Development of controlled release formulations of thiabendazole ()
Figure 2. Adsorption kinetics of Thiabendazole on M48 at 40˚C. Bars represent standard deviation.
![]()
Figure 3. Adsorption of thiabendazole on M48 at different pH values. Bars represent standard deviation.
instance, it can be suggested that at pH 3 or pH 5.5 large fractions of Thiabendazole molecules became positively charged molecules consequently large adsorbed amount were obtained. At high pH value (pH 9) large fraction of Thiabendazole molecules became negatively charged or non charged molecules. In this case raw bentonite par- ticles can not adsorb a negatively charged Thiabenda- zole molecule due to repulsion between the negatively charged bentonite surfaces and Thiabendazole. Further- more, the hydrophilic bentonite surfaces can not adsorb large fraction from Thiabendazole molecules due to the hydrophobocity of Thiabendazole at high pH value. The effects of pH values on the ionization status of Thiaben- dazole molecules are shown in Figure 4. Our suggestion is supported by the previous results of El-Nahhal and Safi [29], who provided acceptable ionization status of bromoxynile adsorption on organo-clays and by Rodri- guez et al [30] who achieved Thiabendazole separation using a buffer of formic acid-ammonium formate at pH 3.5 with 2% of methanol using solid phase extraction.
In addition, further support to our discussion can also be obtained from previous reports [24,31] who provided different KowlogP values for Thiabendazole at different pH values.
3.3. Adsorption Isotherms at Different pH
Figure 5 illustrates the adsorption isotherms of Thia- bendazole on bentonite clay different pH values. As ex- pected, Thiabendazole was poorly adsorbed on raw ben- tonite (M48) high pH (pH 9), whereas low pH (pH 5.5 and pH 3) significantly enhanced the adsorbed amounts of Thiabendazole. It can be seen that at low equilibrium concentration (less than 0.5 mg/l) a sharp increase in the adsorbed amounts of Thiabendazole was observed. In- creasing Thiabendazole concentration in the equilibrium solution did not produce further increase in the adsorbed amount of Thiabendazole, and the adsorption reached to plateau. This feature of adsorption was obtained in the organic cation and clay minerals. Moreover, reaching a plateau suggests that the available adsorption site in bentonite surfaces were nearly occupied by Thiabenda- zole molecules. Similar feature was obtained with ad- sorption of cationic herbicides, paraquat and diquat, ad- sorption on clay [32] and organic cations to clays [25]. The explanation of pH effect on the adsorption of Thia- bendazole is given above.
Figure 3 shows the adsorption isotherms of Thiaben- dazole on bentonite at various pH values.
Thiabendazole has an acid dissociation constant (pKa) of 4.73 and is mostly neutral at pH < 4.73, becoming deprotonated (anionic) at pH > 7.
Accordingly, the adsorbed amount of Thiabendazole obtained at pH 3 is higher than the adsorbed amount
![]()
Figure 4. Influence of pH value on the ionization status of Thiabendazole.
![]()
Figure 5. Adsorption of Thiabendazole on clay (M48) at various values of pH at 291 K.
obtained at pH 5.5 and pH 9 (Figure 3). Statistical ana- lysis showed a significant difference, p-value with t-test = 0.004.
The high adsorbed amount at pH 3 was because, at pH 3, Thiabendazole molecules are mostly protonated, and have 2 positive charges in the N-atoms, in this case they may be adsorbed as cations on hydrophilic sites of the bentonite surfaces. At pH 5.5, a part of Thiabendazole molecules has a one positive charge in the N-atoms and the other part is in the neutral form (Figure 4). In this case, the one positive charged molecules are adsorbed on the hydrophilic sites of bentonite surfaces, and the neu- tral forms stay in solution; accordingly, low adsorbed amounts of Thiabendazole molecules were observed. At pH 9 the majority of Thiabendazole molecules are nega- tively charged or in equilibrium with the neutral form. In this case the negatively charged molecules stay in the solution due to the electronic repulsion between the anio- nic form of Thiabendazole and the negatively charged bentonite surfaces. Moreover, very little of the neutral Thiabendazole molecules are adsorbed the negatively charged bentonite surfaces through water bridge. The low adsorption in this case is due to the hydrophilic sur- face of bentonite particles and the hydrophobic proper- ties of Thiabendazole molecules. This explanation is sup- ported by previous reports of [29] that found a high ad- sorbed amount of bromoxynil in soil and wheat char at low pH. Furthermore, the shape of the isotherm tends to be L-type and adsorption occurs as the Longmuir type at various pH values, due to changes in the electronic form of Thiabendazole a result of pH changes.
