Use of
the regular melting-quench method allowed the isolation of a small glass domain
within the ternary system Li
2O-P
2O
5-Cr
2O
3 at 1000°C. Electrical conductivity and dielectric permittivity measures on
sample glasses and ceramic glasses of this domain were performed at a frequency
of 10 kHz and at temperatures between 25°C and 300°C. The values of dielectric
permittivity and electrical conductivity increase with increasing Li
2O
content. However, increases of Cr
2O
3 content, even at low
concentrations, induce a change in electrical conductivity behaviour from that
of a glass to that of a ceramic glass. The introduction of an increasing amount
of Li
2O content in vitreous phosphorus pentoxide changes its
three-dimensional network; rupture of the P-O-P bond then occurs and there is
formation of easily polarisable entities with quite high values of
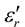
. The vibrational spectroscopy technique I.R has
allowed an efficient investigation of the structural change versus composition
within the above indicated ternary diagram. The maximal dielectric permittivity
obtained at 300°C, both for the glasses and for the ceramic glasses varied in
the order 10
4 to 3 × 10
5,
while the maximum electrical conductivity obtained at 300°C for the ceramic
glasses was in the order of 10
-3 Ωcm
-1.