Tolerability and hydrating effects of an anti-aging gynaecological collagen cream in women in menopause ()
1. INTRODUCTION
Perimenopause and menopause are conditions characterised by several changes that affect the structural integrity of vagina and vulva and that involve also the urinary tract [1-3]. In fact, oestrogen maintains the thickness of the vaginal epithelium and promotes the synthesis of collagen and elastin in the vaginal connective tissue. Collagen is the major macromolecular component of derma, which ensures the tissue elasticity, pliability, thickness and tone. The collagen of types I and III decreases in derma by 30% in the first 5 years following menopause [4-7]. The thinning of the vaginal epithelium, the hyalinisation of collagen and the elastin fragmentation lead to hypotonus, relaxation, dryness deriving from a deficiency in connective supports.
A sedentary lifestyle, lack of exercise, childbirth, or other events may also cause relaxation of the perineal muscles and of the tissues and muscles that surround the genital area and regulate the urinary functions. This condition may cause very frequent disturbances: from small urine leaks when coughing or laughing or dyspareunia or constipation up to severer disorders such as enuresis, recurrent cystitis and prolapse.
The use of moisturisers is a primary option in the treatment and prevention of vaginal symptoms, as showed in several published trials [3].
The principal components of moisturising products improve the skin hydration either by an occlusive or by a humectant mechanism. Fundamentally, the ability of moisturisers to restrain the transepidermal water loss and to increase the water content in the stratum corneum is an essential, though not unique, property required to improve also the skin mechanics [8-12]. However, many other components exert a synergistic beneficial effect on the epidermis through various mechanisms [12]. Polypeptides, frequently included in moisturising preparations, are able to stimulate enzymatic processes, e.g. the collagen neosynthesis, and to enhance the dermal metabolism [13,14]. The percutaneous absorption of hyaluronic acid was proven when applied with topical preparation to the intact skin though the strongly hydrophilic nature of the compound [15]. Vitamins A, C, E and B3 contribute to the anti-aging effect of many topical products. All the 4 vitamins may have an antioxidant effect on the stratum corneum [12,16-22]. Furthermore, both vitamin A in the precursor form of retinol and in the activated form of retinoic acid and vitamins C and B3 can promote the neosynthesis of collagen in the dermis [16,19,22-27]. Very frequently used components are also antioxidants of botanical origin. In particular, polyphenols and soy isoflavones exert a proven anti-aging effect through antioxidant mechanisms.
In the present study, a marketed cream—class II A medical device—containing collagen, hyaluronate, vitamins A, C, E and B3 and isoflavones was investigated in menopausal women who applied it for a period of 3 weeks.
2. METHODS
This mono-centric study comprised 20 women in good health and in menopausal status (natural or surgical) for at least one year. The volunteers were included in the study at the Phase I Unit of CROSS Research S.A., Arzo, Switzerland, from 16NOV11 and 15DEC11.
The volunteers selected for the study presented a reduced elasticity of the perineal skin and vaginal dryness reported by the screened women without any instrumental ascertainment. Before definitive enrolment in the study, an evaluation of the vulvar and vaginal mucosa was done by a gynaecologist and an evaluation of the perineal and abdominal skin was performed by a dermatologist. The gynaecological and dermatological examinations prevented the inclusion of women with relevant skin or mucosal alterations.
Other exclusion criteria were 1) intake of any medication judged by the investigator as incompatible with the study and in particular any substitutive hormonal therapy 2), known sensitivity to skin care products or cosmetic products, 3) clinically relevant abnormal physical findings, 4) history of bacterial infection at the urinary tract in the previous 2 weeks and 5) unwillingness to provide written informed consent.
Primary objective of the study was the investigation of the local tolerability of the test product (Perilei™ Pausa) applied according to a multiple dose regimen. The study was also aiming at investigating the hydration and elasticity of the perineal and abdominal skin and the moisturising of the vaginal mucosa during and after multiple dose application of the product.
