1. Introduction
Ruminant production in the tropics is hampered by scarcity of protein and energy rich feedstuffs especially in the dry season. This is so because tropical forages are generally of low quality and their intake by animals is usually limited by high levels of fibre and low nutrient content, particularly protein which is essential for both animal and rumen microbial growth [1] . The use of multipurpose trees (MPTs) as fodder for livestock is a way of making up for the nutrient limitation of the forages. It is an option for increasing the quality and availability of feed resources for livestock in the arid areas [2] . MPTs contain high levels of crude protein and minerals and many show high levels of digestibility [3] and are potential sources of cheap feed for ruminant animals, especially during dry seasons [4] . They maintain higher protein and mineral contents during growth than do grasses, which decline rapidly in quality with progress to maturity [5] . Currently small-holder farmers of Sub-Saharan African countries [6] are increasingly relying on various potential MPTs that can provide a green feed throughout the year which may be particularly useful as feed supplements to the typical low-quality diets [7] . However, many have been found to contain antinutritional factors which tend to affect both their intake and digestibility [8] .
Fungal treatment has been used to improve nutritive value of antinutritional factors (ANFs) containing feeds. The solid state fermentation has been shown to improve the nutritive value and sensory values of a wide variety of legumes [9] . Degradation of phytic acid by Rhizopus oligosporus [10] and tannins by A. niger has been reported [11] [12] . However, information on the mineral profiles and in sacco degradation of biodegraded products for ruminants is scarce.
At the ruminal level, optimum concentration of minerals is required for microbial growth, protein synthesis and cell wall degradation [13] . Minerals catalyze several enzymatic processes and hormone systems [14] maintain acid-base balance, water balance and osmotic pressure in the blood and cerebral spinal fluids [15] . However, rarely can pasture forages completely satisfy all mineral requirements [15] [16] . Tropical forage is often poor in calcium and especially phosphorus therefore care must be taken to supply the requisite amounts of these minerals [17] . Inadequate mineral intake leads to low productivity [18] . A knowledge of the mineral profile and effect of fermentation of MPTs becomes necessary.
Since the rumen is the primary site of digestion of forages, it is important to monitor their degradation kinetics [7] . The rumen degradation using the nylon bag technique is a major determinant of forage quality [19] . The technique is quicker and cheaper than the whole animal studies [7] . It is useful in screening ruminant feeds; in particular forage plants, for the proportion of rumen degradable nutrients (RDN) and undegradable dietary nutrients (UDN) [20] . In this study therefore, the effects of solid state fermentation on the mineral profile and in sacco degradation characteristics of dry matter in Enterolobium cyclocarpum (EC) and Pterocarpus santalinoides (PS) were evaluated.
2. Materials and Methods
2.1. Location and Forage
The study was conducted at the livestock farm of the international Livestock Research Institute (ILRI), Ibadan, South Western Nigeria. Biodegraded EC and PS leaves were used. The leaves were previously inoculated with A. niger and incubated at 30˚C for five periods (0, 7, 14, 21 and 28 days). A. niger was originally isolated at the Department of Yam pathology, International Institute of Tropical Agriculture and maintained on potato dextrose agar (PDA) slants [21] . After each incubation period, samples were oven dried at 60˚C, allowed to cool and ground for mineral analysis and in sacco dry matter degradability. Samples for mineral analysis were ground to pass 1mm sieve while those for dry matter degradability were ground to pass through 2.5 mm sieve.
2.2. Mineral Analysis
Analysis for Mg, Mn, Ca, Fe and Zn were determined by air acetylene flame absorption spectrophotometry on a Perkin Elmer model 703. Sodium, Potassium and Calcium were determined using the phlame photometer, while P was by use of autoanalyser 11 Technicon. All analyses were done in triplicate [22] .
2.3. Animals and Feeding
Three fistulated West African Dwarf (WAD) rams (Ovis aries) were used. Each animal was fed Guinea grass and supplemented with 2 kg wheat per day with water and salt licks provided ad libitum.
2.4. Rumen degradability of MPTs
The dry matter degradation was determined using the nylon bag technique [19] . Five grams of sample was emptied into nylon bag (Polymon, Switzerland) with pore size of 4 µ and effective size 5 cm × 18 cm. The bags were incubated in the rumen for 6, 12, 24, 48, 72 and 96 incubation periods. Bags were withdrawn at the end of each incubation and washed under running water until rinse water remained clear. The zero time washing loss was estimated by washing under tap after soaking in water at 37˚C for 1 hour. After washing, bags were oven dried in forced air oven at 60˚C for 48 hr, left at room temperature for at least 2 h before weighing residue. The results of dry matter disappearance from nylon bags were fitted into the exponential formula of [19] as follows:

where p = degradability at time t;
a = water soluble fraction (%);
b = water insoluble but potentially degradable fraction (%);
c = rate at which b is degraded per hour (rate constant);
t = incubation time (h).
