Scheme 2. Different reaction pathways proposed for the hydrolysis reaction of LAS.
on the reaction of hydrolysis reagent LAS, is given by the expression:
 (4)
(4)
It is obvious to note that the kinetic law of pseudo first-order Equation (3) can be easily connected to changes in optical densities as a function of time, especially, only the hydrolyzed product absorbs at the measurement wavelength (lmax = 300 nm). Indeed, if D0, Dt and D∞ are respectively the values of optical density at t = 0, at time t and at the end of the reaction and if esal is the molar extinction coefficient of salicylate ion, we can write the following relations:
 ;
; ;
;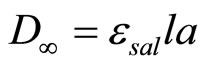 (5)
(5)
where l is the optical path, a and x are the respective concentrations of LAS reagent and hydrolyzed product. From the kinetic law of Equation (3) and the preceding relations Equation (5), we can establish relationships that express the evolution of the optical density versus time t.
 (6)
(6)
and we see immediately that the value of the apparent rate constant kobs is obtained from the slope of the straight lines
 (7)
(7)
The graphic in Figure 2 illustrates the good linearity of the observed experimental lines.
Several experiments on the hydrolysis reaction of the reagent LAS, were performed for pH values from 0.50 to 13, with HCl and NaOH as agents, respectively to determine the pH of the studied solutions. For these experiments, the kinetic experimental lines Equation (6) were plotted and the apparent rate constants kobs were calculated for all studied solutions, from the slopes of obtained
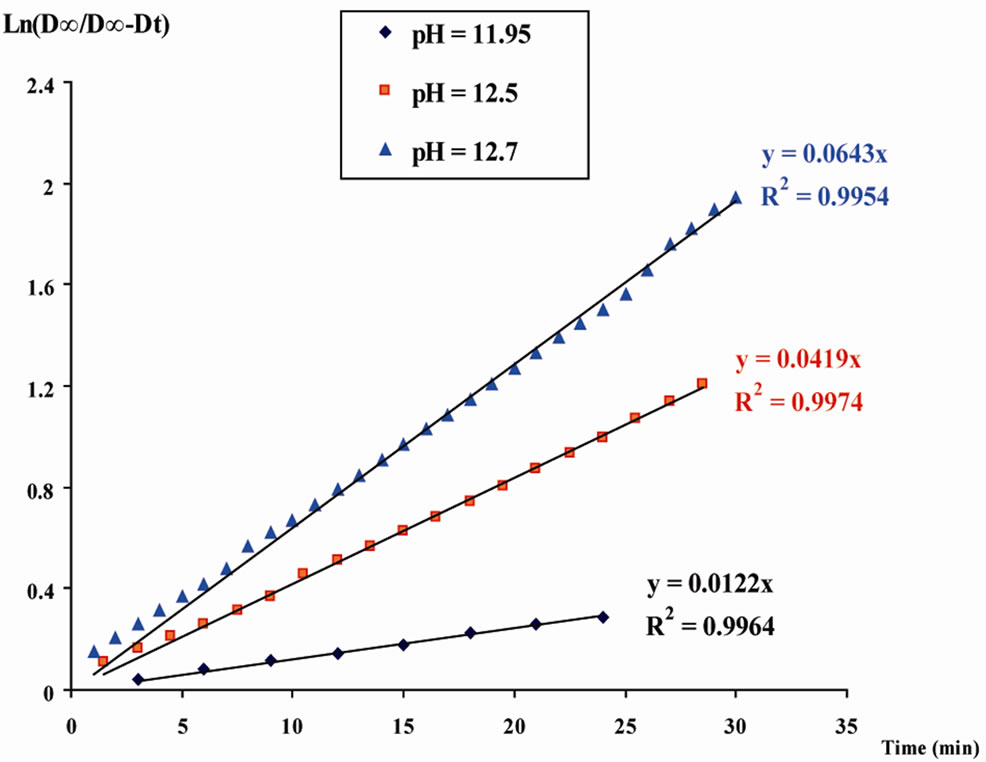
Figure 2. Kinetics for the hydrolysis reaction of LAS reagent, in different basic solutions at 298 K.
lines. Table 1 includes all results related to the phenomenon of hydrolysis of the LAS compound.
These results show, firstly that the rate of hydrolysis reaction of this compound depends on the acidity of the medium and the reaction pathway catalyzed by the HO– ions on the hydrolysis reaction is predominant, on the other hand, values of these apparent rate constants (kobs) change in different ways, according to three pH ranges. From these values, the pH-rate profile graph (log kobs = f(pH)), was established (Figure 3), and the obtained experimental curve shows three different slopes (p = –1, 0 and +1).

Table 1. Evolution of the apparent rate constants (kobs) on the medium acidity.

