Received 20 February 2016; accepted 25 March 2016; published 29 March 2016

1. Introduction
Phalaenopsis orchids are popular and valuable potted ornamental plants worldwide for their beautiful and long- lasting flowers that have a variety of shapes, sizes, and colors. Flowering time must be precisely scheduled to meet market demands. One of the key steps in regulating Phalaenopsis flowering is to control the emergence of the flower stalk or spiking.
Phalaenopsis is a monopodial orchid with leaves alternating on each side of the plant and the bases of the leaves are connected to a shortened stem. One or 2 dormant flower stalk bud(s) emerges from the stem at the base of the third or fifth leaves that are numbered basipetally from the aerial portion of the plant [1] .
Light is an important environmental condition for plant reproductive development. It is commonly recognized that light quantity has a close relationship with photosynthetic performance. The photosynthetic products, sugars, act as signals to control the reproductive development of plants [2] . In Phalaenopsis, many authors emphasized that sucrose concentration in the source leaf was correlated with the breaking of dormant flower stalk buds [3] - [7] ; however, a conflicting observation has also been identified [8] - [10] . Although the leaf is the site of light signal perception, it is reasonable to speculate that the signal in the stem of Phalaenopsis is involved more directly to spike development than in the leaf because the flower stalk bud is connected with the shortened stem. Nevertheless, little is known about the relationships between carbohydrates, especially sucrose levels in the stems and spiking of Phalaenopsis.
Light quality is an important factor that regulates plant reproduction [11] . In response to low red/far-red (R/FR) ratio, flowering was accelerated in Arabidopsis [12] , but was delayed in soybean [13] . Flower bud formation of Petunia was achieved with FR light treatment but inhibited by red light treatment [14] . A red light supplement at the crown delays flower bud induction and reduces flowering percentage in strawberry [15] . Blue light promoted flowering of Chenopodium rubrum but its effect was counteracted by red light [16] . Clearly, the aforementioned results manifested the effect of light quality on flowering varied among different plant species.
In fact, the effect of light quality upon photosynthetic pigments such as chlorophyll a (Chl a), chlorophyll b (Chl b), and/or carotenoids (Car) contents was also species dependent. For example, the pigment content in Atractylodes macrocephala [17] , lettuce [18] , and Petunia [14] was not affected but in Marigolds and Salvia [19] , tomato [20] , cucumber [21] , or poinsettia [22] was significantly affected by light quality. Furthermore, in poinsettia, blue light increased Chl a content and promoted flowering but red light increased the contents of Chl b and delayed flowering [22] . In Arabidopsis, Chl b deficient mutants exhibit late flowering, whereas transgenic plant overproduces Chl b earlier flowering than the wild type [23] . However, there was not any correlation between chlorophyll content and flower bud formation in Petunia [14] . Different to the aforementioned plants, Phalaenopsis is an epiphytic orchid with thick, succulent leaves and exhibits a crassulacean acid metabolism (CAM) [5] [8] [9] [24] - [26] . The main objectives of this study are the impact of light quality on changes of dawn-to-dusk of photosynthetic pigment and carbohydrate concentrations in the leaf, the carbohydrates in the stem, and the nexus between spiking of Phalaenopsis.
Light-emitting diodes (LEDs) are a promising electric light source and have been used for different light regimes to demonstrate the effect of light quality on plant growth and development. Nevertheless, many studies have used LEDs as the sole light source despite of the quality of single wavelength or multiple wavelengths mixed in different ratios [13] [18] [27] . Brazaityte et al. [28] suggested that artificial lighting with too narrow a spectrum was not spectrally optimal for normal plant growth. Furthermore, in a natural environment, it is in conceivable that plants may be supplied with light having a narrow spectrum. Massa et al. [29] commented that broadening the spectrum could achieve better productivity and suggested that LEDs could be used to supplement sunlight or other types of lighting. Hence, in this study, we used white fluorescence tubes as a control treatment and 10% of the control photosynthetic photon flux (PPF) was replaced with red or blue LEDs as light quality treatments at the same irradiance conditions (approximately 70 μmole∙m−2∙s−1 PPF).
