1. Introduction
Hypoxia is a well-known cause of cell injury at plain, with important pathological implications in many disease processes, including cerebral ischemia and myocardial infarction [1] . Cells in hypoxia expressed a variety of adaptive or death gene products to satisfy altered metabolic demands or to remove irreversibly damaged cells [2] . However, in the case of severe hypoxic damage beyond the cell’s adaptive capability, death- promoting genes are expressed, resulting in necrosis or apoptosis [3] .
Hypoxia-inducible factor-1α (HIF-1α), as a kind of transcriptional factor, is known to play a fundamental role in adaptive or death process in response to hypoxia [4] , it is induced, stabilized, and translocated to the nucleus to regulate the transcription of a variety of genes involved in adaptive responses, such as increased O2 delivery and angiogenesis [5] . Our previous study and other’s have demonstrated that the apoptosis rate of gut-associated lymphatic tissue (GALT) increased postburn (PB) with delayed resuscitation [6] - [8] , but the mechanism remained controversial. It was reported that CD3- positive human T cells accumulating in inflammatory tissue and expressing HIF-1α, indicating a role of hypoxia-mediated signals in regulation of T cell function. Surprisingly, accumulation of HIF-1α in human T cells required hypoxia but also TCR/CD3- mediated activation [9] . As the marker of T lymphocyte, CD3+ molecule expressed on the surface of matured T lymphocytes. The CD3+ molecule could stabilize the structure of CD3+-T cell antigen receptor (CD3-TCR) and transfer the signals of activation. There was no report about the effects of HIF-1α expression on the apoptosis and proliferation of T lymphocytes in Peyer’s patches PB at plateau. In this study, a burn model with delayed resuscitation was duplicated at sub-plateau (1517 m above sea level) and plateau (3848 m above sea level) and the effects of HIF-1α expression on the lymphocytes in Peyer’s patch was observed.
2. Material and Method
2.1. Animal Model
Total of 132 Wistar rats were supplied by medical experimental animal center of Gansu Chinese Traditional Medical College. The average weight was 200 ± 30 g. Half of them were female. Animal was kept in single cage under 19˚C - 25˚C for 1 week before injury. They were fasted 12 h before experiment. Animals were anesthetized with an injection of pentobarbital sodium (40 mg/kg). The back of the rats was shaved before experiment and was immersed into 90˚C hot water for 20 s to produce a 30% total body surface area (TBSA) scald burn on the back of the rats at 1517 m and 3848 m above sea level. The experiment at 1517 m altitude was carried out in the animal experimental center of Lanzhou General Hospital in Lanzhou city, Gansu province. The experiment at 3848 m altitude was carried out in an animal experimental base of high altitude medicine in Mahanshan mountain in Yuzhong county, Gansu province, China. Animals were transported from Lanzhou (1517 m above sea level) to Mahanshan mountain (3848 m above sea level) in 1 day and the experiment was started after 3 days acclimation, finished in 10 days. All animals were randomly divided into 6 groups by table of random digit: immediate fluid resuscitation group (IFR, n = 30 at each altitude, the animals received saline injection intraperitoneally according to the Parkland formula immediately after injury for resuscitation, 40 ml/kg body weight); delayed fluid resuscitation group ( DFR, n = 30 at each altitude, the same amount of fluid was given 6h PB; and sham group (SG, n = 6 at each altitude, the back of the animals were immersed into 37˚C warm water for 20 s to imitate scald burn without saline injection).
2.2. Sample Harvesting
At 6, 12, 24, 48 and 72 hours PB, animals (n = 6, at every time point in each group) were euthanized after being anesthetized with intraperitoneal injection of pentobarbital sodium (40 mg/kg). All the Peyer’s patches in the intestine (10 cm distal to the ileocecal junction) were harvested and were immediately fixed in 10% neutral buffered formalin. The samples in sham group were harvested 6 hours after sham burn.
2.3. Histopathological Observation of Peyer’s Patches
The Peyer’s patches were then embedded in parafin and sectioned. After stained with hematoxylin and eosin, histopathological observation and photographing were carried out under optical microscope.
2.4. The Detection of Lymphocyte Apoptosis in Peyer’s Patches
The terminal deoxynucleotidyl transferase (TdT)-mediated d-UDP-biotin nick end labeling (TUNEL) method was used to detect the apoptosis of lymphocytes in Peyer’s patches with modification [10] , using the cell death kit (Roche Company). After DAB coloration and hematoxylin counterstain, they were observed and photographed under microscope. The cell number in 6 high power fields (more than 1000 cells) were counted and the positive cells were identified. The apoptotic percentage was counted as following:
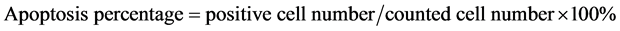
2.5. The Detection of Expression of HIF-1α and CD3+ by Immunohistological Method
The expression of HIF-1α and CD3+ protein from the Peyer’s patches were determined by Strept-Actividin-Biotin-Complex (SABC) method. The paraffin sections (4 µm) were dehydrated in xylene and graded ethanol series, and added in order with primary antibody [HIF-1α (rabbit polyclonal antibody, Wuhan Boster Co., China), CD3+ (rabbit anti-rat monoclonal antibody, Wuhan Boster Co.)], after incubated in a humid box overnight, biotinylated secondary antibody (rabbit anti-rat IgG, Wuhan Boster Co.) added and incubated for 20 min at 37˚C, SABC reagents and DAB solution (Wuhan Boster Co.) added. For negative control, the sections were treated with PBS instead of primary antibody. Immunohistological quantitative analysis was carried out as following: ten sample sections from each group were chosen for analysis. 6 high power visual fields were observed randomly. The Dpcontrollor 7.0 photographing system and the pro-plus 5.0/Ipp5.0 Image analysis system were used to determine the absorbance (A value) of HIF-1α and CD3+ protein.