Furthermore, the shape of the isotherms was similar in all cases. According to Gile et al. [33] classification, iso- therms of Thiabendazole could be classified as L type based on the initial slope of the curve in the concentra- tion range studied. Adsorption isotherms were regular, positive and concave to the concentration axis (Figures 5 and 6), which indicated no strong competition for the adsorption sites between the solvent (water) and Thia- bendazole molecules on bentonite surfaces. This suggests that Thiabendazole adsorption becomes easier as its con- centration in the aqueous phase rises. These isotherms indicate the tendency of large adsorbed molecules to as- sociate rather than to remain as isolated units. Fitting the data in Figures 5 and 6 to the Freundlich equation gives the adsorption parameter, (Q = KC1/n) where Q is the adsorbed amount in mg/g, C is the equilibrium concen- tration of Thiabendazole, K reflects the binding coeffi- cient, and n is the physical adsorption parameter.
It can be seen in Table 1 that the K value of adsorption on raw bentonite at pH 3 is several times higher than at pH 5.5 and pH 9, indication the high affinity of Thia- bendazole to bentonite surfaces at low pH. Furthermore, the binding coefficient (K) is the highest at 291 K. This
![]()
Figure 6. Influence of temperature on the adsorption of Thia- bendazole on M48. Bars represent standard deviation.
![]()
Table 1. Physical parameters of Thiabendazole adsorption at different conditions.
suggests that the optimum adsorption occurred at 291 K. Furthermore, the regression values (R2) in all cases are close to 1 indication of a strong positive association.
3.4. Adsorption Isotherms at Different Temperature
Adsorption of Thiabendazole at different temperature degrees are shown in Figure 6. It can be seen that ad- sorption of Thiabendazole is more pronounced at 291 than at 278 K and at 313 K. The explanation of these results is that at a low temperature (278 K), the chemical potential of Thiabendazole molecules is reduced and the molecules tend to from crystals due to their low solubili- ty in water, 0.16 g/l, 20˚C [26]. At 291 K, nearly the op- timum temperature of solubility in water, Thiabenda- zole molecules are very active and interact with the ad- sorption site at the bentonite surfaces accordingly high adsorbed amount of Thiabendazole was observed. In contrast, at high temperature (323 K), the system absorbs heat energy and an increase in the chemical potential of Thiabendazole molecules may occur. This step makes more Thiabendazole molecules in a dynamic mobility in the aqueous solution; accordingly, low adsorption was observed, indicating low adsorption affinity at high tem- perature. Statistical analysis of the data in Figure 6 indi- cates significant difference among the adsorbed amount at 278, 291 and 323 K, as the error bars which represent the standard deviation do not overlap at any point of the equilibrium concentration (Figure 6). Furthermore cal- culation of p-values among the three isotherms showed value ranges between 0.034 - 0.029, indicating signifi- cant differences among all cases.
This supports the suggestion that optimal adsorption occurred at temperature 291 K.
Changes in molar free energy (DGo) of the adsorption reaction of Thiabendazole at different temperatures were calculated using the following equation [21]:
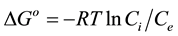
where, DGo is the molar free energy change (Kcal mol−1), R is the gas constant (1.986 cal K−1 mol), T is the abso- lute temperature of the adsorption reaction, and Ci/Ce are the initial and equilibrium concentrations, respectively.
The values of DGo for adsorption at 278 K, 291 K and 313 K from distilled water were −0.802 ± (0.313), −2.147 ± (0.504) and −0.369 ± (0.18), respectively. These results are in accordance with the data in Figure 6. These values are within the expected range for physical adsorption [34]. Analysis of variance between the calcu- lated free energy at 278 K and 291, and between 291 and 313 gave p-values of 0.0005 and 8.86E−06 respectively.