2. Materials and Methods
2.1. Plant Material and Culture Conditions
Phalaenopsis aphrodite subsp. formosana plants with 5 leaves were purchased from the Wusulin Farm of the Taiwan Sugar Corp., Tainan County, southern Taiwan (23˚34'N, 120˚38'E), and grown in transparent plastic pots (7.5 cm diameter) filled with sphagnum moss in a computer controlled heating-ventilation system by forcing air through a diesel oil-burning heater to maintain the greenhouse at 28.0˚C ± 1.2˚C. Additionally, the plants were placed under a natural photoperiod without supplying any artificial light for 1 month. The average irradiance on the planting bench was maximum 150 μmole∙m−2∙s−1 PPF at noon on a fine day and measured by LI-250radiometer (Li-Cor, Lincoln, NE, USA).
Six plants were transferred to a growth chamber (ST3-1, SAINT TIEN, Kaohsiung, Taiwan) for each light condition for 6 weeks. Pots were randomly rearranged every week to minimize any effect of edge or position with each treatment. The day and night temperature was set at 28.0 ± 0.5 and 20.0˚C ± 0.5˚C, respectively. The relative humidity was maintained at 70% ± 5%. Plants were irrigated once a week, alternating between HYPONEX No.1 (N:P:K, 7:6:19) and HYPONEX No.5 (N:P:K) (Hyponex, Marysville, OH, USA) water- soluble fertilizer (1.0 g/L) at approximately 1.149 mS/cm and 397.5 mS/cm, respectively, as measured by an electric conductivity meter (3250, JENCO, CA, USA).
2.2. Light Treatments
Cool white fluorescence tubes (F20T12, GE, NY, USA) were used as the main light source and were placed vertically. Two LED panels (200 × 200 mm, HiPoint, Kaohsiung, Taiwan) of red (peak at 660 ±10 nm) or blue (peak at 450 ± 10 nm) were placed horizontally approximately 25 cm above the plant canopy inside the growth chamber to achieve approximately 10% light substitution. The 3 experimental plots were named control (WC), blue enhanced (WB), and red enhanced (WR). All treatments were maintained with a 14 h photoperiod (06:00- 20:00 light/20:00-06:00 dark) and approximately 70 μmole∙m−2∙s−1 PPF above the plant canopy (Table 1). Light spectral distribution was scanned from 380 to 780 nm at 1-nm intervals using a spectroradiometer (LCS-100, Isuzu Optics, Hsinchu, Taiwan) and the spectrum was recorded at the top of the plant canopy (Figure 1). The contribution of blue (400 - 500 nm), green (500 - 600 nm), red (600 - 700 nm), far-red (700 - 780 nm) and total PPF were determined by bandwidth integration. From the spectroradiometer data for each light treatment, the red to far-red ratio (R/FR) was calculated as (integrated photon flux density at 655 - 665 nm) (integrated photon flux density at 725 - 735 nm−1) [30] , and the value of phytochrome photostationary state (PSS) was calculated according to the following equation described by Sager et al. [31] :
![]()
Figure 1. Spectral distribution of (a) the cool white fluorescence tubes control (WC), (b) white light with supplemental blue light (WB), and (c) white light with supplemental red light (WR).
![]()
Table 1. Spectral quality of 3 light sources, cool white fluorescence light (WC), white light with supplemental blue light (WB) and white light with supplemental red light (WR). The value in parentheses represents the percentage over photosynthetic photon flux (PPF). Spectra were recorded and averaged at four locations at the top of plant canopy with a spectroradiometer. Different letters show significant difference among three treatments (P < 0.05).x Total photon flux of the background white fluorescent lamp and supplemental light-emitting diodes. yCalculated according to Kurata et al. (2007). zCalculated according to Sanger et al. (1988).
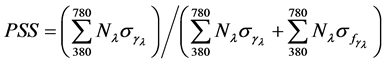
Here, N and s denote irradiance and the photochemical cross-section area, respectively, at each wavelength (l) from 380 - 780 nm against the relative absorption for Pr and Pfr forms of phytochrome. The range of wavelength was not the same as that used by Sager et al. [31] from 300 - 800 nm due to the limitation of spectroradiometer used.
2.3. Determination of Spiking Characteristics
Spiking was defined as the appearance of a flower stalk equal or larger than 0.5 cm long [9] . The spiking ratio represented the ratio for all plants from 5 batches of experiments, with 6 plants for each batch and each treatment. The spike length was measured from the base of the flower stalk at the junction between the base of the leaves and short internode stem to the apex of the flower stalk [9] .
2.4. Sampling Procedure
The leaves were numbered basipetally, and the second leaf (approximately 25 cm2) was approximately two-fold the size of the first leaf. Leaf discs (approximately 0.28 cm2) from the second leaf were cut from the middle and approximately 0.5 cm away from the main vein of the second leaf with use of a cork borer [8] . After all leaves were completely removed, a knife was used to harvest the stem from the junction between the base of the flower stalk bud and the shortened stem, and removed the flower stalk. Three plant replicates were separately obtained at 06:00 (dawn) and 18:00 (dusk) for each treatment. The leaf discs and stems were rapidly weighed and frozen in liquid N2 for analysis.
