Received 9 March 2016; accepted 14 June 2016; published 17 June 2016

1. Introduction
Malaria remains to be a health and economic problem in the tropical Africa and the world at large. In Africa malaria is responsible for over 430,000 children deaths every year [1] . In the year 2013, the World Health Organization (WHO) estimated 198 million cases of malaria leading to 584,000 deaths worldwide [1] . The disease mostly prevails in the poor countries of tropical and sub-tropical regions of Sub-Saharan Africa, America and Asia [2] - [7] . The Plamodium species namely P. falciparum, P. vivax, P. malariae and P. ovale, and the lately reported species P. knowlesi are known to cause malaria [4] [5] [8] [9] . Of the five species P. falciparum is the most responsible for causing the disease [2] [3] [5] [7] .
Over the recent past years, chloroquine was used to treat malaria [4] [8] . Artemisinin and its derivatives were later discovered and took over to treat the disease [3] . However, the occurrence of resistant strains of Plasmodium to chloroquine and artemisinin based drugs [3] [4] [8] , has necessitated the search and development of new drug targets to combat the disease. Development of resistance to antimalarial drugs is associated to mutations in the parasite active site for the drug target [3] [4] [8] . Thus, identifying new drug targets with new mechanisms of action of the drug may help in fighting the disease [4] [8] . Today, many other natural products and synthetic anti-malaria agents have been designed to target different enzymes involved in parasitic life cycle [6] [10] - [15] .
Enzymes of the glycolytic pathways are thought to be an important drug target due to parasitic dependence on glycolysis for energy production [3] [4] . Plasmodium falciparum Lactate Dehydrogenase (PfLDH) enzyme is involved in the final step of glycolysis and catalyzes the interconversion of pyruvate to lactate [3] [4] . The enzyme LDH is further involved in the formation of NAD+ which is required for the glycolytic enzyme glyceraldehyde-3-phosphate dehydrogenase [4] . P. falciparum parasites depend on this enzyme for energy production needed for biochemical process, growth and development. Thus, this enzyme is thought to be an important drug target in malaria treatment. Inhibition of this enzyme activities results in death of the parasite. Thus, some anti- malarial drugs have been designed to target this enzyme. Thus, efforts to search for new potent anti-malarial drugs have been a constant ambition to scientists in this field of research.
In a search for potent anti-malarial compounds, Ngadjui and co-workers isolated anti-malarial compounds namely hoslundin, hoslundal and hoslunddiol from Hoslundia opposita (Figure 1) [16] . The compounds showed anti-malarial activity against multi-drug resistant K1 strain of P. falciparum, hoslundal being the most active [16] . An interesting anti-malarial activity of hoslundal suggested it to have a different mode of action from chloroquine and other anti-malaria drugs [16] . H. opposita is traditionally used in the treatment of malaria and other diseases in East and West Africa [16] [17] . Several research groups have also reported the anti-malarial activities of the compounds from H. opposita [17] - [21] . However, the in silico assessment of molecular interaction of these compounds with PfLDH enzyme remains unreported. In this work, a molecular docking study was carried on hoslundin, hoslundal and hoslunddiol to evaluate their potential inhibitory activity against PfLDH enzyme. The binding interactions were investigated and are reported herein.
2. In Silico Experimental Procedures
The three dimensional protein crystal structures of PfLDH (PDB ID: IT2C) with resolution of 2.01 Å, (Figure 2(a)), [22] was obtained from the RCSB Protein Data Bank and used for docking studies. Docking experiments
![]()
Figure 1. Structures of anti-malaria compounds from Hoslundia opposita [16] .
![]()
![]()
![]()
Figure 2. Structure of PfLDH enzyme (PDB ID: IT2C) retrieved from the RCSB Protein Data bank as shown in (a); (b) shows the binding surface pockets present in PfLDH enzyme. Site A is the cofactor binding pocket as viewed in PyMol software; (c) shows the amino acids binding pocket in blue colour near the surface of the enzyme.