Since no previous clinical investigation of the test product was performed, the sample size determination was not based on a statistical evaluation. A total of 20 subjects were considered adequate to achieve the objectives of the trial.
Measurements of skin hydration and elasticity were performed using a specific multi-probe device in accordance with the instrument producer instructions [28].
The measurement of skin elasticity (done with Cutometer MPA 580) is based upon peculiar mechanical properties of human skin and represents a non-invasive method whose use has become solid and widely spread in dermatology and cosmetics [29-35] and acknowledged by the European Expert Group on Efficacy Measurement of Cosmetics and other Topical Products (EEMCO) [36].
The corneometer CM 825 allows the measurement of stratum corneum hydration. It is regarded as a reliable measure of skin conditions and also this method gained validity and use diffusion in dermatology and cosmetics [37-39]. The reliability and the reproducibility of these measures were previously demonstrated [40-42].
The volunteers evaluated the moisturising effect on the vaginal mucosa using a Visual Analogue Scale (VAS) 0 - 100 mm. The volunteers were asked a list of questions in order to evaluate the comfort of the cream use.
The cream was daily applied for 1 week in the evening. During the following 2 weeks, it was applied on alternate days in the evening, i.e. on days 8, 10, 12, 14, 16, 18 and 20.
The study participants applied the product in the evening before going to bed on the days scheduled for applications. At each application, 1 tube (5 mL) was used for the perineal skin and vaginal mucosa and 1 tube (5 mL) was used for the abdominal skin. During each application to the vaginal mucosa, the subjects were lying in the supine position. The applicator was slowly introduced into the vagina and about the half of the first tube was extruded. The remaining amount of cream was applied to the perineal and vulvar area (see Figure 1). The subject rubbed gently the cream with circular movements without rinsing. The subjects gently spread the cream with circular movements, when they applied it to the abdominal skin.
The time points for the assessments of vaginal and skin moisturising and skin elasticity were scheduled at

Figure 1. The diagonal bar zone indicates the site for the measurement of the skin moisturising. The dazed square zone indicates the site for the elasticity measurements.
the end of the daily application period on day 8 and then at the end of the study, on day 21.
Application site and expected effects on moisturising and elasticity were circumscribed to skin or vaginal mucosa. For this reason, the safety and tolerability assessments were limited to the spontaneous reporting of adverse reactions and to the subjective and objective assessment of local tolerability.
The study documentation was reviewed by the independent ethics committee of Canton Ticino and was conducted in compliance with the Swiss rule “Federal Law on Medicinal Products and Medicinal Devices (Law on Therapeutic Products LATer)” and in accordance with the Declaration of Helsinki and the general principles of ICH Harmonised Tripartite Guidelines for GCP. The study participants did not undergo any study procedure before signing the written informed consent form.
2.1. Study Parameters and Data Analysis
2.1.1. Skin Moisturising Measurement
The moisture retention capacity of the skin relates to the hydric dielectric constant [43] and can be measured in the stratum corneum at a depth of 10 - 20 µm. This ensures that the measurement is not influenced by capillary blood vessels. An increase in capacitance means an increase in skin hydratation.
Triplicate skin hydration measurements were performed at baseline (day 1) and post-dose on days 8 and 21 both in the abdominal right upper quadrant and in the perineal zone. The zone of application of the Corneometer® CM 825 probe was near the centre of the perineum, below the vagina (see Figure 1, diagonal bar zone). The same zone was kept for all the measurements. The instrument software calculated the mean capacitance value of each triplicate measurement.
2.1.2. Skin Elasticity Measurement Using the Cutometer® MPA 580 Probe
Single elasticity measurements were performed at baseline (day 1) and on days 8 and 21 both in the abdominal right upper quadrant and in the perineal zone in an area different from that chosen for the moisturising measurement (see Figure 1, dazed square zone). The same zone was kept for all the measurements. The Cutometer® built-in probe applied to the skin surface originates a vacuum (a negative pressure of 450 mbar was applied) that sucks the skin into the probe. After 2 s of suction, the negative pressure is instantaneously interrupted thus allowing the skin moving back out of the probe and relaxing for 2 s. The movements of the skin inside the probe (elongation and retraction) are measured by a glass prism according to the principle of light ray interruption.