Effective degradability of dry matter was calculated by using the following equation
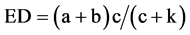
where k is the outflow rate (%).
2.5. Statistical Analysis
Results of effects of period (0, 7, 14, 21 and 28 days) of fermentation on mineral elements (Ca, Cu, Fe, K, Mg, Mn, Na, P and Zn) were analysed as a complete randomized design using ANOVA. The degradation characteristics were analysed as a randomized complete block design with three replicates (fistulated sheep) using NONLIN procedure [23] .
3. Results and Discussion
The mineral concentrations of biodegraded EC samples were affected by incubation period and are presented in Table 1. Mean values of Ca, Cu, K, Na and Zn were significantly increased (0.05), while mean values of Fe, Mg and Mn were first reduced (day 0) before significantly increasing (0.05). However, the composition of P showed significant reduction (0.05), up to day 28 where no change was observed. Mineral concentrations did differ appreciably from mean day 0 values for Ca (0.91%), Cu (9.04%), Fe (76.20%), K (1.16%), Mg (0.53%), Mn (81.14%), Na (53.45%), P (0.14%) and Zn (27.78%) respectively.
Mineral concentrations in biodegraded PS were also affected by incubation period (Table 2). Values of Ca, Fe, K, Mg, Mn, Na and Zn were significantly increased (0.05). However values of Cu and Fe only increased
![]()
Table 1. Changes in mineral composition of biodegraded EC with incubation time (%).
Means along the same column with any identical superscript are not significant (P > 0.05). (a) Ca = Calcium; K = Potassium; Mg = Magnesium; P = Phosphorus; Cu = Copper; Fe = Iron; Mn = Manganese; Na = Sodium and Zn = Zinc.
![]()
Table 2. Changes in mineral composition of biodegraded PS with incubation time (%).
Means along the same column with any identical superscript are not significant (P > 0.05). (a) Ca = Calcium; K = Potassium; Mg = Magnesium; P = Phosphorus; Cu = Copper; Fe = Iron; Mn = Manganese; Na = Sodium and Zn = Zinc.
after day 14, while the value of P was reduced significantly over the different incubation periods. Calcium concentration in PS increased by day 7 (32.56%) and by day 14 (42.38%) further percent increase was had but at day 21 (36.03) lower percentage than day 28 (41.22%).
The Ca concentration for EC at day 0 was higher than the dietary requirements of dairy cattle (0.43% - 0.60% of DM of diet) recommended by NRC [24] and within 0.09% - 3.35% [18] for various forages but lower than 1.01 for Ahdatoda vessica and 2.7 for Impashion bicolor [25] . The P in EC was low (0.14) in comparison with the required minimum of 0.20% [26] it was however within the range of 0.11 - 0.88 reported for various forages [18] but higher than 0.016 in Impashion bicolor [24] . The Ca to P ratio EC was about 7:1 at the brink of what ruminants can tolerate [27] . [25] [28] also observed wide Ca:P ratios among shrub species. The Ca in PS at day 0 were lower (0.87) than that of EC but higher than the requirement for dairy cattle (0.43 - 0.6 of DM of diet) recommended [24] . The P value (0.19) for PS however, was higher than that of EC but below required minimum of 0.20% [26] . The Ca:P ratios for EC (7:1) and PS (5:1) appear to corroborate reports of [29] that trees are rich in Ca and poor in P. [4] also recorded Ca contents of recommended levels but lower P contents for MPTs studied.
The variations in mineral concentrations of biodegraded EC and PS reflect differing effect of incubation periods with A. niger. Both biodegraded EC and PS concentrations of Ca, K, Na, and Zn increased as incubation period was further increased. Concentration of Fe, Mg, Mn and P in the biodegraded EC reduced after seven days incubation periods. In PS, reductions were of Cu, Fe and P, as in EC reduced with further increase in incubation period. The decreased concentrations suggest that the fungus may need the elements to meet specific requirements. The increased concentrations indicate increase in the microbial populations and production of the elements. Rumen microbes have capabilities to extract and concentrate essential minerals [30] and may be the case of the fungus used. Also fungal treatment can lead to release of locked up minerals from breakdown of cell wall components and other fibrous constituents from biodegraded materials. Fodder trees and Shrubs have always played a role in feeding Livestock [18] . They are good sources of minerals for ruminants [31] and often much higher in mineral contents than forages. However, continued reduction of P concentrations in both biodegraded EC and PS might be limiting to both rumen microbes and the animal requirement for P. The trend suggests that A. niger has preferential demand for P. Mineral content such as sulphur and phosphorus, is one of several factors that govern ruminal microbial yield [32] . Studies from tropical countries have indicated that medium to high levels of production at very high feed conversion efficiencies can be achieved by ruminants on poor quality forages adequately supplemented with critical nutrients [33] . Although P in biodegraded EC and PS may not reach critical levels, the reduction may create a demand for supplementation. In addition, high Ca:P ratio reduces absorption of P [24] . Therefore, P supplementation appears to be essential [34] .