were performed following the procedure reported in the literature by [23] [24] . Briefly, Protein binding pockets sites were analysed by using of 3D Ligand Binding Site Predication Server. Possible proteins clashes and amino acid from the active sites were checked by PyMol software v 0.9 (DeLano Scientific LLC). The protein crystal structure was cleaned by using Arguslab software by deleting all heteroatoms, cofactor and water in the protein active sites. Hydrogen atom was added to the geometry. The crystal structure was converted to PDBQT, then refined and geometries optimized using AutoDock Vina in PyRx tools to generate atomic coordinates. Ligands used in this study are chemical structures of hoslundin, hoslundal and hoslunddiol which are anti-plasmodial compounds isolated Hoslundia opposita [16] . A list of ligands was generated and optimized using ChemDraw Ultra 12.0 and ChemDraw 3D Pro. The optimized geometry structures were served in .pdb file format. Ligands were then converted to PDBQT in PyRx. Finally, ligands were automatically docked to PfLDH enzymes using AutoDock Vina in PyRx (v 0.8) virtual screening tool with 8 ligand exhaustiveness. PyRx employs Lamarckian Geometric Algorithm (LGA) in docking processes; ligands were docked using flexible conformation. Ligands were re-docked three times in the active sites. Docking grid from autogrid with dimensions 25 × 25 × 25 Å size was used. A default vina search space grid dimensions of 26.09, 26.99 and 9.106 for x, y, z, coordinates was used, respectively. PyMol v 0.9 software (DeLano Scientific LLC,) was used to visualize the binding sites and orientations of the complex.
3. Results and Discussion
3.1. Binding Pockets Analysis
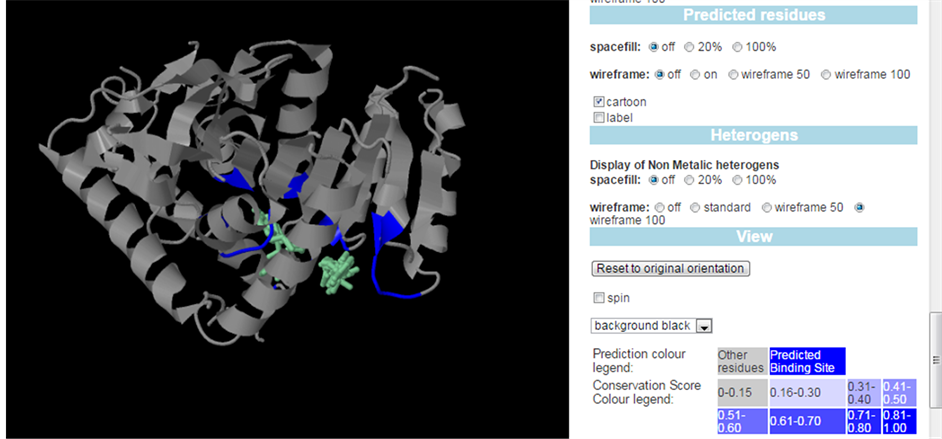
The PfLDH enzyme possesses two important binding pockets, the cofactors binding pocket (Site A) and Site B (Figure 2(b)) [4] . Site A, identified as NADH binding pocket comprised of amino acid residues: Gly29, Met30, Ile31, Phe52, Asp53, Ile54, Val55, Tyr85, Thr97, Ala98, Thr101, Val138, Thr139, Asn140, Val142, Leu163, Leu167 and Pro250 (Appendix). Site B comprised of amino acid residues: Asp230, Lys198, Val233, Lys314, Glu317, Asp230, Leu201, Glu226, Phe229, Val200, Leu237, and Asn241. These amino acid residues were near the surface end of enzyme (Figure 2(c)) [4] . The presence of NADH cofactor affected the distribution and conformation of the docked ligands. It was observed that, in the presence of the cofactors all ligands bound to site B, while in the absence of the cofactor all docked ligands showed a stable conformation at site A.
3.2. Molecular Docking
All ligands were successfully docked to binding sites of PfLDH enzyme. All ligands showed better docking score with stable conformation than the crystalized NADH cofactor when docked in the absence of the cofactor (in the cofactor binding site). In the absence of cofactor, all ligands bound to site A which is the binding site of the cofactor. When ligands were docked in the presence of cofactor, site B was the preferred binding site for all ligands and possessed lower docking scores (Table 1).