3.5. Release of Thiabendazole
The release of Thiabendazole from bentonite-Thiaben- dazole complexes at different pH values are shown in Figure 7. It is evident that % released fraction of Thia- bendazole at pH 3, pH 5.5 and pH 9 are: 1.48 ± 0.05, 25.71 ± 4.46 and 79.88 ± 9.8 respectively. The explana- tion of these results is that adsorption of Thiabendazole at low pH values (3, 5.5) tends to be attraction between the positive charge of Thiabendazole (Figure 4) and the negative charge of the Ca-bentonite clay. This interac- tions is very strong, accordingly the release of Thiaben- dazole molecules from the clay surfaces depends on the pH of added water and the possibility of Thiabendazole molecules to form hydrogen bonding with water mole- cules.
Under low pH values (pH 3, 5.5), the electrostatic in- teractions between Thabendazole molecules and water is stronger than the hydrogen bonding with water mole- cules accordingly low fraction of Thabendazole is availa- ble for solubility and release at water has low pH value. Under high pH value (pH 9), the electronic status of Tha- bendazole changed from positively charged molecules to a neutral or negatively charged ones. Accordingly the possibility of hydrogen bonding between Thabendazole and water molecules is very high beside the fact that the electrostatic interaction between Thabendazole and Ca- bentonite clay became very week. Accordingly, high
fraction of Thabendazole is available for release as evident in Figure 7.
Kinetics of Thiabendazole release at different water volumes are shown in Figure 8. It can be seen that upon application of 0.05 L water (first washing) about 80% of the initial Thiabendazole concentration was released from raw bentonite-Thiabendazole complex at pH 9, whereas 26% and 1.5% Thiabendazole concentrations were released at pH 5.5 and pH 3. Further application of eluting water up to 500 ml resulted in the further release of Thiabendazole up to 97%, 70% and 2.6% from raw bentonite-Thiabendazole at pH 9, pH 5.5 and pH 3 re- spectively. Thus, the release of Thiabendazole from ben- tonite-Thiabendazole complex at pH 3 was significantly slower than at pH 5.5 and pH 9.
The explanation of these results is that the release of Thiabendazole from bentonite-Thiabendazole complexes is controlled by the binding coefficients, (K) values, which are very high at pH 3 and pH 5.5 and low at pH 9 (Table 1). These results are in agreement with a previous report [28], which found that herbicide release was con- trolled by binding coefficients. These results are in ac- cordance with the adsorption data (Figure 5).
![]()
Figure 7. Influence of solution pH on the release fraction of Thiabendazole from bentonite (M48). Bars represent standard deviation.
![]()
Figure 8. Kinetics of Thiabendazole release from bentonite (M48) at different solutions pH. Bars represent standard deviation.
4. Concluding remarks
This study reveals that changing the solution pH of Thiabendazole resulted in changing the ionization status of the molecules and consequently enabled them to be adsorbed on the hydrophilic bentonite surfaces. The ra- tionale for this work is that adsorption of Thiabendazole on bentonite surfaces can be enhanced by changing solu- tion pH. These findings showed that the highest adsorbed amounts were obtained at pH 3 and at 291 K (Figures 3 and 6). The release of Thiabendazole was very slow from bentonite complexes at lower pH (Figures 7 and 8). The environmental relevance of this work is that bentonite clay can be used to develop environmentally acceptable Thiabendazole formulations for safe controlled release formulation.
ACKNOWLEDGEMENTS
Dr Y. El-Nahhal acknowledges Alexander von Humboldt Stiftung Foundation Fellowship Grant no IV-PAL/1104842 STP, Germany. Special thanks go to Prof Dr G.lagaly at Kiel University.
REFERENCES
[1] El-Nahhal, Y. (2004) Contamination and safety status of plant food in Arab countries. Journal of Applied Science, 4, 411-417. http://dx.doi.org/10.3923/jas.2004.411.417
[2] Dogheim, S.M., Gad Alla, S.A., El-Syes, S.M.A., Almaz, M.M. and Salama, E.Y. (1996) Organochlorine and orga- nophosphorus pesticide residues in food from Egyptian local markets. Journal of the Association of Official Ana- lytical Chemists, 79, 949-952.
[3] Nazer, I.K. and Masoud, H.A. (1986) Residues of diclofol on cucumber grown under plastic covers in Jordan. Jour- nal of Environmental Science and Health Part B, 21, 387- 399. http://dx.doi.org/10.1080/03601238609372532
[4] Sawaya, W.N., Al-Awadhi, F.A., Saeed, T., Al-Omair, A., Ahmad, N., Husain, A., Khalafawi, S., Al-Omirah, H., Dashti, B., Al-Amiri, H. and Al-Saqer J. (1999) Kuwait’s total diet study: Dietary intake of organochlorine, carba- mate, benzimidazole and phenylurea pesticide residues. Journal of the Association of Official Analytical Chem- ists, 82, 1458-1465.