4. Rumen Degradation
The dry matter degradation constants for biodegraded EC and PS are presented in Table 3. The water soluble fraction (a) of biodegraded EC for days 0, 7, 14, 21 and 28 were 19.68, 26.72, 25.14, 27.19 and 28.56, respectively. The soluble but potentially degradable fractions (b) decreased after day 7 (31.11%), increase in incubation period only caused reduction in b. The rate constant (c) of biodegraded EC was highest at day 0 (50.79%) but not significantly different from potential degradability of longer incubation periods to day 28 (48.68%). Effective degradability (ED) was highest for day 28 (36.71%) and not significantly different from days 21 (34.83%) and 7 (35.71%) but significantly different from days 0 (31.32%) and 14 (31.75%) respectively.
The degradability constants for biodegraded PS differed significantly (0.05). The soluble fraction (a) at day 0 (19.71%) did not differ significantly from day 28 (19.15%) but differed significantly (0.05) from days 7 (20.86), 14 (22.35) and 21 (21.55) respectively. The insoluble but potentially degradable fraction was highest at day 0 (48.55%) and significantly (0.05) different from day 21 (14.64%) but not different from days 7 (23.31%), 14 (32. 54%) and 28 (40.64%) respectively. Rate constants (c) for biodegraded PS for day 0 (0.010573%) did not significantly (0.05) differ from days 7 (0.013612%), to 28 (0.011056%) however days 14 (0.007171%) significantly differed from day 21 (0.020637%). Potential degradability (PD) for biodegraded PS was highest at day 0 (68. 26%) and only differed significantly (0.05) from day 21 (36.19%). The effective degradability ED was also highest for day 0 (31.39) samples and significantly differed from other incubation periods, further increase in period only showed lower but similar ED values for days 7 (27.52%), 14 (26.76%), 21 (27.42%) and 28 (26.86%) respectively.
The dry matter degradability of both EC and PS was affected by length of incubation with A. niger. Mean values for potentially degraded fractions of EC were similar but significantly (0.05) different from day 0. The PD value for day 0 was high with a low ED value. Though 21 day biodegraded EC had highest PD with significantly (P < 0.05) high ED, 28 day biodegraded EC had highest ED with fastest degradation rate. The slowest degradation rate was obtained with 28 days biodegraded EC but there was no difference.
Rumen degradation is useful in ranking MPTs in terms of nutritive value [35] [36] and for comparing the digestive capacities of ruminal species [37] . PS has been ranked low compared to other MPTs [38] [39] . In this study, PS had higher PD than EC. The insoluble but degradable fraction b was 48.55% in PS and 31.11% in EC leaves; similarly, the greatest and least values of PD were recorded for PS (68.26%) and EC (50.79%), respectively. Biodegradation resulted in lowering the PD values. 21 day biodegraded PS with significantly (P < 0.05) lowest PD degraded least. In spite of the high PD values, PS comparatively degraded faster than EC. The degradability of PS was higher than range 29% to 34 % reported for Calliandra calothrysus [40] but similar to those obtained by [20] for four MPTs foliage using WAD goats.
The degradability constants varied appreciably. Variabilities in rumen degradability constants have been reported [7] [39] [41] - [43] . The possible reason could be linked to variation in plant species [7] , their chemical composition and cell wall characteristics and their effects on rumen ecology in various ruminant species [44] .
![]()
Table 3. Dry matter degradation constants for biodegraded substrate of EC and PS.
Means along the same column with any identical superscript are not significant (P > 0.05). A = water soluble fraction (%); B = water insoluble fraction (%); C = rate at which b is degraded (rate constant); PD = potential degradability (%) and ED = effective degradability (%).
Cellulose in plants is composed of amorphous and crystalline structures [45] , the latter more resistant to microbial degradation [46] . Enzymes secreted by aerobic and anaerobic fungi are capable of solubilising crystalline cellulose [47] . However, in fungal decayed material, where part of the lignin was already removed by the fungi, the limitation in the digestion of structural carbohydrates may be due to other factors such as crystallinity of cellulose [46] [47] . Variation in rumen degradation characteristics may also be attributable to cell wall polysaccharide configuration in the different species and their effect on rumen microbial attachment and colonization of digesta particles [48] .
Degradation constants as measured by the in sacco nylon bag technique are strongly related to digestible DM intake [49] . Thus, interspecies variations in OM degradability could result in different intakes of the plants when given as sole diets to animals [7] .
5. Conclusion
In this study degradation showed improvements in PD and ED values of fungal biodegraded EC and reduction in PS, suggesting improved quality of EC. However, animal output is a better reflection of forage quality. Further research on animal performance was therefore necessary to determine whether the high PD and ED values in EC could be translated into improved animal output.