The cutometer generates an elongation and a retraction curve depicting each immediate skin deformation (see Figure 2). The amplitudes (from R0 to R8) and the areas (F0 and F1) measured by the instrument, while constructing the curves, are indices which allow the interpretation of the viscoelastic properties of the skin. The parameters obtained through the viscoelasticity measurements are listed in Table 1.
Mode 1 was applied to all study measurements. Each measurement had a duration of at least 4 s, including 2 s of suction and 2 s of relaxation.
2.2. Statistical Analysis
The values measured on days 8 and 21 were compared vs. day 1 by a paired t-test (or by a Wilcoxon signed-rank test in case of lack of normality) with a 0.05 two-sided significance level.
VAS values were described using classic descriptive statistics and were compared vs. baseline (day 1) using a paired t-test (or by a Wilcoxon signed-rank test in case of lack of normality) with a 0.05 two-sided significance level.
Questionnaire items were summarised by presenting tables.
2.3. Safety Variables
Safety variables included the local tolerability on the abdominal and perineal skin and vaginal mucosa evaluated by the dermatologist, the gynaecologist and the study participants, the recording of adverse events (AEs)

Figure 2. Example of a skin elongation curve obtained after 1 cycle of skin suction-relaxation done in mode 1 using a Cutometer® MPA 580 probe.

Table 1. Linear and superficial parameters derived from the skin deformation curve by measuring the skin viscoelasticity.
and the physical examination.
Both the dermatologist and the volunteers scored the product local tolerability at the application sites of abdominal and perineal skin on days 8 and 21. The local tolerability of the product at the application site of the vaginal mucosa was also scored by both the gynaecologist and the volunteers. For all the evaluations, the following 5-point scale was used:
1 = excellent (no skin reaction);
2 = good (small skin reaction that spontaneously resolves);
3 = moderate (skin reaction tolerated with difficulty by the subject);
4 = poor (skin reaction needing interruption of treatment);
5 = bad (serious skin reaction).
The subjective evaluation was done as response to the dermatologist’s or gynaecologist’s question: “How do you judge the skin tolerability of the product?”.
3. RESULTS
3.1. Analysis of Moisturising of the Vaginal Mucosa Evaluated by Subjects
Mean VAS values given by the study participants to evaluate the moisturising of the vaginal mucosa on days 1, 8 and 21 and their change from baseline are summarised in Table 2.
Study subjects generally perceived an increase in moisturising of the vaginal mucosa from day 1 to 8. The VAS value increased more than twice as much as the baseline on average. From day 8 to 21, the moisturising of the vaginal mucosa increased less markedly. The change from baseline was statistically significant at both day 8 and day 21.
3.2. Evaluation of Skin Moisturising: Capacitance
For each triplicate measure of capacitance, one mean value calculated by the instrument software was recorded. Capacitance values and their change from baseline are summarised in Table 3. In comparison with baseline, the increment of capacitance was statistically significant both on day 8 and on day 21 only in the abdominal area (p-values < 0.0001 and 0.0119).
The increment observed in the perineal area was not statistically significant.
3.3. Skin Elasticity Measurement
Mean elasticity measures taken at the perineal and abdominal application sites with statistically significant changes from baseline are summarised in Table 4 for baseline, day 8 and 21 respectively.
An improvement in the elastic properties of skin was denoted by few significant changes. In detail, R1 de-

Table 2. Mean ± SD of visual analogue scale (VAS) value (mm) for vaginal mucosa moisturising on days 1, 8 and 21, the change from day 1 to day 8 and day 21 with the corresponding statistical test outcomes.

