The inclusion complexation behavior of 2,5-Bis(5-tert-butyl-benzoxazol-2yl)-thiophene (UVOB) with native
β-cyclodextrin (
βCD) and
βCD-monochlorotriazinyl (
βCD-MCT) was evaluated by fluorescence spectroscopy. The association constant (K
s), stoichiometry,
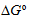
,

and
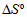
were evaluated at 25
℃ ± 0.1
℃ in phosphate buffer solution (pH = 10.5, 0.1 mo
•dm
–3) in order to find out the complex formation ability and stability. Fluorescence enhancement for UVOB and UVBNB with both CDs has been observed as a result of the complex formation. A stoichiometry 1:1 for UVOB in both CDs was observed; a stoichiometry 3:1 for UVBNB in both CDs has been observed. The
Ksvalues for UVOB were 4916 ± 137 M
–1 and 655 ± 19 M
–1 (acetone: water 90/10, v/v) with
βCD and
βCD-MCT, respectively. The
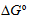
value obtained indicates a spontaneous and stable complex formation, but the complex
βCD-UVOB showed high
Ks value as an indicative of a high concentration of complex formed. Additionally,
Ks and thermodynamic parameters
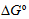
and

were evaluated in a commercial product UVBNB (UVOB, 13%, v/v). In aqueous solution, the values obtained were 2552 ± 115 and 1787 ± 75 M
–1 respectively. Complexation of UVOB with CDs is an interesting approach for utilization of UVOB in aqueous systems without the need of solvents and or surfactants used in commercial product (UVBNB).