[5] Tahir, S., Anwer, T., Ahmad, I., Aziz, S., Mohammad, A. and Ahad, K. (2001) Determination of pesticide residues in fruits and vegetables in Islamabad market. Journal of Environmental Biology, 22, 71-74.
[6] Safi, J., Abou Foul, N., El-Nahhal, Y. and El-Sebae, A. (2001) Monitoring of pesticide residues on green pepper, potatoes, Vicia faba, green bean and green peas in Gaza Governarate (PNA), Palestine. Journal of Pest Control and Environmental Science, 9, 55-72.
[7] Garcia-G, J.E., Kirchhoff, J. and Grossmann F. (1990) Behavior of captafol residues after prolonged application in a wheat monoculture. Journal of Environmental Sci- ence and Health Part B, 2, 185-204. http://dx.doi.org/10.1080/03601238609372532
[8] Schirra, M., D’Aquino, S., Palma, A., Marceddu, S., An- gioni, A., Cabras, P., Scherm, B. and Migheli, Q. (2005) Residue level, persistence, and storage performance of ci- trus fruit treated with fludioxonil. Journal of Agriculture and Food Chemistry, 53, 6718-6724. http://dx.doi.org/10.1021/jf051004w
[9] Stromqvist, J. and Jarvis, N. (2005) Sorption, degradation and leaching of the fungicide iprodione in a golf green under Scandinavian conditions: measurements, modelling and risk assessment. Pest Management Science, 61, 1168- 1178. http://dx.doi.org/10.1002/ps.1101
[10] Padovani, L., Capri, E., Padovani, C., Puglisi, E. and Trevisan, M. (2006) Monitoring tricyclazole residues in rice paddy watersheds. Chemosphere, 62, 303-314. http://dx.doi.org/10.1016/j.chemosphere.2005.05.025
[11] Palma, G., Sanchez, A., Olave, Y., Encina, F., Palma, R. and Barra, R. (2004) Pesticide levels in surface waters in an agricultural-forestry basin in Southern Chile. Chemos- phere, 57, 763-770. http://dx.doi.org/10.1016/j.chemosphere.2004.08.047
[12] Turiel, E. and Martín, E. (2012) A Molecularly imprinted stir bars for selective extraction of thiabendazole in citrus samples. Journal of Separation Science, 35, 2962-2969. http://dx.doi.org/10.1002/jssc.201200554
[13] García-Reyes, J.F., Gilbert-López, B., Molina-Díaz, A. and Fernández-Alba, A.R. (2008) Determination of pesti- cide residues in fruit-based soft drinks. Analytical Chemi- stry, 80, 8966-8974. http://dx.doi.org/10.1021/ac8012708
[14] He, Q., Kong, X., Zhao, J., Li, J., Yue, A. and Zhang, Y. (2008) Determination of benomyl, carbendazim and thia- bendazole in apple juice concentrate using solid-phase extraction coupled with ion exchange chromatography. Chinese Journal of Chromatography, 26, 563-567.