2.5. Determination of Photosynthetic Pigment Concentration
The contents of Chl a, Chl b, and Car were determined according to the method described by Lichtenthaler [32] . In this method, fresh leaf discs (ca. 200 mg) were extracted in 80% (v/v) acetone. Extracts were centrifuged at 10,000× g for 5 min. The absorbance of supernatant was measured at 663.2, 646.8, and 470 nm with a spectrophotometer (DU640, Beckman Coulter, USA).
Chl a, Chl b, and Car contents (μg/g FW) were calculated using the following formulas:
Chl a = (12.25 A663.2 − 2.79 A646.8) × F;
Chl b = (21.21A646.8 − 5.1 A663.2) × F; and
Car = [(1000 A470 − 1.8 Chl a − 85.02 Chl b)/198] × F.
where F= volume of supernatant(mL)/sample FW (g).
2.6. Maintaining the Integrity of the Specifications
Leaf discs and stems were separately homogenized and sugars were extracted with 80% (v/v) ethanol at 80˚C to determine the soluble sugar concentrations. The supernatant was evaporated in an N2 gas stream and dissolved in deionized water. Sugars were separated by anion exchange chromatography (BioLC, Dionex) with CarboPac PA10column [8] . Sediment was digested with a mixture of pullulanase and amyloglucosidase, and the amount of liberated glucose was estimated by a glucose oxidase and peroxidase method to determine starch content [8] .
2.7. Statistical Analysis
Data are indicated as mean ± SE. The data were subjected to one-way ANOVA and the differences between means were measured at the 5% probability level using Fisher’s protected least significant difference (LSD) test (CoHort Software, Monterey, CA).
3. Results
3.1. Light Source Characteristics
The control (WC) light sources had the spectrum distribution (Figure 1(a)) defined as the percentage over PPF of 28.5% at 400 - 500 nm, 50.6% at 500 - 600 nm, and 20.9% at 600 - 700 nm (Table 1). The blue enhanced (WB) had an additional supply of 11.5% of the total PPF in the blue spectrum (400 - 500 nm) compared to the control. Thus, it changed the spectrum distribution (Figure 1(b)) to 40.0% of the PPF at 400 - 500 nm, 42.6% at 500 - 600 nm, and 17.4% at 600 - 700 nm (Table 1). The red enhanced (WR) had an additional supply of 9.2% of the total PPF in the red spectrum (600 - 700 nm) compared to the control and resulting in a spectrum distribution (Figure 1(c)) of 25.3% of the PPF at 400 - 500 nm, 44.6% at 500 - 600 nm, and 30.1% at 600 - 700 nm (Table 1). Although the mean total PPF of WR was 67.3 μmole∙m−2∙s−1 slightly lower than that of 69.5 and 71.9 μmole∙m−2∙s−1 in WB and WC, respectively, the value of PSS in WR was 0.842, which was significantly higher than for 0.785 and 0.811 in WB and WC, respectively. Meanwhile, the ratio of red to far-red in WR was 12.19, significantly higher than for 3.63 and 4.03 in WB and WC, respectively (Table 1). These results clearly show that a slight increase in red supplement greatly elevated the level of PSS and the R/FR ratio in WR compared to WB and WC.
3.2. Spike Development
The mean spiking ratio was 78% in WR under almost equivalent PPF levels, which was much higher than 14% and 2% of WB and WC, respectively (Table 2). The mean spike length was 3.6 cm in WR, which was longer than the just-emerged spiking of both WB and WC (Table 2). Evidently, a 10% of red light supplementation indeed enhanced spike development, and WB and WC could not show such enhancement.
3.3. Photosynthetic Pigments
The concentrations of Chl a, Chl b, Chl a + Chl b, and the ratio of Chl a/Chl b did not show differences among 3 treatments at either dawn or dusk. At dawn, the amounts of Chl a, Chl b, and Chl a + Chl b were always lower, but the mean ratio of Chl a to Chl b were higher than at dusk in all treatments (Table 3).
![]()
Table 2. Influence of light quality on the spike development of P. aphrodite exposed to WC, WB and WR for 6 weeks. The mean spiking ratio and length are obtained from 5 batch experiments, with 6 individual plants each. Different letters show significant difference among 3 treatments (P < 0.05).
