In this study, series of hydroxyapatites containing Mg
2+ and CO
2-3 are prepared by the precipitation method with independently varying concentrations of CO
2-3 and Mg
2+. All the compounds are characterized by infrared spectra (IR); powder X-ray diffraction (PXRD) and elemental analysis. The physical analysis results show that the prepared compounds are pure B-type carbonate apatite. The presence of Mg
2+ and CO
2-3 in the apatite cause the following effects on its physical properties: a decrease in a-dimension but no changes in c-dimension and a decrease in crystallinity as shown in XDR patterns and IR spectra. The results of the chemical analysis allow us to predict the predominant substitution mechanisms for the CO
2-3 and the Mg
2+ incorporations in the calcium hydroxyapatites and to calculate their relative contributions x, y and z.
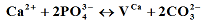
(II);
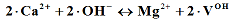
2. (IV);
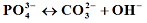
(V). Statistical studies of the results “multiple linear regression, analysis of variance (ANOVA) and t-test of the regression coefficients” allow us to determine and to test the mathematical model proposed. Finally, the present study makes it possible to write the general formula for these com-pounds.