1. Introduction
Recently, the binary compounds such as TlAs, AlAs, ScN, GaN and their mixtures such as TlAlAs, ScGaN have been studied theoretically [1] -[3] , because the wide range of the band gap is important for microelectronics devices. Generally, monoxide compounds are known as rock salt structures (B1) at room temperature and under pressure. Y. Duan et al. [4] have been handled the electronic properties of XO (X = Be, Mg, Ca, Sr, Ba, Zn and Cd) for wrutzite, zincblende and rock salt structure. Ponce et al. [5] have analyzed theoreticaly and experimentaly electronic and optic properties of CaS nad CaO. Albuquerque and Vasconcelos [6] are reported structual, electronic and optical properties of CaO. Karki et al. [7] [8] have inversigated structual, dynamic and electronic properties of liquid MgO via density functional theory. Makaremi and Nourbakhsh have declerated structual, electronic and magnetic properties of Mgo nanolayers. Nishi et al. [9] have investigated experimentally metastable 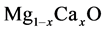 solid solutions film ZnO layers. Stolbov and Cohen [10] have studied the electronic structure for equilibrium MgO-CaO. Miloua et al. [11] have calculated the electronic properties of
solid solutions film ZnO layers. Stolbov and Cohen [10] have studied the electronic structure for equilibrium MgO-CaO. Miloua et al. [11] have calculated the electronic properties of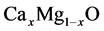 , theoretically. Besides, A. Srivastava et al. [12] calculated phase translations in
, theoretically. Besides, A. Srivastava et al. [12] calculated phase translations in 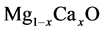 alloys.
alloys.
In this paper, we represent the bowing parameter of 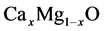 alloys by first principles density functional theory. To the best of our knowledge, no theoretical as well as experimental work has been performed thus far for the bowing parameter of
alloys by first principles density functional theory. To the best of our knowledge, no theoretical as well as experimental work has been performed thus far for the bowing parameter of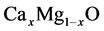 . The aim of this paper is to understand the attitude of the bowing parameters and contribution to the gap bowing parameters. The paper is methodized as follows: computational methodology is given in Section 2. Results and discussion are represented in Section 3. The study is concluded in Section 4.
. The aim of this paper is to understand the attitude of the bowing parameters and contribution to the gap bowing parameters. The paper is methodized as follows: computational methodology is given in Section 2. Results and discussion are represented in Section 3. The study is concluded in Section 4.
2. Computational Details
The calculations for CaO, MgO and 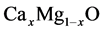 in the rock salt structure were investigated within the generalized gradient approximation (GGA) of density functional theory (DFT) using the PWSCF code [13] . In Quantum Espresso, the examining is performed by utilizing the Kohn-Sham [14] formation established on the DFT. Total energies have been calculated by using ultrasoft pseudopotentials and plane-wave basis sets. The exchange- correlation potentials in the GGA [15] is separately used in the calculations. The electronic configurations used for the pseudo potentials were Ca(3p64s), Mg(2p63s) and O(2s22p4). The Khon-Sham [14] orbitals were described using a plane wave basis set. The highest kinetic energy of a plane wave in the chosen fundamental set is known the cutoff energy. Specialize assignation of the cutoff energy is important for achieving accurate results with available computational process. The values of cutoff energies used in our calculations are summarized in Table 1.
in the rock salt structure were investigated within the generalized gradient approximation (GGA) of density functional theory (DFT) using the PWSCF code [13] . In Quantum Espresso, the examining is performed by utilizing the Kohn-Sham [14] formation established on the DFT. Total energies have been calculated by using ultrasoft pseudopotentials and plane-wave basis sets. The exchange- correlation potentials in the GGA [15] is separately used in the calculations. The electronic configurations used for the pseudo potentials were Ca(3p64s), Mg(2p63s) and O(2s22p4). The Khon-Sham [14] orbitals were described using a plane wave basis set. The highest kinetic energy of a plane wave in the chosen fundamental set is known the cutoff energy. Specialize assignation of the cutoff energy is important for achieving accurate results with available computational process. The values of cutoff energies used in our calculations are summarized in Table 1.
The plane wave energy cut off is selected 90 Ry. Accurate Brillounin zone investigations are carried out using the standard special k-points technique of Monkhorst and Pack [16] . The Brillouin zone investigation was performed over a 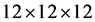 mesh points. Our calculations involve an 16 atom for
mesh points. Our calculations involve an 16 atom for 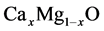 alloys in a supercell. We start at MgO cluster and finish at CaO cluster.
alloys in a supercell. We start at MgO cluster and finish at CaO cluster.