2.6. Statistical Analysis
Results are expressed as means ± s. Group comparisons were carried out by variance analysis, using SPSS 10.0 medical statistical software. P-value less than 0.05 was considered significant.
3. Result
3.1. Result of Histopathological Observation of Peyer’s Patches
The construction of Peyer’s patches in SG at 1517 m was very clear. There was no inflammatory cell infiltration. The construction of Peyer’s patches was also clear in IFR group, only some germinal center of some follicle enlarged. There was light inflammatory cell infiltration 6 - 12 h PB. Inflammatory cell infiltration was observed in DFR group, apoptotic lymphocytes distributed in the germinal center singularly or areataly. The pathological changes aggravated gradually after 6 - 12 h PB.
The construction of Peyer’s patches in SG at 3848m was clear too, but both the B cell zone and T cell zone enlarged and the volume of Peyer’s patches increased too. There was a little inflammatory cell infiltration in IFR group, single apoptotic cell distributed in the germinal center, the membrane of apoptotic cell shrinked, the neuclous karypyknosis and margination of chromatin and round and ellipse apoptotic body were observed. The above pathological changes alleviated 48 - 72 h PB.
3.2. Immunohistochemistry Observation of Apoptotic Cell and Apoptosis Rate Change at Different Time Point (Table 1)
Apoptotic cells were observed in Peyer’s patches at two altitudes in experimental groups. The apoptotic cell number increased gradually 6 - 12 h PB and reached the zenith at 12 h PB. The nucleus of apoptotic cell showed brown color, the chromosome condensed, and distributed in the germinal center of Peyer’s patches clusterly (Figure 1).
![]()
Table 1. Comparison of lymphocyte apoptosis rate in Peyer’s patches in burned rats at two altitudes (ap%, 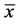 ± s).
± s).
Notice: a: vs SG at the same altitude, P < 0.05; b: vs IFR at the same altitude, P < 0.05; c: vs corresponding group at low altitude, P < 0.05; Blank means no data.
![]()
Figure 1. A lot of Tunel positive apoptotic lymphocytes were discerned in the germinal center of Peyer’s patch 12 h PB in IFR group at 3848 m altitude. The apoptotic lymphocytes showed deep brown color. TUNEL ×400.
Quantitative analysis of lymphocyte apoptosis rate after 6 - 24 h PB showed: 1) the apoptosis rate in DFR group was higher than that in IFR group at the same altitude 6 - 24 h PB (P < 0.05). The highest apoptotic rate in experimental group was at 12 h PB (P?0.05). 2) The apoptosis rate in both DFR and IFR group at high altitude was higher than that in low altitude (P?0.05) (Table 1).
3.3. The Change of HIF-1α Expression in Every Group at Different Altitude (Table 2)
The HIF-1α expression increased both in IFR and DFR group at 6 - 72 h PB, compared with that in SG at the same altitude. The positive expression mainly distributed in the nucleus of lymphocytes, showing a buffy colour granules (Figure 2). The HIF-1α expression in DFR and IFR group at every time point at different altitude were higher than that in SG (P < 0.05 all). The expression of HIF-1α at 6 h, 24 h, 72 h PB in DFR group at low altitude, at 6 h, 12 h, 24 h, 48 h PB in DFR at high altitude were higher than that in IFR group (P < 0.05). The HIF-1α expression in DFR group at 6 h, 12 h, 24 h, 48 h PB at high altitude were higher than that in groups at low altitude (P < 0.05).
3.4. The Change of CD3+ Expression at Different Altitude (Table 3)
Compared with the CD3+ expression at the same altitude in SG, the CD3+ expression decreased both in IFR and DFR groups, mainly distributed on the cell membrane of T lymphocyte, showing brow-yellow color. The quantitative analysis showed that the CD3+ expressions at both altitudes in DFR and IFR groups at different time point were lower than that in SG (P value all less than 0.05). The CD3+ expressions in DFR group at low altitude and in DFR group at 6 h, 12 h, 24 h, 48 h, 72 h PB at high altitude were all lower than that in IFR group at the same altitude. The CD3+ expressions both in DFR and IFR groups from 6 h to 72 h PB were all lower than that in corresponding low altitude groups (P < 0.05).