Figure 3. pH-rate profile for hydrolysis reaction of the LAS reagent at 298 K.
Therefore, the apparent rate constant kobs, expression is composed of three terms and is identified with the Equation (4). The shape of this curve on the pH-rate profile clearly indicates that the hydrolysis reaction of the reagent LAS, is using three different reaction pathways, for three different pH ranges and therefore, this reaction presents a phenomenon of acid-base catalysis. For pH values below 2, the curve has a negative slope (–1) and in kobs expression Equation (4), the term kH is dominant, and this hydrolysis reaction proceeds according to the reaction pathway catalyzed by H3O+ ions. The zero slope of this curve, presented for pH values between 2 and 8, clearly indicates that in this pH range, the k0 term is predominant Equation (4), the apparent rate constant kobs depends on the concentration of HO– ions, and the hydrolysis process of LAS, results from the intervention of these ions, according to the reaction pathway catalyzed by these ionic species HO–. From the curve in Figure 3, we determined the values of three rate constants kH, k0, and kOH, for different reaction pathways that constitute the global mechanism for the hydrolysis process of the LAS reagent. Table 2 shows the values for these constants and the definitive expression of the apparent rate constant kobs at 298 K, is given by the following equation:
 (8)
(8)
These results indicate that the hydrolysis reaction of the LAS reagent, is achieved using an acid-base catalysis mechanism, the reaction pathway catalyzed by HO– ions is dominant, and the degradation of this compound is important in basic medium. This value of the rate constant kOH on the hydrolysis reaction of LAS in basic medium is the same order of magnitude as the values of the constants kOH determined for the picryl chloride and trinitro-anisole hydrolysis reactions [16-18]. It is considerably less than the value calculated for the hydrolysis of 7-chloro-4,6-dinitrobenzofurazan [19], while this value is much higher than the values announced for the hydrolysis reactions of styrene oxides [20].
3.2. Determination of Activation Parameters and Reaction Mechanism
To elucidate the temperature effect on the evolution of the kinetic parameters relating to the reaction pathway catalyzed by HO– ions, to determine the activation parameters, activation energy (Ea), enthalpy (ΔH≠) and entropy (ΔS≠) for the transition state of the rate-determining step and propose a mechanism for this predominant reac-

Table 2. The three rate constants for the hydrolysis phenomenon of LAS.
tion pathway in the hydrolysis process of studied reagent (LAS). We followed the kinetics of the studied hydrolysis reaction in basic medium of constant pH (10.12), by varying the temperature between 298 and 323 K. The experimental values of the apparent rate constant kobs were calculated in the same way as the previous section, and Table 3 summarizes all results.
The linear relationship between Ln kobs and the inverse of absolute temperature (1/T) represented by the graph in Figure 4, indicated firstly, that a consistent solvolysis mechanism operated for this hydrolysis reaction pathway catalyzed by HO– ions, on the other hand, the Arrhenius law is well verified for the studied temperatures and the activation parameters for this process were calculated from the slope and intercept on the experimental right.
Ln kobs = f(1/T), plotted for the interval of studied temperatures, with the following values for the activation energy Ea = (65.60 ± 0.05) kJ.mol–1 and the preexponential factor in the Arrhenius relationship A = (1.803 ± 0.005) 106 s–1.
From these activation parameters, we determined at temperature T = 298 K, the magnitude of activation enthalpy ΔH≠ and entropy ΔS≠, according to the following expressions derived from the theory of the transition state:
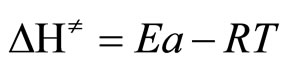 and
and  (9)
(9)
After calculation we obtain the values:


The ΔH≠ and ΔS≠ values associated with the solvolysis phenomenon of LAS reagent, can propose a structure of transition state on the studied reaction. In fact, a very negative value for the activation entropy ΔS≠ reflecting a late transition state, very organized with a structure similar products and indicates that probably, for this reaction pathway catalyzed by HO– ions, the intervention of the water molecule is in the rate-determining step, after the fast attack of the LAS reagent by very active HO– ion, and that in the transition state there is a transfer of hydrogen atoms from one site to another. Indeed, some kinetic studies [21,22] on hydrolysis reactions of some organic compounds, have suggested mechanisms which involve a water molecule in the rate-determining step, and for this reaction pathway we propose the following mechanism:

Table 3. The apparent rate constant kobs evolution vs. the temperature.

Figure 4. The Arrhenius representation Ln kobs = f(1/T) for the hydrolysis reaction of the LAS reagent at pH = 10.12.

Figure 5. Mechanism for hydroxide ion catalyzed hydrolysis of the LAS reagent.
This mechanism for the reaction pathway catalyzed by HO– ions, composed of three steps, indicating that Step 2 on the association of a water molecule with the carbanion produced by Step 1, is rate-determining step, with a highly ordered transition state whose structure resembles the products of the reaction. The passage through this highly ordered transition state with cyclic and internal transfer of hydrogen atoms from one site to another, well justifies the activation energy required for the formation of this transition state, and the very negative value of the activation entropy (ΔS≠ = –133.42 J.K–1.mol–1). Moreover, this very negative value is in favor of a bimolecular process involving two species, to imagine that highly ordered transition state. Indeed, the value of this parameter (ΔS≠) is very useful for identifying the rate-determining step molecularity. Recent studies on the degradation kinetics of some compounds [23,24], indicate clearly that for the unimolecular process, the values of the activation entropy ΔS≠ are positive.
4. Conclusion
Studies in this work have identified the stability of the active reagent LAS as a function of the medium acidity, to determine the different rate constants for the three reaction pathways involved in the hydrolysis process of this compound. Finally a study of the temperature influence has been achieved for the hydrolysis reaction in basic medium, and the activation parameters for transition complex were determined. The analysis of these results has identified an acid-base catalysis type for the hydrolysis process of this studied compound, with a clear predominance for the reaction pathway catalyzed by HO– ions in basic medium, and for this reaction pathway, the activation parameters on the transition complex was determined based on expressions derived from the transition state theory. On the one hand, the values of these parameters allowed us to propose a reaction mechanism consisting of three steps for the hydrolysis phenomenon of reagent LAS in basic medium, and secondly, to clearly specify the rate-determining step for this proposed mechanism.
NOTES