In the present study, the anti-malarial compound hoslunddiol from Hoslundia opposita had a good docking score (−8.0 kcal/mol) higher than all the other ligands to PfLDH enzyme. The best docking score was obtained in the absence of the cofactor (Table 1). When re-docked in the absence of the cofactor, similar binding affinities were obtained (Table 2). The interaction of hoslunddiol with amino acid residues of PfLDH involved four hydrogen bonds which were formed as follows: The first hydrogen bond was formed by the oxygen atom of the ligand (Figure 3(d)). The second hydrogen bond was formed by the hydroxyl group bonding with -NH2 of Met30 (HO----Met30, 2.38 Å). The third and fourth hydrogen bonds involved oxygen of methoxy group (MeO ----Thr97, 3.22 Å), carbonyl group (C=O----Thr97, 3.54 Å) and oxygen atom of the pyran ring (-O----Thr97, 3.30 Å). Figure 3(e) shows the binding of hoslunddiol in the PfLDH active site A. The docking results showed hoslundin to have the second docking score rank with −7.8 kcal/mol. Docking of hoslundin involved four hydrogen bonds between PfLDH and oxygen of the ligand (Figure 3(b) and Figure 3(c)). The first hydrogen bond formed between C=O of Gly99 (C=O----Gly99, 3.06 Å) while the second hydrogen bond was formed between -NH2 of Asn140 (C=O----Asn140, 3.05 Å). The hydroxyl (OH) group in the ligand formed the third hydrogen bond with -NH2 group of Phe100 (HO----Phe100, 3.02 Å) whereas the fourth bond was formed between amine group of Thr97 with the carbonyl group in the ligand (C=O----Thr97, 2.35 Å). Molecular docking studies of hoslundal showed only two hydrogen bonds, which involved Thr101 (3.08 Å) and Asn140 (3.26 Å) (Figure 3(a)). Other amino acid residues which interacted with hoslundal in the PfLDH binding pocket were; Gly99, Thr97, Ala98, Gly27, Lys102, Ala103, Phe101, Met30, Ile31, Gy29, Gly32, Pro246, Asn116, Thr139, Ser28, Met36, Tyr247, Gly33, Ala251, Asp110, Leu112, Ser245 and Val248.
Docked ligands showed higher binding affinity than NADH in absence of the cofactor, implying a possible competitive inhibition. However, on closer examination, the interaction of NADH and PfLDH indicated many more hydrogen bonds than the ligands (Figure 4). Concomitantly, other studies have reported many hydrogen bonds formed between NADH and PfLDH, regardless of the lower binding energy obtained for NADH in the
![]()
Table 1. Best binding energy (kcal/mol) of the favourable conformation based on PyRx
![]()
![]()
Figure 4. Interaction of NADH to PfLDH (left) and binding position in the pocket (right).
![]()
Table 2. Best binding affinity on re-docking experiments
PfLDH enzyme active site [3] . The presence of a high number of hydrogen bonds may reduce the binding affinity of other ligands in the cofactor binding sites. Hydrogen bonds stabilize the ligand interaction in the active site [4] [25] . Docking of ligands in the presence of NADH reduced the affinity and frequency of binding to the cofactor active site. Such phenomenon has also been reported when ligands were docked in the presence of NADH [4] , the frequency and affinity of binding to the cofactors binding site was found to be low. In the present study, all ligands showed lower binding affinity than NADH when they were docked in the presence of the NADH (Table 1). However, in the absence of NADH all ligands showed higher docking score than NADH. This suggests competitive inhibition in the NADH cofactor binding site, thus, inhibiting PfLDH enzyme from producing energy [3] . Binding of ligands to site B may suggest allosteric inhibition [4] . The amino acid residues forming H-bonds and the bond length in the active site with hoslundal, hoslundin, hoslunddiol and NADH are presented in Table 3.
4. Conclusion
Plasmodium falciparum is an important malaria parasite in Africa and other parts of the world. The glycolytic PfLDH enzyme is thought to be one of the important anti-malarial drug targets due to the parasite dependence on glycolysis for energy production. The present study investigated the in silico inhibition effects of the three Hoslundia opposita compounds (hoslunddiol, hoslundin and hoslundal) to PfLDH enzyme. Results indicated that hoslunddiol (−8.0 kcal/mol) and hoslundin (−7.8 kcal/mol) had better interaction with the target. Hoslundal showed less binding energy (−7.3 kcal/mol) and had few interactions. The former two compounds showed four hydrogen bonds while the latter showed only two hydrogen bonds. Since hoslundal was reported to be the most active anti-malarial compound against the multi-drug resistant K1 strain yet weakly active against the chloroquine sensitive strain NF54 in vitro, the present findings further affirm that hoslundal possesses antimalarial activities with probable different modes of action. Within the limitations of the docking procedure, these findings suggest that the studied compounds could act as competitive inhibitors as they had higher binding energy than the cofactor. The pharmacokinetics and in vivo studies of these promising anti-malarial compounds are recommended. Furthermore, studies on the interaction and mode of action with other Plasmodium falciparum enzymes
![]()
Table 3. Amino acid residues forming H-bonds with their bond length in PfLDH active site
and any other life cycle target points are recommended for the studied and other anti-plasmodial compounds from H. opposita.
Acknowledgements
DMS express his sincerely thanks to the NAPRECA (T) for sponsorship and facilitation on In Silico studies during the 16th NAPRECA Pre-Symposium workshop. JJEM is grateful to the International Foundation for Sciences (IFS) for financial support.
Appendix
Predicated binding active site in the PfLDH enzyme by 3D ligand site prediction server.
![]()
Selected letter amino acid code sequence of PfLDH in docking server.
![]()
NOTES
![]()
*Corresponding author.