[15] Prousalis, K.P., Polygenis, D.A., Syrokou, A., Lamari, F.N. and Tsegenidis, T. (2004) Determination of carben- dazim, thiabendazole, and o-phenylphenol residues in le- mons by HPLC following sample clean-up by ion-pair- ing. Analytical and Bioanalytical Chemistry, 379, 458- 463. http://dx.doi.org/10.1007/s00216-004-2614-1
[16] Piccirilli, G.N. and Escandar, G.M. (2009) Flow injection analysis with on-line nylon powder extraction for room- temperature phosphorescence determination of thiaben- dazole. Analytica Chimica Acta, 646, 90-96. http://dx.doi.org/10.1016/j.aca.2009.05.033
[17] Harino, H., Mori, Y., Yamaguchi, Y., Shibata, K. and Senda, T. (2005) Monitoring of antifouling booster bio- cides in water and sediment from the port of Osaka, Japan. Archives of Environmental Contamination and Toxicolgy, 48, 303-310. http://dx.doi.org/10.1007/s00244-004-0084-2
[18] García-Reyes, J.F., Jackson, A.U., Molina-Díaz, A. and Cooks, R.G. (2009) Desorption electrospray ionization mass spectrometry for trace analysis of agrochemicals in food. Analtical Chemistry, 81, 820-829. http://dx.doi.org/10.1021/ac802166v
[19] Grogan, H.M. and Jukes, A.A. (2003) Persistence of the fungicides thiabendazole, carbendazim and prochloraz- Mn in mushroom casing soil. Pest Management Science, 59, 1225-1231. http://dx.doi.org/10.1002/ps.759
[20] Castillo, L.E., Martínez, E., Ruepert, C., Savage, C., Gi- lek, M., Pinnock, M. and Solis, E. (2006) Water quality and macroinvertebrate community response following pesticide applications in a banana plantation, Limon, Costa Rica. The Science of the Total Environment, 367, 418-432. http://dx.doi.org/10.1016/j.scitotenv.2006.02.052
[21] Kim, H.J., Lee, D.S. and Kwon, J.H. (2010) Sorption of benzimidazole anthelmintics to dissolved organic matter surrogates and sewage sludge. Chemosphere, 80, 256- 262. http://dx.doi.org/10.1016/j.chemosphere.2010.04.029
[22] García-Calzón, J.A., Muñíz-Alvarez, J.L. and López- Fonseca, J.M. (1999) Adsorption and two-dimensional condensation of thiabendazole at a mercury/solution in- terface. Journal of Colloid and Interface Science, 219, 144-148. http://dx.doi.org/10.1006/jcis.1999.6455
[23] Spanoghe, P., Ryckaert, B., Van-Gheluwe, C. and Van- Labeke, M.C. (2008) Fate of vinclozolin, thiabendazole and dimethomorph during storage, handling and forcing of chicory. Pest Management Science, 66, 126-131. http://dx.doi.org/10.1002/ps.1838
[24] USEPA Prevention, Pesticides and Toxic Substances (2002) Thiabendazole and salts. EPA-738-F-02-002.
[25] El-Nahhal, Y., Nir, S., Polubesova, T., Margulies, L. and Rubin, B. (1998) Leaching, phytotoxicity and weed con- trol of new formulations of alachlor. Journal of Agricul- tural Food Chemistry, 46, 3305-3313. http://dx.doi.org/10.1021/jf971062k
[26] Tomlin, C.D. (2000) The pesticides manual. 11th Edition, British Crop Protection Council.
[27] El-Nahhal, Y. and Lagaly, G. (2005) Salt effects on the adsorption of a pesticide on modified bentonite. Colloid and Polymer Science, 283, 968-974. http://dx.doi.org/10.1007/s00396-004-1244-7
[28] El-Nahhal, Y. and Safi, J. (2006) Bentonite for controlled release of linuron. Journal of Pest Control and Environ- mental Science, 14, 57-71.
[29] El-Nahhal, Y. and Safi, J. (2010) Adsorption of bromox- ynil to modified bentonite: Influence of pH, and temper- ature. Journal of Pesticide Science, 35, 333-338. http://dx.doi.org/10.1584/jpestics.G09-41
[30] Rodríguez, R., Picó, Y., Font, G. and Mañes, J. (2002) Analysis of thiabendazole and procymidone in fruits and vegetables by capillary electrophoresis-electrospray mass spectrometry. Journal of Chromatography A, 949, 359- 366. http://dx.doi.org/10.1016/S0021-9673(01)01395-4
[31] European Commission, Directorate-General Health and Comsumer Protection (2001) Thiabendazole. 7603/VI/97- Final, 22 March 2001.
[32] Rytwo, G. and Tavasi, M. (2003) Addition of a monova- lent cationic pesticide to improve efficacy of bipyridyl herbicides in Hulah Valley soils. Pest Management Sci- ence, 59, 1265-1270. http://dx.doi.org/10.1002/ps.732
[33] Giles, C.H., Mac-Ewan, T.H., Nakhwa, S.N. and Smith, D.A. (1960) System of classification of solution adsorp- tion isotherms, and its use in diagnosis of adsorption me- chanisms and in measurement of specific surface areas of solids. Chemical Society, 111, 3973-3993.
[34] Kishk, M.F., Abu-Sharar, M.T., Bakry M.N. and Abu- Donia B.M. (1979) Sorption-desorption characteristics of methyl parathion by clays. Archives of Environmental Contamination and Toxicology, 8, 637-645. http://dx.doi.org/10.1007/BF01054866