3. Results and Discussion
3.1. Structural Properties of Binary Compounds
The ternary compounds 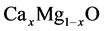 are bordered by two binary compounds of CaO and MgO. In order to be able to analyze the energy band gaps and bowing parameters of
are bordered by two binary compounds of CaO and MgO. In order to be able to analyze the energy band gaps and bowing parameters of 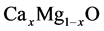 ternary alloys, it is wholesome to study the CaO and MgO binary compounds in terms of their structural and electronic properties. By lessening the total energy with regards to the atomic positions and lattice parameters we carried out the structural optimization. Equilibrium lattice parameters are obtained by fitting the total energy with the different volumes according to the Birch equation of states.
ternary alloys, it is wholesome to study the CaO and MgO binary compounds in terms of their structural and electronic properties. By lessening the total energy with regards to the atomic positions and lattice parameters we carried out the structural optimization. Equilibrium lattice parameters are obtained by fitting the total energy with the different volumes according to the Birch equation of states.
The Birch equation of states [17] can be seen in the Equation (1):
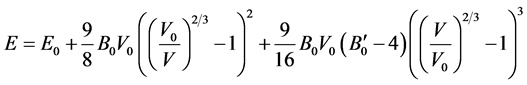 (1)
(1)
where 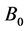 and
and ![]() are the bulk modulus and its pressure derivative at the equilibrium volume
are the bulk modulus and its pressure derivative at the equilibrium volume![]() . The calcu-
. The calcu-
![]()
Table 1. Ground state energies for equilibrium MgO and CaO with various cutoff energies.
lated values for the equilibrium of CaO and MgO are 4.805 Å and 4.263 Å, respectively. We represented and compered the equilibrium lattice parameter, bulk modulus ![]() and bulk modulus derivation
and bulk modulus derivation ![]() in Table 2.
in Table 2.
3.2. Structural Properties of ![]()
In this paper, we examined the effectiveness of the Vegard’s law for rocksalt ![]() alloys in its ground states. Ca composition
alloys in its ground states. Ca composition ![]() dependent lattice constant of
dependent lattice constant of ![]() compounds is expressed as:
compounds is expressed as:
![]() (2)
(2)
where![]() ,
, ![]() and
and ![]() are the lattice constants of the
are the lattice constants of the![]() , CaO and MgO, respectively. The volve of the deflection from Vegard’s law can be calculated by Equation (2). The numerical calculations are fulfilled for the different situations. Seven compositions of
, CaO and MgO, respectively. The volve of the deflection from Vegard’s law can be calculated by Equation (2). The numerical calculations are fulfilled for the different situations. Seven compositions of ![]() alloys were checked: 0.125, 0.25, 0.375, 0.5, 0.625, 0.75 and 0.875. The calculated results of lattice parameters with respect to
alloys were checked: 0.125, 0.25, 0.375, 0.5, 0.625, 0.75 and 0.875. The calculated results of lattice parameters with respect to ![]() compositions are itemized in Table 3 from which we find that the lattice parameter increases with increasing Calcium composition because these results are due diognosis atomic radius. The atomic radius of Calcium and Magnesium are 194 and 145 picometers, respectively. We represented and compered the equilibrium lattice parameter of
compositions are itemized in Table 3 from which we find that the lattice parameter increases with increasing Calcium composition because these results are due diognosis atomic radius. The atomic radius of Calcium and Magnesium are 194 and 145 picometers, respectively. We represented and compered the equilibrium lattice parameter of ![]() ternary alloys in Table 3.
ternary alloys in Table 3.
The results also propose that the composition-dependent lattice parameter of the ![]() ternary alloys can be represented by a third-order polynomial equation,
ternary alloys can be represented by a third-order polynomial equation,![]()
![]() .
.
The bowing parameter, ![]() , is significant for investigating the band gap energy ternary alloys. The band gap energy of ternary alloys describe by the band gap energy of binary compounds, a quadratic interpolation of
, is significant for investigating the band gap energy ternary alloys. The band gap energy of ternary alloys describe by the band gap energy of binary compounds, a quadratic interpolation of
![]()
Table 2. Itemized lattice parameter a, bulk modulus ![]() and bulk modulus derivation
and bulk modulus derivation ![]() for the binary compounds MgO and CaO in rocksalt structure at equilibrium volume.
for the binary compounds MgO and CaO in rocksalt structure at equilibrium volume.
![]()
Table 3. Itemized lattice parameter a for the ternary alloys ![]() in rocksalt structure at equilibrium volume. All values are Å.
in rocksalt structure at equilibrium volume. All values are Å.
composition amount ![]() and the bowing parameter.
and the bowing parameter.
The band gap energy of ternary alloys ![]() given by
given by
![]() . (3)
. (3)
Here, ![]() is the band gap energy of the ternary
is the band gap energy of the ternary ![]() compound,
compound, ![]() is the band gap energy of the CaO compound,
is the band gap energy of the CaO compound, ![]() is the band gap energy of the MgO compound and
is the band gap energy of the MgO compound and ![]() is the band gap bowing parameter of
is the band gap bowing parameter of![]() . Bowing parameter is associated with the band gap energy. Figure 1 shows bowing parameter as a function of
. Bowing parameter is associated with the band gap energy. Figure 1 shows bowing parameter as a function of ![]() for ternary alloys. We calculated nearly linear variation for
for ternary alloys. We calculated nearly linear variation for ![]() different composition,
different composition, ![]() , determinating powerful. Our calculations show that according to the band gap energy of ternary alloys, bowing parameter decreases. The results are given by Table 4 and our results show that an average bowing parameter of
, determinating powerful. Our calculations show that according to the band gap energy of ternary alloys, bowing parameter decreases. The results are given by Table 4 and our results show that an average bowing parameter of ![]() is ~0.583 eV.
is ~0.583 eV.