Table 3. Mean ± SD capacitance for abdominal and perineal skin moisturising on days 1, 8 and 21, the change from day 1 to day 8 and day 21 with the corresponding statistical test outcomes.

Table 4. Mean ± SD measures of skin elasticity taken on the perineal and abdominal skin on days 1, 8 and 21, the change from day 1 to day 8 and day 21 with the corresponding statistical test outcomes.
creased significantly from day 1 to day 8 and R8 decreased significantly from day 1 to day 21 in the perineal region. Indeed, both parameters are inversely proportional to skin elasticity and their decrement denotes an improvement in skin elasticity. With respect to the abdominal region, a significant increase in R0 also denoted some improvement in skin elasticity. The parameter continued to increase slightly from day 1 to 8 and then to 21.
3.4. Analysis of Use Comfort
The product comfort was evaluated positively by the totality of the subjects with respect to most of the asked questions. In detail, all the subjects could easily spread the cream without flowing or greasing or sticking or maculating. With respect to the smell, the 80% of subjects did not perceive any smell, while the 20% perceived a pleasant smell.
The 45% of subjects perceived that the vaginal hydration feeling lasted enough time, while the 55% found that the hydration feeling lasted for too long time.
3.5. Adverse Events
Generally, the treatment was well tolerated by the subjects. The occurrence of side effects was low. In fact, reproductive system disorders affected 3/20 subjects (15%) with occurrences of vulvovaginal burning sensation and of vulvovaginal discomfort. No other side effects judged as related to the application of the investigational cream occurred.
3.6. Local Tolerability
Local tolerability evaluated by the dermatologist at the perineal and abdominal skin, by the gynaecologist at the vaginal mucosa and by the volunteers themselves for all 3 regions is presented in Table 5.
Local tolerability of the cream was excellent at both assessment time points according to both dermatologist’s and gynaecologist’s evaluation. The 95% of the women rated the local tolerability as excellent. The volunteers, who rated the local tolerability of the product as good (10% on day 8 and 5% on day 21), experienced some adverse reactions at the reproductive system. The woman, who evaluated the local tolerability of the test product as good on days 8 and 21, experienced a vulvovaginal burning sensation starting from the 9th application up to the last application. Another woman, who evaluated the local tolerability of the test product as good on day 8, had experienced a vulvovaginal burning sensation on the first 3 days of application. After the 3rd application the adverse reaction resolved spontaneously.
4. DISCUSSION
The present study demonstrated the safety and the tolerability of a marketed gynaecological cream. The potential effects of the cream on vaginal mucosa and perineal and abdominal skin moisturising and viscoelasticity were also investigated. The study results showed a significant improvement in the moisturising of the vaginal mucosa and the perineal and abdominal skin.
The improvement in the conditions of the application areas was apparent from the objective epidermic hydration measurements on one hand and from the subjective judgement on the other. The topical hydration increased with the daily applications of the cream both at the perineal and the abdominal application sites, as shown by the slight increase in the values of capacitance in the stratum corneum. The change vs. baseline was more marked after 8 days than after 21 days. The improvement in skin moisturising was statistically significant at the abdominal skin application site. The results of the present study are consistent with the literature data [37-40, 44-48]. In particular, the results are consistent with those of Li et al. [44], who also showed that the measures taken one hour of the application are predictive of the stratum corneum moisturising obtained after a multiple applications for one or 2 weeks. Similarly, Wanitphakdeedecha et al. [48] showed, consistently with the results of the present study, that a cream containing 0.1% mucopolysaccharide polysulphate improves the hydration of the face skin after multiple applications for 28 days and of the skin of the volar side of the forearm after one application.
On average, an improvement in the moisturising of the vaginal mucosa was observed according to the volunteers’ evaluation from day 1 to 21 in the present study. In particular, the volunteers judged that the moisturising of the vaginal mucosa increased more than twice as much as the baseline with daily applications of the cream for the initial 8 days. Afterwards, the moisturising continued to increase up to day 21, though less markedly. The improvement in vaginal moisturising evaluated through a VAS was statistically significant (p-value < 0.0001) both on day 8 and 21 as compared with baseline.