The concentrations of carotenoids at dawn were all lower than at dusk and did not exhibit distinct differences among 3 treatments at dusk. However, at dawn, the lowest level was shown in the WR, resulted in the mean magnitude of dusk-to-dawn oscillation was 46 μg/g FW, which was slightly higher than 34 and 40 μg/g FW in WC and WB, respectively (Table 3).
3.4. Carbohydrates
Table 4 compares the influence of light quality on the dawn and dusk concentration of carbohydrate of the second leaf and stem. In the leaf, no differences in glucose and fructose contents and dawn-to-dusk fluctuations among the three treatments. Sucrose was the major component of free soluble sugars, accounting for 95%, 94%, and 94% at dawn, and 76%, 80%, and 85% at dusk of total sugars (the sum of glucose, fructose, and sucrose) of WC, WB, and WR treatments, respectively. However, a slight dawn-to-dusk fluctuation of sucrose concentration in WC and WR appeared higher at dawn than at dusk, but this fluctuation did not occur in WB. By contrast, the starch showed a significant dawn-to-dusk deposition in all three treatments and its amplitude was 7.0, 6.6, and 8.1 mg/g FW in WC, WB, and WR, respectively.
![]()
Table 3. Influence of light quality on the dawn and dusk concentration of photosynthetic pigments of the second leaf from P. aphrodite exposed to WC, WB, and WR for 6 weeks. All values are mean ± SE of the 3 individual plants. Different letters show statistically significant differences among the 3 treatments at the same sampling time (P < 0.05). The asterisks indicate significant differences between dawn and dusk in samples under the same treatment (P < 0.05).
![]()
Table 4. The influence of light quality on the dawn and dusk concentration of carbohydrates of the second leaf and stem from P. aphrodite exposed to WC, WB, and WR for 6 weeks. All values are mean ± SE of the 3 individual plants. Different letters show statistically significant differences among the 3 treatments at the same sampling time (P < 0.05). The asterisks indicate significant differences between dawn and dusk in samples under the same treatment (P < 0.05).
Compared with the leaf, the stem carbohydrate contents including glucose, fructose, sucrose, and starch were rather stable, except that fructose showed a slight difference at dusk. Sucrose was also a major component of free soluble sugars and accounted for 93%, 98%, and 94% at dawn and 94%, 88%, and 95% at dusk of total sugars in WC, WB, and WR, respectively. Moreover, the sucrose concentrations in the stem was about 5-, 7-, and 4-fold at dawn and 15-, 7-, and 13-fold at dusk higher than in the leaf in WC, WB, and WR, respectively.
4. Discussion
It is commonly recognized that the quantity of light energy correlates directly with photosynthetic performance. Although the magnitude of dawn-to-dusk starch biosynthesis in the leaf was slightly increased in WR, the contents of glucose, fructose, and sucrose in the leaf and stem, and starch in the stem did not show significant differences among the three treatments (Table 4). Hence, under the almost equivalent light quantity condition, the carbon pool metabolism of Phalaenopsis is not much affected by a 10% change in light quality with red or blue light supplementation.
Sucrose concentration was found to be higher in the leaf of Phalaenopsis under a spike-emergence state than a spike-inhibited state [3] - [7] . Tsai et al. [6] also demonstrated that sucrose concentration in the leaf was negatively related to the length of time to reach spiking. However, these studies all examined the new and fully expanded leaf. Leaves are the primary site of perception for light cues that are transmitted to the shoot apical meristem via the phloem to evoke the transition from vegetative growth to reproductive development [33] . In this study, the sucrose concentration in the stem was significantly higher than in the leaf; meanwhile, there was no difference in the 3 light treatments either in the second leaf or in the stem (Table 4). These results are concurrent with Liu et al. [9] , which manifested the sucrose concentration in the stem was profoundly higher than in the different ages of leaves of Phalaenopsis, irrespective of spiking or not. Therefore, the relationship between spiking and sucrose concentration is seemingly not so tight in Phalaenopsis.
The effect of light quality upon photosynthetic pigment content, and the correlation between pigment change and developmental transition reported so far appeared to be rather complex. In this study, Chl a, Chl b, and Car all were degraded during nighttime and a 10% red or blue LED supplementation did not greatly affect the contents of these pigments, even though the magnitudes of dusk-to-dawn fluctuation in Chl a/b ratio and Car content had slight changes among the three treatments (Table 3). Evidently, the spiking of Phalaenopsis induced by a 10% red LED supplementation is not mediated by the alternation of photosynthetic pigments.