The combination-dependent bowing parameter function [21] ![]() was described as
was described as
![]() (4)
(4)
The band gap of the ternary alloys are correlated with the band gaps of the binary compounds. The band gap bowing parameters ![]() have three physically contributions [17] .
have three physically contributions [17] .
Finally, the total band gap bowing parameter can be written by resolving into its components as:
![]() . (5)
. (5)
The effect of volume deformation causes the first term of bowing parameter, ![]() , in Equation (5). The no-
, in Equation (5). The no-
![]()
Figure 1. Composition dependence of the bowing parameter for ![]() ternary alloys.
ternary alloys.
![]()
Table 4. Itemized bowing parameter and contribution of the bowing parameter a for the ternary alloys ![]() in rocksalt structure at equilibrium volume.
in rocksalt structure at equilibrium volume.
tional response of MgO(CaO) to hydrostatical pressure states this term via the effect of the contribution of balanced lattice constant ![]() to the lattice constant of the alloy value
to the lattice constant of the alloy value![]() .
.
![]() (6)
(6)
A charge transfer in MgO and CaO at ![]() indicates the second term,
indicates the second term, ![]() , of the total band gap bowing parameter,
, of the total band gap bowing parameter,
![]() (7)
(7)
The third term, ![]() refers to the structural relaxation which takes place during the passing from the unrelaxed to the relaxed alloy
refers to the structural relaxation which takes place during the passing from the unrelaxed to the relaxed alloy
![]() (8)
(8)
In Table 4, the values of bowing parameter, ![]() and its volume deformation component
and its volume deformation component![]() , charge exchange component
, charge exchange component ![]() and structural relaxation component
and structural relaxation component ![]() are expressed for
are expressed for ![]() ternary alloys.
ternary alloys.
The results also propose that the combination-dependent bowing parameter of the ![]() alloys can be represented by a second-order polynomial equation,
alloys can be represented by a second-order polynomial equation, ![]() eV. The results clearly show that
eV. The results clearly show that ![]() and
and ![]() are weak and composition dependent. The prime contribution to the gap bowing is from the
are weak and composition dependent. The prime contribution to the gap bowing is from the![]() . The interval of the calculated band gap bowing coefficient for
. The interval of the calculated band gap bowing coefficient for ![]() alloys is from 2.900
alloys is from 2.900 ![]() to 4.234
to 4.234![]() . One can note that for all contributions of
. One can note that for all contributions of ![]() the main addition to the gap bowing is owing to the
the main addition to the gap bowing is owing to the ![]() effect. This could be correlated to the strong ionicity dissociable of the corresponding binary compounds (CaO and MgO). The contribution of the volume deformation term to the bowing parameter
effect. This could be correlated to the strong ionicity dissociable of the corresponding binary compounds (CaO and MgO). The contribution of the volume deformation term to the bowing parameter ![]() is gradually decreasing with
is gradually decreasing with ![]() concentration. Suddenly, in the case of
concentration. Suddenly, in the case of ![]() the addition of the
the addition of the ![]() has increasing. The addition of the other structural relaxation
has increasing. The addition of the other structural relaxation ![]() is weak. Furthermore,
is weak. Furthermore, ![]() and
and ![]() is nearly no effect to the bowing parameter. Namely, the
is nearly no effect to the bowing parameter. Namely, the ![]() is the only are component which enables the bowing parameter for
is the only are component which enables the bowing parameter for ![]() alloys.
alloys.
4. Conclusion
We have examined the electronic properties of the rocksalt ![]() ternary alloys as a function of Calcium composition
ternary alloys as a function of Calcium composition ![]() by using the GGA method within DFT. The electronic band structures, which are calculated by using the lattice parameters composed from Vegard’s law. In the GGA, a band gap bowing parameters are achieved for rock salt
by using the GGA method within DFT. The electronic band structures, which are calculated by using the lattice parameters composed from Vegard’s law. In the GGA, a band gap bowing parameters are achieved for rock salt ![]() ternary alloys. These results propose the physical condition of a composition dependent band gap energy for
ternary alloys. These results propose the physical condition of a composition dependent band gap energy for ![]() ternary alloys. Our calculations show that the
ternary alloys. Our calculations show that the ![]() ternary alloy’s the bowing parameters are very strong Calcium composition. The average bowing parameter is 0.583 eV for
ternary alloy’s the bowing parameters are very strong Calcium composition. The average bowing parameter is 0.583 eV for ![]() ternary alloys. The results clearly show that the
ternary alloys. The results clearly show that the ![]() is the only dominant component of the bowing parameter for
is the only dominant component of the bowing parameter for ![]() alloys. Additionally
alloys. Additionally ![]() and
and ![]() are weak.
are weak.