![]()
Figure 2. An amount of HIF
-1a
positive lymphocytes appeared in the paracortical area of the Peyer’s patch 12 h PB at 3848 m in DFR group, and the cells of positive expression showed brown color. SABC ×400.
![]()
Table 2. The HIF-1α expression in Peyer’s patch at two altitudes (A value 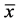 ± s).
± s).
a: vs SG at the same altitude, P < 0.05; b: vs IFR at the same altitude, P < 0.05; c: vs corresponding group at low altitude, P < 0.05, Blank means no data.
![]()
Table 3. CD3+ expression in Peyer’s patches after burn in rats at two altitudes (A value  ± s, n = 132).
± s, n = 132).
a: vs SG at the same altitude, P < 0.05; b: vs IFR at the same altitude, P < 0.05;c: vs corresponding group at low altitude, P < 0.05, Blank means no data.
4. Discussion
Immune cells are exposed to different oxygen tension, including hypoxia, as they develop, migrate and function in primary, secondary, and tertiary lymphoid organs with different infrastructure, vasculature, and oxygen supply [11] [12] . Hypoxia results in adaptationally appropriate alterations of gene expression through the activation of hypoxia-inducible factor-1 (HIF-1) to overcome any shortage of oxygen. HIF-1 is a basic helix-loop-helix/Per-ARNT-Sim protein consisting of HIF-1α and HIF-1β subunits. It was shown that the expression of HIF-1α, but not HIF-1β, is significantly enhanced by hypoxia [13] - [15] . HIF-1α expression in T lymphocytes can be induced by hypoxia [16] . T-cell adaptation to hypoxia is characterized by a modulation of cytokine and cell cycle protein expression, associated with an inhibition of T-cell activation and proliferation [17] [18] . Other reports demonstrated that hypoxia induces apoptosis in Jurkat cells, a lymphoblastic cell line [19] . An increase in HIF-1 transcriptional activity enhances apoptosis in thymocytes in vivo in mice that are deficiency in von Hippel Lindau (VHL) protein [20] .
Our previous study has demonstrated that the apoptosis rate of gut-associated lymphatic tissue (GALT) increased postburn (PB) with delayed resuscitation [6] [7] . To observe the relationship between HIF-1α expression and apoptosis and proliferation of lymphocytes in Peyer’s patches, animals were subjected to scald burn with delayed resuscitation at 1517 m and 3848 m altitude above sea level. In this model, the intestinal lymphocytes of the rats suffered from double hits of hypoxia and ischemia-reperfusion. The results showed that a positive expression of HIF-1α occurred at 6 h PB and reached the zenith at 12 h PB in DFR and IFR group, the expression was obvious especially in DFR group, revealed that a high expression of HIF-1α in Peyer’s patches could be induced by hypoxia and ischemia-reperfusion caused by delayed fluid resuscitation at plateau. The positive expression of HIF-1α paralleling to the change of apoptosis rate of lymphocytes in Peyer’s patches, suggested the involvement of HIF-1α in the induction of T lymphocyte apoptosis in Peyer’s patches.
There was a little expression of HIF-1α at 3848 m altitude in sham group; this revealed that hypoxia may be involved in the induction of apoptosis. The result is in agreement with the report that hypoxia could enhance the HIF-1α accumulation and apoptosis of T cells, and inhibit T-cell proliferation [21] .
CD3+ protein, as a marker of T lymphocytes, the trough value of CD3+ expression also appeared at 12 h PB, and the value in DFR group were all lower than that in IFR group at different altitude. And the lowest expression of CD3+ occurred at 12h PB. These results suggested that the apoptotic lymphocytes were mainly T cells. The expression of CD3+ protein was antiparalleled to the apoptosis of lymphocytes [6] [7] , suggested that the proliferation of lymphocytes in Peyer’s patches were decreased after burn and delayed resuscitation at plateau. It was reported that hypoxia could inhibit the proliferation of T cells [21] .
It is well known that hypoxia increase cellular ROS generation, probably from mitochondrial electron transport complexes [22] . It has been demonstrated that Noxa, a member of BH3-only Bcl-2 family proteins, is an HIF-1α-dependent hypoxia-inducible gene and mediates hypoxic cell death in an ROS-dependent manner. Suppression of Noxa expression by antisense oligonucleotides rescued cells from hypoxia-induced cell death and decreased infarction volumes in an animal model of ischemia [23] . Therefore, the mechanism which the expression of HIF-1α promotes the apoptosis of T lymphocytes in Peyer’s patches in this model might be related to the production of ROS caused by hypoxia and ischemia-reperfusion due to delayed resuscitation [24] .
ROS could stabilize HIF-1α and prevent the degradation of HIF-1α. The HIF-1α could be detected in T lymphocytes in sham group, because the half-life of HIF-1α was very short under normoxia. They were ubiliquitinated quickly after production, and then dissolved by protease [25] - [27] . Therefore, ischemia and hypoxia induced the expression of HIF-1α after severe scald with delayed resuscitation at high altitude, and aggravated the injury of T lymphocytes.
Funding
Supported by: The general program of medical science and technology of Chinese PLA during the 11th “five year” plan. Fund No: (06G030).