The flowering of many Phalaenopsis cultivars was hastened when grown under the red shade net as compared to blue and black shade ones [34] . Recently, Dueck et al. [35] published that a high red:far red ratio via supplemental high pressure sodium resulting in a high PSS value can enhance flower bud break of two Phalaenopsis cultivars, Quincy and Red Stones, compared to the supplement of a combination of high pressure sodium and far-red LEDs with a low PSS value. In this study, likewise, a 10% increase in red supplement significantly elevated the ratio of red to far-red light and the level of PSS (Table 1). Accordingly, the spiking development increased (Table 2). These results strongly highlight that the spike emergence of P. aphrodite closely depends on the relative amount of active phytochrome expressed as the photostationary state.
The light spectrum affects the hormone balance in plants [36] . Red light induces a great increase in the expression of a key gene for gibberellin (GA) biosynthesis in lettuce [37] . Although GAs affect flowering in a species-dependent manner, Ben-Tal and Erner [38] demonstrated that GAs were routinely used to promote flowering in many ornamental plants, such as Chrysanthemum, Hypericum, Gypsophilla, and Campanula. Similarly, GA content increased in the flowering shoots of Phalaenopsis under low temperature conditions when flowering development was promoted [39] . Applications of GA and cytokinin, such as benzyladenine (BA) simultaneously or BA alone promoted flowering of Phalaenopsis; however, BA treatment has the risk to develop abnormal inflorescence [40] . Phytochrome negatively controlled GA sensitivity and/or biosynthesis [41] , and the level of R/FR ratio affected biosynthesis or distribution of other hormones, such as auxin, cytokinin, ethylene, and brassinosteroid [42] . Hence, mechanisms for the phytochrome-mediated spike emergence of Phalaenopsis via changing the type and/or concentrations of various phytohormones or other downstream mediators acting as a leaf-to-stem transported signal remain to be explored.
Temperature is a primary factor that affects the transition from a vegetative into a reproductive state in Phalaenopsis [43] . It should be mentioned that the temperature regime in this study was set at the spike-inducing condition as follows: warm day (28˚C) and cool night (20˚C), as in our previous study [8] . In our earlier study, plants with 7 leaves were grown in 10.5 cm diameter pots, the size of the second leaf was ca. 85 cm2, and the spike emergence was significant in 4 weeks [8] . In this study, however, under the same temperature conditions, no significant spiking occurred in the control (WC) plants (Table 2), which had 5 leaves and were grown in 7.5 cm diameter pots and the experimental leaf was only ca. 25 cm2 in size. These results are consistent with Paradiso and De Pascale [44] addressed that the time for spike emergence was strongly influenced by the plant size, and Blanchard et al. [45] reported that a longer cooling duration may be required for smaller Phalaenopsis plants.
For Phalaenopsis, it is a rather complex and time-consuming process from spike emergence to flowering. It takes about 4 - 5 months for P. aphrodite from spike emergence to the first floret appearance. Meanwhile, the culturing conditions in commercial production are different between spike induction and inflorescent development [44] [46] . In this study, we focused on the effects of light quality on spike emergence because breaking the dormancy of flower stalk/spike bud(s) was a key step to produce flowers in Phalaenopsis. Orchid growers induce spike emergence when the orchid is grown in a pot of 10.5 cm diameter. We examined the plants cultured in 7.5 cm diameter pot in this study because they were limited by the space of the growth chamber (620 × 520 × 1300 mm) and the size of LED panel (200 × 200 mm). Nonetheless, there are some commercial markets for those grown in smaller pots. For Phalaenopsis breeders, it takes a long time to know the flower characteristics after hybridization. Therefore, accelerating the spike emergence can shorten the breeding time.
Overall, this study offers some potentially practical value for promoting spike development in Phalaenopsis by supplementing red light and opens up a new academic approach via the induction of PSS to control spiking in Phalaenopsis.
Acknowledgements
The authors would like to thank Professor Jong-Ching Su, Professor Fure-Chyi Chen and Doctor Yung-I Lee for helpful comments on drafting this manuscript. The authors sincerely thank HiPoint Co. (Taiwan) for lending the spectroradiometer (LCS-100; Isuzu Optics Co., Hsinchu, Taiwan) to scan light spectral distribution. This work was supported by the grant NSC 98-2313-B-390-001-MY3 from the National Science Council, Executive Yuan, Taiwan.
NOTES
![]()
*Corresponding author.