Results obtained for perineal and abdominal skin viscoelasticity were consistent with previously published data of other similar studies [29,45-48]. In the present study, small changes in the skin elasticity parameters were observed in both perineal and abdominal skin, but their majority did not have statistical significance. A small significant improvement in the elastic properties was observed for the perineal region (R1 and R8 decrease) and secondarily also for the abdominal region (R0 increase). Interestingly, the mean values of R6 and R7 found by Elsner et al. [29] for the vulvar and forearm skin in untreated pre and post-menopausal women are consistent
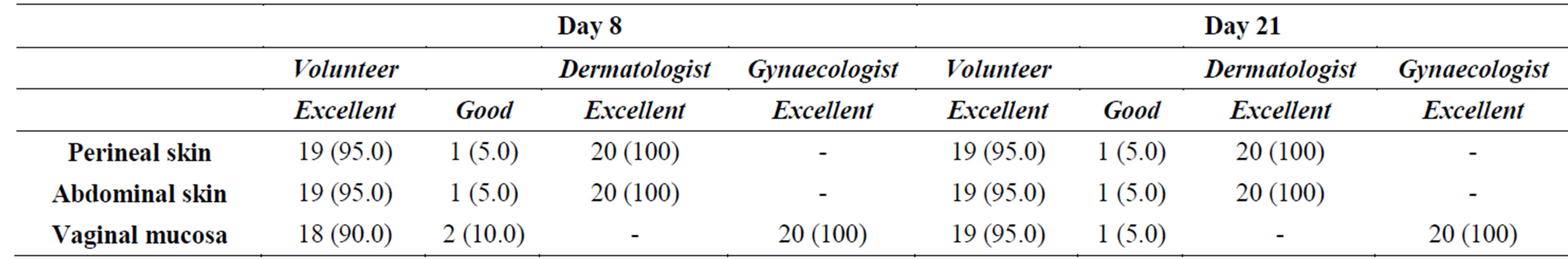
Table 5. Local tolerability of the cream applied to the perineal and abdominal skin and to the vaginal mucosa according to the objective evaluation of the dermatologist and the gynaecologist and the subjective evaluation of the volunteers. Number and percentage of subjects are presented [N (n%)].
with the results obtained in the present study for the perineal and abdominal skin, respectively. Generally, the use of the cream was judged comfortable by the majority of the volunteers with respect to the majority of the questions concerning the use comfort. Vaginal, perineal and abdominal tolerability of the cream was excellent at both assessment time points according to both dermatologist’s and gynaecologist’s evaluation. Vaginal, perineal and abdominal tolerability was excellent also for almost all the volunteers (90% - 95%). Only 3 of them experienced vulvovaginal burning sensation and vulvovaginal discomfort. Among the reported adverse events, only 10 episodes were judged as related to the cream use at a frequency of 15%. In conclusion, the tested cream represents a safe product with proven moisturising effect towards the vaginal mucosa and the perineal skin when applied daily by menopausal women.
ACKNOWLEDGEMENTS
A.C.R.A.F. S.p.A., Italy, gave the financial support to the project. The relationships between the Sponsor, A.C.R.A.F. S.p.A. and CROSS Research S.A., the dermatologist and the gynaecologist were regulated by financial agreements.
The Sponsor reviewed and approved the study design, was informed about the collection of data and reviewed and approved the analysis and the interpretation of data. The Sponsor also reviewed and approved the manuscript for publication.
MMR, AME and GC reviewed and approved the design of the study, were responsible for the clinical activities and collected the data, AV performed the statistical analysis, FF and ES proposed the study design and reviewed the draft manuscript, AFDD wrote the clinical study report and drafted the manuscript.
All authors read and approved the manuscript.